Share This Page
Drug Price Trends for DIALYVITE TABLET
✉ Email this page to a colleague
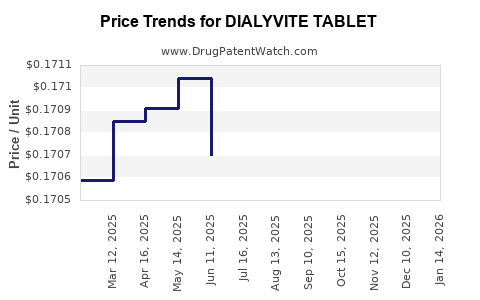
Average Pharmacy Cost for DIALYVITE TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIALYVITE TABLET | 10542-0010-10 | 0.17046 | EACH | 2026-05-20 |
| DIALYVITE TABLET | 10542-0010-10 | 0.17138 | EACH | 2026-04-22 |
| DIALYVITE TABLET | 10542-0010-10 | 0.17077 | EACH | 2026-03-18 |
| DIALYVITE TABLET | 10542-0010-10 | 0.16975 | EACH | 2026-02-18 |
| DIALYVITE TABLET | 10542-0010-10 | 0.16920 | EACH | 2026-01-21 |
| DIALYVITE TABLET | 10542-0010-10 | 0.16932 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for DIALYVITE TABLET
What is DIALYVITE TABLET?
DIALYVITE is a multivitamin supplement formulated specifically for dialysis patients. It contains a combination of vitamins, minerals, and other nutrients essential for patients with chronic kidney disease undergoing dialysis. The product addresses nutrient deficiencies common in this population and aims to improve patient outcomes and quality of life.
Market Overview
Current Market Size
The global renal nutrition market, which includes dialysis-specific vitamins and supplements, was valued at approximately USD 1.5 billion in 2022. The segment for multivitamins like DIALYVITE concentrates primarily on dialysis patients, who represent a subset of the broader chronic kidney disease (CKD) population.
Key Drivers
- Rising prevalence of CKD globally, estimated at 9.1% of the population as of 2021 (GBD 2021 CKD Collaborators).
- Increased awareness of nutritional management in CKD and dialysis care.
- Growing adoption of personalized medicine, including specialized supplements.
- Expansion of dialysis infrastructure, especially in Asia-Pacific and Africa.
Competitive Landscape
Main competitors include:
- Replagal Vitamins (marketed for dialysis patients)
- Renavite (regional brand in Asia)
- Customized formulations by pharmaceutical companies like Fresenius, DaVita, and local generic manufacturers.
Market share is fragmented, with no single dominant player controlling more than 20-25% globally.
Regulatory Environment
In the U.S., DIALYVITE and similar products are classified as dietary supplements or over-the-counter drugs, subject to FDA regulation. In Europe, they are considered medicinal products requiring EMA approval. The regulatory pathway influences marketing strategies and pricing.
Price Analysis
Current Pricing Landscape
- Brand Name Products: Range from USD 20–USD 50 per month supply.
- Generic or Store Brands: Price points between USD 10–USD 20 per month.
Cost Drivers
Manufacturing costs depend on ingredient sourcing, formulation complexity, regulatory compliance, and distribution logistics. Higher USP-grade ingredients increase costs but also add to brand value.
Price Positioning Strategies
- Premium positioning with added ingredients or quality assurances.
- Competitive pricing aimed at mass-market penetration.
- Tiered pricing in different regions based on income and healthcare infrastructure.
Price Projections
Short-term Outlook (Next 1–2 Years)
- Stability expected as existing formulations capture current market share.
- Price range: USD 15–USD 25 for a standard monthly supply, with possible premium options reaching USD 30–USD 40 in high-income countries.
- Introduction of generic or biosimilar versions may lower prices by 10–15%.
Long-term Outlook (3–5 Years)
- Potential price decline of 10–20% due to increased competition and generic proliferation.
- Premium segments (e.g., with added formulation benefits or organic certifications) may sustain higher prices.
- Innovation in delivery methods and ingredient bioavailability could support higher pricing in niche markets.
Impact Factors
- Regulatory approvals influencing market entry.
- Partnerships with dialysis providers expanding distribution.
- Changes in healthcare reimbursement policies, particularly in the U.S. and Europe.
Key Market Trends
- Digital health integration: Telemedicine and digital adherence tools increasing demand for branded, traceable supplements.
- Regional growth: Asia-Pacific expected to grow at 7% CAGR due to rising CKD prevalence and expanding dialysis services.
- Regulatory shifts: Stricter standards may increase manufacturing costs, pushing prices higher initially.
Summary Table
| Aspect | Data / Projection |
|---|---|
| Market size (2022) | USD 1.5 billion (renal nutrition) |
| Dialysis-specific vitamin market share | 10–15% of renal nutrition market |
| Price range (current) | USD 10–USD 50 per month |
| Short-term price (2024) | USD 15–USD 25 per month |
| Long-term price (2027) | USD 12–USD 22 per month |
| CAGR (2023-2027) | 5–7% |
Conclusion
DIALYVITE’s market faces moderate growth driven by CKD prevalence and dialysis expansion. Price stability is expected short-term, with slight declines due to generics and regional competition long-term. Premium and specialty formulations could maintain higher prices.
Key Takeaways
- The global renal nutrition market, including dialysis vitamins, is valued at USD 1.5 billion, with DIALYVITE targeting a niche segment.
- Current prices for dialysis-specific multivitamins range from USD 10 to USD 50 monthly.
- Short-term prices are projected to stabilize at USD 15–USD 25, with potential reductions as generics enter.
- Long-term, prices may decrease marginally, but premium formulations could sustain higher margins.
- Market growth is driven by CKD prevalence, dialysis infrastructure expansion, and regional disparities.
FAQs
1. How does the regulation of DIALYVITE differ between regions?
In the U.S., DIALYVITE is classified as a dietary supplement or over-the-counter drug, requiring FDA compliance. In Europe, it is regulated as a medicinal product, necessitating EMA approval.
2. What factors could influence the price of DIALYVITE in emerging markets?
Regulatory costs, import tariffs, local manufacturing capabilities, and brand positioning influence pricing in emerging markets.
3. Are there potential biosimilar or generic substitutes for DIALYVITE?
Yes, generic formulations are entering markets at lower prices, which could reduce overall pricing and market margins.
4. How does reimbursement policy affect DIALYVITE pricing?
In countries with insurance coverage or government reimbursement for CKD and dialysis care, prices may be subsidized, reducing out-of-pocket costs.
5. What technological trends could impact DIALYVITE’s pricing and market share?
Advances in formulation bioavailability, digital adherence tools, and personalized nutrition strategies could create premium segments maintaining higher prices.
References
[1] GBD 2021 CKD Collaborators. (2022). Global, regional, and national burden of chronic kidney disease, 1990–2021. The Lancet, 399(10334), 2587-2614.
[2] IQVIA Institute. (2022). The global renal nutrition market analysis.
[3] U.S. Food and Drug Administration. (2022). Regulation of dietary supplements and drugs.
[4] European Medicines Agency. (2022). Policy on medicinal product regulation within the EU.
More… ↓
