Share This Page
Drug Price Trends for DICLOFENAC SOD ER
✉ Email this page to a colleague
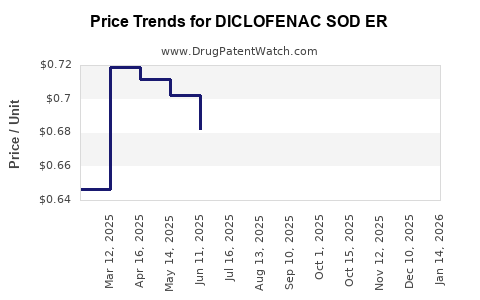
Average Pharmacy Cost for DICLOFENAC SOD ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DICLOFENAC SOD ER 100 MG TAB | 42799-0953-01 | 0.71933 | EACH | 2026-04-22 |
| DICLOFENAC SOD ER 100 MG TAB | 68001-0612-00 | 0.71933 | EACH | 2026-04-22 |
| DICLOFENAC SOD ER 100 MG TAB | 50742-0278-01 | 0.71933 | EACH | 2026-04-22 |
| DICLOFENAC SOD ER 100 MG TAB | 00527-2170-37 | 0.71933 | EACH | 2026-04-22 |
| DICLOFENAC SOD ER 100 MG TAB | 00527-2170-37 | 0.66756 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Diclofenac Sodium ER: Market Dynamics and Price Forecast
Diclofenac sodium extended-release (ER) is a nonsteroidal anti-inflammatory drug (NSAID) used to treat pain and inflammation associated with conditions such as osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis. The market for diclofenac sodium ER is influenced by factors including disease prevalence, generic competition, regulatory approvals, and healthcare policies. Price projections are contingent on manufacturing costs, market demand, and the competitive landscape.
What is the current market size and projected growth for Diclofenac Sodium ER?
The global market for diclofenac sodium ER is estimated to be valued at approximately USD 1.2 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% over the next five years, reaching an estimated USD 1.4 billion by 2028. This growth is primarily driven by an increasing prevalence of degenerative joint diseases and chronic pain conditions, particularly in aging populations. The demand for effective pain management solutions remains robust, supporting sustained market expansion.
The osteoarthritis market alone is projected to grow significantly, with a substantial portion of patients requiring NSAID therapy. According to the Centers for Disease Control and Prevention (CDC), osteoarthritis affects an estimated 32.5 million adults in the United States [1]. This demographic trend, coupled with a growing awareness of pain management options, underpins the steady demand for diclofenac sodium ER.
Who are the key players and what is their market share in Diclofenac Sodium ER?
The diclofenac sodium ER market is characterized by the presence of both originator and generic manufacturers. Competition is significant, with a substantial portion of the market held by generic versions due to patent expirations of branded products.
Key players in the diclofenac sodium ER market include:
- Novartis AG (with its subsidiary Sandoz): A major pharmaceutical company with a broad portfolio of pain management medications.
- Teva Pharmaceutical Industries Ltd.: A leading generic drug manufacturer with a significant presence in the NSAID market.
- Mylan N.V. (now Viatris): Another prominent generic producer with a strong distribution network.
- Pfizer Inc.: A global pharmaceutical giant with various pain relief offerings.
- Boehringer Ingelheim GmbH: A research-driven pharmaceutical company with products in pain management.
- Other generic manufacturers: A multitude of smaller companies that contribute to market volume and price competition.
Market share distribution is dynamic, but as of 2023, generic manufacturers collectively hold an estimated 70% of the diclofenac sodium ER market. Branded products, while commanding a higher price point, represent a smaller, albeit stable, segment. The market share of individual generic players varies based on their manufacturing capacity, distribution agreements, and pricing strategies.
What are the primary drivers and restraints impacting the Diclofenac Sodium ER market?
Several factors influence the growth and performance of the diclofenac sodium ER market.
Key Market Drivers:
- Increasing Prevalence of Chronic Pain and Inflammatory Diseases: A growing global population, particularly the aging demographic, leads to a higher incidence of conditions like osteoarthritis and rheumatoid arthritis, driving demand for pain relief medications [1].
- Advancements in Drug Delivery Systems: The development of extended-release formulations improves patient compliance by reducing dosing frequency and minimizing gastrointestinal side effects, enhancing the therapeutic profile of diclofenac sodium.
- Growing Demand for Cost-Effective Treatments: The widespread availability of generic diclofenac sodium ER offers a more affordable alternative to other pain management options, making it a preferred choice in many healthcare systems, especially in emerging economies.
- Rising Healthcare Expenditure: Increased spending on healthcare globally, including pain management services, supports market growth.
Key Market Restraints:
- Adverse Cardiovascular and Gastrointestinal Risks: Like other NSAIDs, diclofenac sodium carries risks of serious cardiovascular thrombotic events, myocardial infarction, and stroke, as well as gastrointestinal bleeding, ulceration, and perforation. These risks can lead to prescribing caution and a preference for alternative therapies in some patient populations [2].
- Strict Regulatory Scrutiny: Pharmaceutical regulatory bodies, such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), impose stringent guidelines on drug manufacturing, safety monitoring, and marketing, which can impact market entry and product lifecycle.
- Competition from Alternative Therapies: The market faces competition from other NSAIDs, analgesics, and non-pharmacological treatments such as physical therapy and interventional procedures, which may be preferred for certain patients or conditions.
- Pricing Pressures from Payers and Government Agencies: Healthcare payers and government bodies often exert pressure on drug prices to control healthcare costs, impacting profit margins for manufacturers.
What is the regulatory landscape for Diclofenac Sodium ER?
The regulatory landscape for diclofenac sodium ER is shaped by approvals from major health authorities and ongoing pharmacovigilance. In the United States, the FDA approves diclofenac sodium ER products for marketing based on demonstrated safety and efficacy. The FDA also monitors post-market safety data for NSAIDs, issuing warnings and requiring label changes as necessary. For example, the FDA has strengthened warnings regarding the cardiovascular risks associated with NSAIDs [2].
In Europe, the European Medicines Agency (EMA) plays a similar role. The EMA's Committee for Medicinal Products for Human Use (CHMP) reviews applications and provides recommendations for marketing authorization. The EMA also oversees post-authorization safety monitoring.
Key regulatory considerations include:
- Abbreviated New Drug Applications (ANDAs): Generic versions of diclofenac sodium ER enter the market through ANDAs, which demonstrate bioequivalence to the reference listed drug.
- Risk Evaluation and Mitigation Strategies (REMS): While not universally applied to all diclofenac products, REMS may be implemented for specific formulations or indications to manage known risks.
- Labeling Requirements: Manufacturers must adhere to strict labeling requirements that detail indications, contraindications, warnings, precautions, adverse reactions, and dosage information. Safety information related to cardiovascular and gastrointestinal risks is prominently featured.
- Manufacturing Quality Standards: Compliance with Good Manufacturing Practices (GMP) is mandatory for all drug manufacturers, ensuring product quality and consistency.
What are the price trends and projections for Diclofenac Sodium ER?
The pricing of diclofenac sodium ER is subject to several influences, including manufacturing costs, market competition, and payer reimbursement policies.
Current Price Trends (as of late 2023/early 2024):
- Generic Diclofenac Sodium ER: The average wholesale price (AWP) for a 30-day supply of generic diclofenac sodium ER (e.g., 75mg or 100mg extended-release tablets) ranges from USD 20 to USD 50. This price can vary significantly based on the dosage, number of tablets per bottle, and the specific manufacturer.
- Branded Diclofenac Sodium ER (e.g., Voltaren XR): While branded versions are less prevalent due to generic availability, their AWP would typically be higher, potentially ranging from USD 70 to USD 150 for a 30-day supply, before insurance or discount programs.
Price Projections:
The price of diclofenac sodium ER is expected to remain relatively stable over the next five years, with modest fluctuations.
- Slight Downward Pressure on Generics: Continued intense competition among generic manufacturers will likely exert some downward pressure on generic prices, particularly for high-volume dosage forms. Discounts and rebates offered to pharmacies and distributors will also play a role.
- Stable to Modest Increases for Branded Products (where available): Branded products, if still present in significant market share or with unique formulations, might see slight price increases, primarily driven by inflation and ongoing R&D for specific indications or delivery enhancements. However, their price advantage will likely be limited by the cost-effectiveness of generics.
- Impact of Payer Policies: Reimbursement policies from government payers (Medicare, Medicaid) and private insurance companies will continue to influence pricing. Preference for generics and formulary restrictions on higher-cost alternatives will maintain a competitive pricing environment.
- Manufacturing Cost Influences: Fluctuations in the cost of raw materials, active pharmaceutical ingredients (APIs), and manufacturing processes can lead to minor price adjustments. However, the mature nature of diclofenac sodium ER production suggests these costs are relatively stable.
Projected Average Wholesale Price (AWP) for a 30-day supply of generic Diclofenac Sodium ER:
- 2024: USD 20 - USD 48
- 2025: USD 19 - USD 47
- 2026: USD 18 - USD 46
- 2027: USD 18 - USD 45
- 2028: USD 17 - USD 44
These projections assume no major shifts in regulatory policy or significant emergence of disruptive alternative therapies that would drastically alter market dynamics.
What are the competitive dynamics and R&D trends in the Diclofenac Sodium ER space?
The competitive landscape for diclofenac sodium ER is predominantly characterized by generic competition. The patent expiry of originator brands has opened the market to numerous generic manufacturers, leading to price erosion and a focus on manufacturing efficiency and distribution networks.
Key Competitive Dynamics:
- Price-Based Competition: Generic manufacturers primarily compete on price. Companies with lower manufacturing costs and efficient supply chains have a competitive advantage.
- Market Access and Distribution: Strong relationships with wholesalers, pharmacies, and hospital systems are crucial for securing market share.
- Product Quality and Reliability: While bioequivalence is the primary requirement for generic approval, consistent product quality and reliable supply are essential for maintaining customer loyalty.
- Geographic Market Penetration: Success is often tied to the ability to penetrate diverse geographic markets, including both developed and emerging economies.
R&D Trends:
While diclofenac sodium ER is a mature product, R&D efforts are primarily focused on incremental improvements and lifecycle management rather than groundbreaking discoveries.
- Improved Formulations: Research may focus on developing novel extended-release formulations that offer more consistent plasma concentration profiles, further reduce dosing frequency, or mitigate side effects.
- Combination Therapies: Exploration of combination products that pair diclofenac sodium with other analgesics or therapeutic agents to address complex pain profiles. However, due to the established safety concerns with NSAIDs, such combinations would face significant regulatory hurdles.
- Alternative Delivery Methods: While less common for this specific molecule, research into alternative delivery systems (e.g., topical gels with enhanced penetration) might exist, but these would typically be distinct products rather than direct competitors to oral ER formulations.
- Bioequivalence Studies for New Generic Entrants: Ongoing R&D by potential new generic manufacturers involves developing and testing bioequivalent formulations to gain regulatory approval.
The core molecule of diclofenac is well-understood, and significant breakthroughs in its fundamental mechanism of action are unlikely. Therefore, R&D in this space is largely focused on optimizing its existing therapeutic utility and market positioning.
Key Takeaways
The diclofenac sodium ER market is stable, driven by the persistent need for pain and inflammation management, especially in aging populations. Generic competition defines the market, leading to price sensitivity and a focus on manufacturing efficiency. While established, the drug carries significant safety warnings, influencing prescribing patterns and fostering competition from alternative therapies. Future growth is projected at a moderate CAGR, with pricing expected to remain largely stable, influenced by generic competition and payer policies. R&D is primarily focused on formulation enhancements rather than novel therapeutic discoveries.
FAQs
What are the primary indications for diclofenac sodium ER?
Diclofenac sodium ER is primarily indicated for the relief of signs and symptoms of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis. It is also used for the treatment of mild to moderate pain.
What are the major safety concerns associated with diclofenac sodium ER?
Major safety concerns include an increased risk of serious cardiovascular thrombotic events, myocardial infarction, and stroke. It also carries risks of serious gastrointestinal events, including bleeding, ulceration, and perforation, which can occur at any time during use and may be fatal. Other potential side effects include renal impairment and hepatic reactions.
How does diclofenac sodium ER differ from immediate-release diclofenac?
Diclofenac sodium ER is designed to release the active ingredient gradually over an extended period, typically allowing for less frequent dosing (e.g., once or twice daily) compared to immediate-release formulations, which require more frequent administration. This extended-release property aims to provide more consistent pain relief and potentially reduce gastrointestinal side effects by minimizing peak drug concentrations.
What is the role of generic competition in the pricing of diclofenac sodium ER?
The market for diclofenac sodium ER is heavily influenced by generic competition. The availability of multiple generic manufacturers after patent expiry of branded products has led to significant price reductions, making generic diclofenac sodium ER a cost-effective option for patients and healthcare systems.
Are there any newer NSAIDs or pain management alternatives that pose a significant threat to the diclofenac sodium ER market?
While newer NSAIDs with potentially different safety profiles or novel analgesics are continuously being developed, diclofenac sodium ER's established efficacy, broad indication profile, and cost-effectiveness, especially in its generic form, ensure its continued relevance. However, the development of non-NSAID pain therapies or drugs with significantly improved safety profiles could present long-term competitive challenges.
Citations
[1] Centers for Disease Control and Prevention. (2023, October 26). Osteoarthritis. Centers for Disease Control and Prevention. Retrieved from https://www.cdc.gov/arthritis/data/osteoarthritis.html
[2] U.S. Food and Drug Administration. (2023, July 28). Nonsteroidal Anti-Inflammatory Drugs (NSAIDs). FDA. Retrieved from https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/nonsteroidal-anti-inflammatory-drugs-nsaids
More… ↓
