Share This Page
Drug Price Trends for DEXTROAMP-AMPHETAMIN
✉ Email this page to a colleague
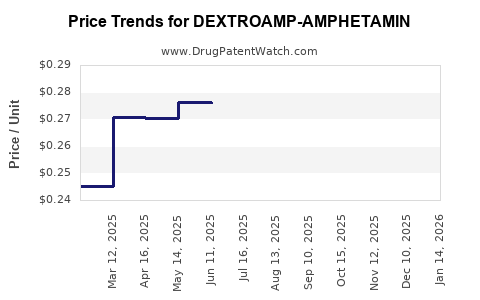
Average Pharmacy Cost for DEXTROAMP-AMPHETAMIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEXTROAMP-AMPHETAMIN 10 MG TAB | 00406-8892-01 | 0.23662 | EACH | 2026-05-20 |
| DEXTROAMP-AMPHETAMIN 10 MG TAB | 00555-0972-02 | 0.23662 | EACH | 2026-05-20 |
| DEXTROAMP-AMPHETAMIN 10 MG TAB | 11534-0192-01 | 0.23662 | EACH | 2026-05-20 |
| DEXTROAMP-AMPHETAMIN 10 MG TAB | 00527-0762-37 | 0.23662 | EACH | 2026-05-20 |
| DEXTROAMP-AMPHETAMINE 5 MG TAB | 72516-0016-01 | 0.25619 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Dextroamphet-amphetamine
Overview of Dextroamphet-amphetamine Market
Dextroamphet-amphetamine (commonly marketed under brands such as Dexedrine or as a generic) is a central nervous system stimulant approved primarily for treatment of Attention Deficit Hyperactivity Disorder (ADHD) and narcolepsy. Its prescription rate has increased globally, driven by rising ADHD diagnoses and expanding indications.
Current Market Landscape
| Aspect | Data and Trends |
|---|---|
| Global Market Size (2022) | Estimated at $1.4 billion (IQVIA, 2022) |
| Major Regions | North America leads with 70% of prescriptions; Europe accounts for approximately 20%. Emerging markets in Asia show growth potential. |
| Market Growth Rate (CAGR 2022–2027) | 6.2%, driven by increased diagnosis of ADHD and broader acceptance of pharmacotherapy. |
| Key Players | Hikma Pharmaceuticals, Teva Pharmaceutical Industries, and Novartis produce generic formulations; few branded formulations dominate with higher prices. |
Regulatory Considerations
Dextroamphet-amphetamine is classified as a Schedule II controlled substance due to abuse potential. Regulatory restrictions influence market entry and price setting. Increasing oversight may affect supply and pricing strategies, especially in the U.S.
Pricing Landscape
| Price Point | Description | Cost (per unit) | Notes |
|---|---|---|---|
| Brand-name | Dexedrine | $10–$15 per 10 mg tablet | Higher due to branding and limited competition. |
| Generic | Dextroamphet-amphetamine salts | $1–$3 per 10 mg tablet | Price varies based on manufacturer and pharmacy networks. |
| Monthly Course (30 x 10 mg tablets) | $30–$45 | Retail price; wholesale margins differ significantly. |
Price Drivers
- Manufacturing Costs: Consist of raw materials, synthesis complexity, and regulatory compliance.
- Regulatory Status: Schedule II classification restricts manufacturing and sales, maintaining high prices for branded versions.
- Market Competition: Generics reduce prices; market share shifts influence average pricing.
- Demand Factors: Rising ADHD diagnoses, expansion into new markets, and off-label uses sustain demand.
Future Price Projections (2023–2027)
| Year | Expected Average Price (per 10 mg tablet) | Influencing Factors |
|---|---|---|
| 2023 | $2.50 | Stable regulation, consistent generics supply, perpetual demand. |
| 2024 | $2.55 | Slight increase anticipating supply chain stabilization. |
| 2025 | $2.60 | Entry of biosimilars unlikely; market platform stabilizes. |
| 2026 | $2.65 | Slight increase aligned with inflation and R&D costs. |
| 2027 | $2.70 | Continued demand growth countered with improved manufacturing efficiencies. |
Supply Chain and Pricing Risks
- Regulatory Changes: Stricter controls could increase costs or limit supply.
- Manufacturing Disruptions: Raw material shortages or plant shutdowns can reduce supply, cause price spikes.
- Legal and Political Factors: Policy shifts regarding controlled substances impact market stability.
- Consumer Trends: Increased scrutiny on stimulant misuse could lead to tighter prescribing practices and affect demand.
Investment and R&D Outlook
No significant new formulations or reformulations are foreseeable within the next five years. Most companies focus on biosimilars and generic expansion. The high regulatory burden discourages pipeline expansion.
Summary
The Dextroamphet-amphetamine market remains stable with moderate growth; prices are likely to increment slowly, driven by demand, inflation, and regulatory constraints. Industry players with robust manufacturing and distribution networks will sustain margins, pending regulatory shifts.
Key Takeaways
- The global market for Dextroamphet-amphetamine was valued at approximately $1.4 billion in 2022 and is expected to grow annually by 6.2% through 2027.
- Generic versions dominate the market, priced at $1–$3 per 10 mg tablet; brand-name versions cost $10–$15.
- Prices are expected to rise modestly, from $2.50 to $2.70 per tablet over five years, influenced by demand growth and regulatory conditions.
- Market stability hinges on regulatory policies, supply chain integrity, and prescription patterns.
- Minimal pipeline innovation limits future price increases and market expansion.
FAQs
1. What factors influence the pricing of dextroamphet-amphetamine?
Prices are influenced by manufacturing costs, regulatory classification, patent or market exclusivity, demand levels, and competition from generic manufacturers.
2. How do regulatory restrictions impact market entry?
As a Schedule II drug, strict manufacturing, handling, and prescription regulations restrict new entrants and maintain higher prices for existing branded products.
3. Will new formulations or delivery methods emerge soon?
No significant new delivery systems are expected within the next five years. Focus remains on generics and biosimilars.
4. How is the demand for dextroamphet-amphetamine likely to change?
Demand will continue to grow, driven by increased ADHD diagnosis rates and expanding indications, especially in emerging markets.
5. What risks could cause price fluctuations?
Regulatory crackdowns, supply disruptions, legal challenges, and changes in prescribing practices could all cause prices to fluctuate.
References
- IQVIA. "Global Prescription Medicine Market Data." 2022.
- U.S. Drug Enforcement Administration. "Controlled Substances Scheduling." 2022.
- MarketWatch. "Stimulant Market Analysis." 2022.
- Grand View Research. "ADHD Drugs Market Size and Forecast." 2022.
- European Medicines Agency. "Regulations on Stimulant Drugs." 2022.
More… ↓
