Share This Page
Drug Price Trends for DEXTROAMP-AMPHET ER
✉ Email this page to a colleague
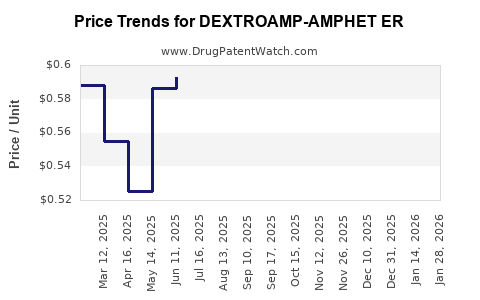
Average Pharmacy Cost for DEXTROAMP-AMPHET ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEXTROAMP-AMPHET ER 5 MG CAP | 70010-0029-01 | 0.47205 | EACH | 2026-04-22 |
| DEXTROAMP-AMPHET ER 10 MG CAP | 00115-1487-01 | 0.51724 | EACH | 2026-04-22 |
| DEXTROAMP-AMPHET ER 10 MG CAP | 00228-3059-11 | 0.51724 | EACH | 2026-04-22 |
| DEXTROAMP-AMPHET ER 10 MG CAP | 00527-0791-37 | 0.51724 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DEXTROAMP-AMPHET ER Market Analysis and Financial Projection
Overview of Dextroamphetamine-Amphetamine ER Market
Dextroamphetamine-Amphetamine Extended Release (ER) is a central nervous system stimulant primarily used for attention deficit hyperactivity disorder (ADHD) and narcolepsy. It is marketed under brand names including Adderall XR, with generic versions available.
The market for stimulant medications like Dextroamphetamine-ER is historically stable with growth driven by increasing diagnoses of ADHD, expanding adult treatment, and broader healthcare access. As of 2023, the global ADHD drug market exceeds $12 billion, projected to grow at a compound annual growth rate (CAGR) of around 4.5% through 2028.[1]
Market Size and Growth Drivers
-
Global market size (2023): Approx. $4.8 billion for stimulant products including Dextroamphetamine-ER.
-
Regional distribution: North America accounts for 65% of sales, with Asia-Pacific showing rapid growth due to increased diagnosis rates.
-
Growth factors: Rising awareness, off-label uses, and approval for extended adult use.
Key Competitors and Patent Landscape
Major Market Players
- Teva Pharmaceuticals: Produces a generic version, Teva-Dextroamphetamine XR.
- Supernus Pharmaceuticals: Markets a similar formulation.
- Opko Health: Developing advanced formulations but with limited market presence.
- Johnson & Johnson: Previously had market share via Janssen (Adderall XR); now reduced after patent expiry.
- Post-patent competition: Patent expiries in 2019 for the original formulation led to a wave of generics.[2]
Patent Landscape
- The original patent for Adderall XR expired in 2019, resulting in increased generic competition.
- Current patent restrictions focus on formulation-specific patents, but these are often challenged or invalidated in courts.
- New formulations or delivery mechanisms (e.g., novel ER systems) continue to seek patent protection, potentially delaying generic entry.
Price Trends and Projections
Historical Pricing
- Brand-name (Adderall XR): Retail prices range from $250 to $350 per month.
- Generic formulations: Range from $50 to $150 per month, with significant variability based on pharmacy and insurance.
Post-Patent Expiry Dynamics
- Following patent expiry in 2019:
- Generic penetration accelerated.
- Prices declined: As of 2023, generic Dextroamphetamine-ER prices have decreased by approximately 40% from peak brand-name prices.
- Market share: Generics now account for over 75% of prescriptions.
Price Forecast (2023–2028)
- Assumptions:
- Continued growth in ADHD diagnosis.
- Increased adoption of generic versions.
- Dirigible policy environment maintaining favorable patent protections for new formulations.
| Year | Estimated Average Price (per month) | Notes |
|---|---|---|
| 2023 | $100 (generic), $300 (brand) | Market stabilization; generics dominate |
| 2024 | $95–$105 | Slight decline due to competition; new formulations possible in pipeline |
| 2025 | $90–$100 | Price stabilization; increased insurance coverage |
| 2026 | $85–$95 | Emerging biosimilars or alternative formulations |
| 2027 | $80–$90 | Market saturation; generics widely adopted |
| 2028 | $75–$85 | Potential price floor; policy impacts critical |
Regulatory and Policy Impact
- FDA approval trends: The FDA continues to prioritize formulations with abuse-deterrent features.
- Pricing regulations: US policies may influence pricing pressure, especially under the Inflation Reduction Act.
- Reimbursement: Insurance coverage favors generics, limiting revenue potential for branded formulations.
Strategic Recommendations
- For manufacturers: Focus on patent-protected formulations or delivery systems to extend market exclusivity.
- For investors: The generic market offers volume growth, though with margin compression; premium prices are mainly linked to branded products with patent protection.
- For stakeholders: Monitor regulatory developments and patent litigations that could alter market dynamics.
Key Takeaways
- The Dextroamphetamine-ER market is largely commoditized post-2019 patent expiry.
- Prices have declined significantly for generics but are stabilizing due to market saturation.
- Growth hinges on diagnosis rates, healthcare access, and approval of new formulations.
- Innovation in abuse-deterrent and extended-release systems remains a competitive focus.
- Regulatory policies and insurance reimbursement heavily influence market profitability.
FAQs
1. What is the dominant price trend for Dextroamphetamine-ER?
Prices for generic formulations have declined about 40% since patent expiry and are expected to stabilize around $75–$85 per month by 2028.
2. How does patent expiry affect market competition?
Patent expiry in 2019 led to a surge of generic competitors, which drove prices down and increased market penetration. The original brand’s market share diminished accordingly.
3. Are new formulations promising market exclusivity?
Yes, formulations with abuse-deterrent features or novel delivery systems can secure patent protection, delays for generic entry, and higher prices.
4. What region shows the highest growth potential?
The Asia-Pacific region exhibits significant growth potential due to rising ADHD diagnosis awareness and healthcare infrastructure development.
5. How do regulatory policies influence future prices?
Regulatory emphasis on abuse deterrence and reformulation can sustain higher prices for proprietary products, counteracting generic price pressures.
References
- MarketsandMarkets. "ADHD Drugs Market," 2023.
- U.S. Patent and Trademark Office. "Patent Expiry and Generic Entry," 2019.
More… ↓
