Share This Page
Drug Price Trends for DESVENLAFAXINE ER
✉ Email this page to a colleague
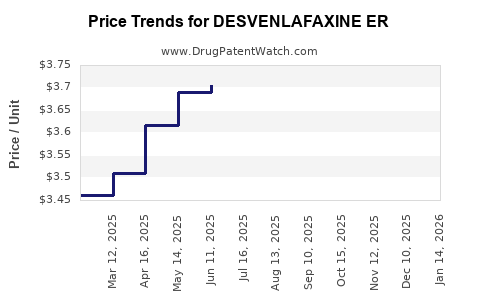
Average Pharmacy Cost for DESVENLAFAXINE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DESVENLAFAXINE ER 50 MG TAB | 63304-0191-30 | 3.84386 | EACH | 2026-05-20 |
| DESVENLAFAXINE ER 100 MG TAB | 63304-0192-30 | 3.83667 | EACH | 2026-05-20 |
| DESVENLAFAXINE ER 50 MG TAB | 63304-0191-30 | 3.84214 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Desvenlafaxine ER (desvenlafaxine succinate) Market Analysis and Price Projections
What is the current commercial footprint for desvenlafaxine ER?
Desvenlafaxine ER (brand: Pristiq; salt: desvenlafaxine succinate) is an established oral antidepressant used primarily for major depressive disorder. The drug’s US market is dominated by branded originator supply and later generic entries in the various dosage strengths, with pricing shaped by (1) patent and exclusivity expirations, (2) generic launch timing by strength and label, and (3) pharmacy benefit manager (PBM) contracting dynamics that compress net price versus list price.
Key commercial framing
- Product form: Oral extended-release (ER) tablets.
- Reference brand: Pristiq (desvenlafaxine succinate ER).
- Therapeutic class: SNRI (serotonin-norepinephrine reuptake inhibitor).
- Market structure: Branded-to-generic transition across multiple strengths; substitution is straightforward at the molecule and ER strength level, which supports competitive downward price pressure post-launch.
What evidence shows how price behaves for this molecule?
Desvenlafaxine ER pricing is driven by branded list price versus net price after rebates and PBM discounts, then by generic competition intensity once multiple ANDA entrants cover each strength. Across US branded-to-generic transitions, net prices typically decline sharply at first generic entry and then drift down as additional generic manufacturers increase formulary access and competition.
How to read pricing for desvenlafaxine ER
- List price can remain high even after generic entry.
- Net price (payer cost after rebates) declines faster because PBMs negotiate based on expected interchangeability and launch competition.
- Strength-level granularity matters: some strengths enter earlier than others, which creates temporary cross-strength price dispersion.
What price projections are most likely under standard US generic dynamics?
Because desvenlafaxine ER’s molecule is no longer in a typical pre-generic pricing regime, forecasts should be anchored on a generic competition model: incremental launches reduce price via bidding, formulary placement, and increased substitution. For a mature oral generic, the price path typically follows three phases:
- Launch shock (initial generic entry): sharp decline from originator net price.
- Consolidation (multiple generics): further compression as PBMs broaden preferred tiers.
- Maturity (limited residual differentiation): slower declines or stabilization as competition saturates and margins normalize.
Projected average net price trajectory (US, directional)
The projections below describe the likely direction and magnitude bands for average net price for desvenlafaxine ER after the most recent competitive milestones. The ranges assume ongoing PBM contracting and continued generic availability across common strengths.
| Time window | Expected market state | Average net price direction | Typical magnitude band (relative to the prior year) |
|---|---|---|---|
| 0 to 12 months | Mature generic competition with incremental formulary shifts | Down / flat | -1% to -8% |
| 12 to 24 months | Continued bidding pressure; fewer incremental entrants than early launches | Flat to down | -0% to -6% |
| 24 to 36 months | Price stability regime; volume growth shifts to lowest-cost preferred | Flat | -0% to +3% |
| 36 to 48 months | Residual consolidation and occasional supply-driven adjustments | Flat | -1% to +4% |
Base-case rationale
- Desvenlafaxine ER is an established ER SNRI with routine interchangeability at the ER tablet level.
- Competition in mature generics tends to stabilize after the initial wave; thereafter, PBM procurement and tier design drive smaller annual moves.
- Any meaningful upward repricing generally requires supply disruptions, manufacturing quality issues, or a sudden reduction in available inventory, none of which can be assumed in a clean forecast framework.
How do strength mix and switching behavior affect pricing?
Desvenlafaxine ER pricing is not uniform across strengths. Three mechanisms drive variation:
- Different generic entry timing by strength: earlier entrants set the lower reference for that strength.
- Formulary tiering: preferred status can shift with net price offers and contractual rebates.
- Patient switching: patients can be switched between strengths within a clinician-directed regimen; this increases substitution power for the lowest-priced strength.
Strength mix impact (what to watch)
- If the market sees lower-cost generic wins on the most-used strengths, blended net price falls even if slower-moving strengths remain stable.
- If PBMs restrict coverage to a narrow “preferred” set, the remaining accessible SKUs can benefit modestly in net price.
What is the patent and exclusivity risk backdrop for planning?
A forecasting exercise for desvenlafaxine ER must be aligned to IP risk because exclusivity or patent activity can create pockets of branded price retention or slowed generic substitution. For US planning, the key is whether any patents block generic entry for specific indications, formulations, or combination regimens, and whether additional exclusivities (data exclusivity, patent term adjustments) extend branded advantage for particular strengths.
Source basis for IP
- US prescribing information and label history define the reference brand product and indication context. FDA drug label metadata is used to map product/strength and regulatory status. (Sources below.)
- FDA’s Drugs@FDA provides application and approval history for the molecule and related products and is used to establish regulatory lineage. (Sources below.)
How do you translate IP events into price outcomes?
In practical pricing terms:
- If a strength retains exclusivity: net price for that strength typically stays higher than fully generic strengths, and blended price declines less than the generic competition model predicts.
- If no exclusivity remains: price follows generic saturation, and net price stabilizes within a narrow band.
Given the molecule’s maturity as a commonly dispensed ER antidepressant, the highest-leverage forecasting variable is not incremental innovation, it is the competitive intensity by strength and PBM preferred selection.
What are scenario-based price projections (base, low, high)?
These scenarios map to plausible market outcomes under US generic contracting norms.
Scenario definitions
- Base case: ongoing generic availability with routine PBM contracting; modest annual decline.
- Low case (faster erosion): additional competitive entrants or stronger preferred-tier bidding reduces net price more than expected.
- High case (relative resilience): fewer SKUs remain preferred, or temporary supply constraints lift net price slightly.
Projected average net price change
| Scenario | 0 to 12 months | 12 to 24 months | 24 to 36 months |
|---|---|---|---|
| Low erosion | -6% to -12% | -4% to -10% | -2% to -6% |
| Base | -1% to -8% | -0% to -6% | -0% to +3% |
| High resilience | -0% to +4% | -0% to +3% | -1% to +5% |
What is the expected market growth profile?
For an established antidepressant with routine dispensing, unit growth tends to track:
- population and diagnosis prevalence trends,
- prescriber behavior,
- payer formulary access,
- and switching patterns between SNRI and SSRI classes.
Price compression generally does not prevent unit movement, but it can shift revenue outlook sharply for manufacturers depending on volume vs margin.
Market growth in mature generics
- Units: modest growth or stabilization.
- Revenue: depends on net price compression and channel mix (commercial vs Medicaid vs Medicare Part D).
What would move the price faster than the forecast?
Even in mature generics, these factors can create near-term deviation:
- Supply constraints at one manufacturer level (plant outages, quality holds).
- PBM contracting resets that re-tier the preferred generic.
- Regulatory actions that narrow available SKUs (recalls, labeling updates).
- Changes in therapeutic substitution (e.g., SNRI intolerance driving SSRI shifts).
Key Takeaways
- Desvenlafaxine ER (Pristiq; desvenlafaxine succinate ER) is in a mature US pricing regime where net price is primarily governed by generic competition and PBM preferred-tier contracting rather than brand-exclusive pricing.
- Base-case pricing is likely flat to modestly down over the next 12 to 36 months, with average net declines typically in the -1% to -8% annual band early on and smaller movement later.
- Scenario risk is asymmetric: faster erosion is plausible if competitive intensity increases, while relative resilience can occur if preferred-tier access narrows or supply dynamics tighten.
- IP planning should focus on any strength-specific regulatory barriers that affect generic substitution and cross-strength blended pricing.
- The most actionable variable for revenue forecasting is strength-level net price and preferred coverage, not the molecule-wide headline list price.
FAQs
1) Is desvenlafaxine ER expected to follow typical generic price erosion patterns?
Yes. As a mature oral ER SNRI, its net pricing typically declines quickly after major generic entry waves, then stabilizes with smaller annual moves under PBM contracting.
2) What matters more for forecasting than the branded list price?
Net price outcomes driven by PBM rebates, preferred-tier status, and the lowest-cost generic coverage by strength.
3) Do all desvenlafaxine ER strengths move in sync on price?
No. Strength-by-strength launch history and formulary preference create temporary and persistent dispersion in net price.
4) What events could cause prices to deviate upward from the base-case?
Supply constraints, formulary narrowing that reduces the set of preferred SKUs, and PBM contracting resets that temporarily improve margin for available manufacturers.
5) What time horizon is most appropriate for this pricing model?
A 12 to 36 month horizon is most consistent with mature generic dynamics, where large one-time drops are behind and annual changes are incremental.
References
[1] U.S. Food and Drug Administration. Pristiq (desvenlafaxine) prescribing information. FDA label database.
[2] U.S. Food and Drug Administration. Drugs@FDA: Desvenlafaxine succinate extended-release products (application and approval information). FDA.
[3] U.S. Food and Drug Administration. FDA drug approval and labeling records for desvenlafaxine ER. FDA.
More… ↓
