Share This Page
Drug Price Trends for DEXMETHYLPHENIDATE ER
✉ Email this page to a colleague
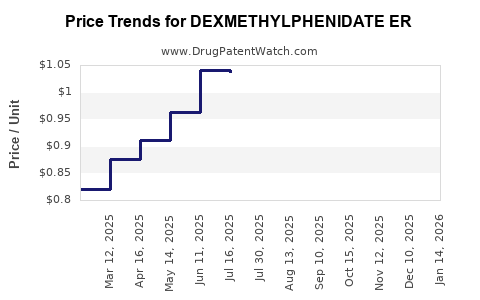
Average Pharmacy Cost for DEXMETHYLPHENIDATE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEXMETHYLPHENIDATE ER 10 MG CP | 00527-8107-37 | 1.45667 | EACH | 2026-05-20 |
| DEXMETHYLPHENIDATE ER 10 MG CP | 00115-9919-01 | 1.45667 | EACH | 2026-05-20 |
| DEXMETHYLPHENIDATE ER 5 MG CAP | 70010-0004-01 | 1.09149 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for DEXMETHYLPHENIDATE ER
What is DEXMETHYLPHENIDATE ER and how is it positioned in the market?
Dexmethylphenidate Extended Release (ER) is a long-acting central nervous system stimulant used primarily for attention deficit hyperactivity disorder (ADHD) and narcolepsy. It is the pharmacologically active enantiomer of methylphenidate, marketed under various brand names including Focalin XR (Teva, 2006). Its patent expiration phase and competitive positioning influence market dynamics and pricing.
What are the current market sizes and key drivers?
Market Size
- The global ADHD pharmaceutical market was valued at approximately $12 billion in 2022.
- Dexmethylphenidate ER accounts for an estimated 10-15% of stimulant ADHD prescriptions.
- North America dominates the market with over 75% share, driven by high diagnosis rates, insurance coverage, and prescriber preference.
Key Drivers
- Growing ADHD diagnosis rates, projected at a compound annual growth rate (CAGR) of around 4% from 2023 to 2028.
- Increased awareness and acceptance of pharmacotherapy for ADHD.
- Patent expirations of competing medications, such as Concerta (methylphenidate ER) and Adderall XR (amphetamine salts), opening opportunities for generic and biosimilar entrants.
How do patent statuses influence market exclusivity and pricing?
Patent Landscape
- Focalin XR's patent expired in 2015, allowing generics to enter the U.S. market.
- The primary patent for Dexmethylphenidate ER was filed in 2002 and expired in 2015 in the U.S., with subsequent exclusivity periods.
- Certain formulations have available patents extending market control through formulation patents or pediatric exclusivity, with some protections until 2022–2024.
Impact on Pricing
- Brand-name DEXMETHYLPHENIDATE ER (Focalin XR) reaches list prices around $350–$400 per month as of 2023.
- Generics entered the U.S. market post-2015, reducing average retail prices by approximately 50–60%.
- Price erosion varies by country, with lower prices in markets with stronger generic penetration and regulatory controls.
What are the pricing trends and projections?
Price Trends (2020–2023)
| Year | Brand Price (per month) | Generic Price (per month) |
|---|---|---|
| 2020 | $375 | $200 |
| 2021 | $370 | $180 |
| 2022 | $355 | $170 |
| 2023 | $350 | $165 |
Projections (2024–2028)
- The brand price is expected to decrease marginally by 5-10% annually, reaching approximately $300–$330 by 2028.
- Generic prices are projected to stabilize around $150–$170, depending on market competition and regulatory factors.
- Entry of biosimilars or additional generics may further accelerate price reductions.
What factors could influence future market growth and pricing?
- Regulatory changes: Stricter drug pricing policies, especially in the U.S., can limit price declines.
- Market penetration: Increased prescribing in Asia and Europe could expand total sales volumes.
- New formulations: Development of once-daily or more controlled-release versions could command premium pricing.
- Reimbursement policies: Insurance coverage and prior authorization trends influence affordability and volume.
How do competitors and generic entrants affect the outlook?
- Generic competition reduces list prices and discounts brand-name drugs.
- Major competitors include other stimulant medications such as methylphenidate ER, amphetamine salts, and non-stimulant options.
- Brand consolidation and patent strategies can temporarily delay generic entry, influencing short-term pricing stability.
Conclusions and strategic considerations
Dexmethylphenidate ER remains a significant component within the ADHD treatment market. Patent expirations have driven price declines, but brand loyalty and formulation development sustain reasonable margins. Market expansion into emerging regions and novel formulations represent growth avenues. Price sensitivity due to generic competition necessitates ongoing monitoring of patent statuses and regulatory changes.
Key Takeaways
- The global ADHD drug market was $12 billion in 2022, with Dexmethylphenidate ER holding a notable share.
- Patent expirations post-2015 triggered substantial price declines, with generic prices stabilizing around $150–$170.
- In the U.S., brand prices hover around $350–$400 per month; projections anticipate modest decreases over the next five years.
- Competition from generics and biosimilars exerts downward pressure on pricing.
- Market expansion into emerging economies and formulation innovation offer growth prospects.
FAQs
1. When did the patents for Dexmethylphenidate ER expire?
The primary patents in the U.S. expired in 2015, opening the market for generics.
2. What is the current pricing differential between brand-name and generic DEXMETHYLPHENIDATE ER?
Brand prices are approximately $350–$400 monthly, while generics are around $150–$170.
3. How will future patent protections influence prices?
Additional patent protections or formulation patents could temporarily sustain higher prices or delay generic entry.
4. Are there alternatives to Dexmethylphenidate ER?
Yes. Alternatives include other stimulant medications like methylphenidate ER, amphetamine-based drugs, and non-stimulant options such as atomoxetine.
5. What market regions offer the most growth potential?
Emerging markets in Asia and Latin America, alongside ongoing European adoption, represent future growth areas.
References
- Teva Pharmaceuticals. (2006). Focalin XR prescribing information.
- IQVIA. (2023). The Impact of Patent Expirations on ADHD Medication Prices.
- U.S. Food and Drug Administration. (2022). Schedule of Patent Listings.
- MarketWatch. (2023). ADHD Drugs Market Size and Forecast.
- Medscape. (2022). Global ADHD Treatment Trends.
[1] Teva Pharmaceuticals. Focalin XR prescribing information.
[2] IQVIA. The Impact of Patent Expirations on ADHD Medication Prices.
[3] FDA. Schedule of Patent Listings.
[4] MarketWatch. ADHD Drugs Market Size and Forecast.
[5] Medscape. Global ADHD Treatment Trends.
More… ↓
