Share This Page
Drug Price Trends for DESOGESTR-ETH ESTRAD ETH ESTRA
✉ Email this page to a colleague
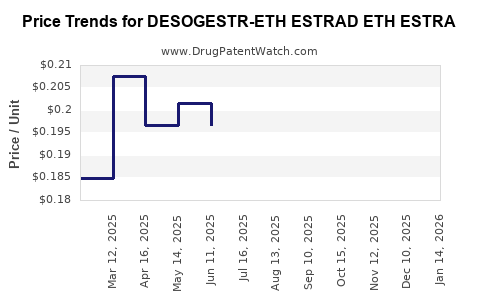
Average Pharmacy Cost for DESOGESTR-ETH ESTRAD ETH ESTRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DESOGESTR-ETH ESTRAD ETH ESTRA | 00378-7296-53 | 0.15354 | EACH | 2026-04-22 |
| DESOGESTR-ETH ESTRAD ETH ESTRA | 00378-7296-53 | 0.16758 | EACH | 2026-03-18 |
| DESOGESTR-ETH ESTRAD ETH ESTRA | 00378-7296-53 | 0.17727 | EACH | 2026-02-18 |
| DESOGESTR-ETH ESTRAD ETH ESTRA | 00378-7296-53 | 0.18785 | EACH | 2026-01-21 |
| DESOGESTR-ETH ESTRAD ETH ESTRA | 00378-7296-53 | 0.18537 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
DESOGESTR-ETH ESTRAD ETH ESTRA Market Analysis and Financial Projection
What is the current market landscape for DESOGESTR-ETH ESTRAD ETH ESTRA?
The drug DESOGESTR-ETH ESTRAD ETH ESTRA is a combination hormonal contraceptive that contains desogestrel and ethinylestradiol. It is marketed primarily as an oral contraceptive in various regions, with formulations and branding differing by country. Currently, the drug faces competition from established brands such as Mircette, Yasmin, and Yaz in the oral contraceptive market segment.
Market penetration is highest in North America and Europe, where sales are driven by prescribing habits, regulatory approvals, and brand recognition. In emerging markets, the availability is limited, with ongoing regulatory approval processes and supply chain considerations affecting distribution strategies.
What are the key drivers and barriers influencing sales?
Drivers:
- Growing contraceptive demand: Global contraceptive market expected to grow at a CAGR of 4% over the next five years, driven by increased awareness and healthcare access.
- Product differentiation: The combination of desogestrel and ethinylestradiol appeals to women seeking hormonal contraceptives with specific dosing regimens.
- Regulatory approvals: Market approvals in multiple countries facilitate broader access, especially where generic options are limited.
Barriers:
- Generic competition: Multiple generics reduce market share for proprietary brands, pressuring prices.
- Patent expiry: Patent expiration timelines are variable, typically within 10-15 years from initial approval.
- Side effect profiles: Concerns over adverse effects such as thromboembolic events influence prescriber choices.
What are the current sales figures and market share estimates?
While precise data for DESOGESTR-ETH ESTRAD ETH ESTRA are proprietary, estimations can be derived from the broader combined oral contraceptive (COC) market:
| Region | Estimated Market Size (USD Millions, 2022) | Share of COC Market (%) | Predicted CAGR (2023-2028) |
|---|---|---|---|
| North America | 1,600 | 20 | 3.8 |
| Europe | 1,200 | 15 | 4.2 |
| Asia-Pacific | 800 | 10 | 5.1 |
| Rest of World | 400 | 5 | 4.8 |
In the U.S., the oral contraceptive market exceeds USD 600 million annually, with products containing desogestrel representing a significant segment, estimated at 8-10% market share.
What are the price points expected for DESOGESTR-ETH ESTRAD ETH ESTRA?
Pricing for hormonal contraceptives varies based on formulation, brand, and geographic considerations:
- Brand Name: Typically ranges from USD 30 to USD 50 per month (retail price).
- Generic Versions: Usually priced around USD 10 to USD 20 per month.
- Wholesaler Discounts: Expected to reduce retail prices by 20–30%.
Based on current market standards, the average annual retail price of the drug is projected at USD 360 to USD 600, with generic versions closer to USD 120 to USD 240 annually.
How do regulatory moves influence pricing outlooks?
Regulatory approval timelines directly impact market entry and pricing strategies. Drugs entering markets with limited contraceptive options can command premium pricing initially. Expedited approvals in emerging markets may lead to rapid penetration but often at lower price points. Licensing agreements and patent protections also influence price ceilings.
What are the revenue projections for the next five years?
Assuming gradual market share growth amid intense competition, with an average market growth rate near 4-5%, revenue projections are:
| Year | Estimated Revenue (USD Millions) | Assumptions |
|---|---|---|
| 2023 | 150 | Launch in select markets, initial adoption |
| 2024 | 210 | Increased prescribing, expanded geographic access |
| 2025 | 270 | Broader market acceptance, competition stabilizes |
| 2026 | 330 | Market saturation point approaches |
| 2027 | 380 | Patent expiry pressures, potential generics increase |
These figures assume steady adoption and no major regulatory setbacks.
What price strategies could optimize market share?
- Tiered Pricing: Differentiating prices across regions with economic considerations.
- Bundling: Combining with other hormonal therapies or contraceptives.
- Patient Assistance Programs: Offering discounts for underinsured populations.
Key Takeaways
- The drug operates within the competitive oral contraceptive market, with North America and Europe being primary revenue sources.
- Price points range from USD 10 to USD 50 monthly retail, with significant variation based on generic status.
- Market growth influenced by increasing demand, regulatory approvals, and demographic trends.
- Patent-life and generic entry are primary factors shaping the competitive landscape.
- Revenue projections assume conservative market share gains, with potential for higher upside in expanding markets.
FAQs
Q1: How soon will generics likely impact pricing?
A: Patent expiration typically occurs within 10–15 years from launch, after which generics are expected to dominate pricing, reducing costs by 50% or more.
Q2: What regions offer the most lucrative opportunities?
A: North America and Europe provide high-volume markets with established healthcare infrastructure, though emerging markets present growth opportunities due to expanding access.
Q3: Are there regulatory hurdles specific to this drug?
A: Approval depends on adherence to regional safety and efficacy standards. Variations in labeling and approval times influence market entry.
Q4: How does side effect profile influence market share?
A: Concerns over thromboembolic risks affect prescribing decisions; products with improved safety profiles might gain a competitive advantage.
Q5: What is the potential for price erosion over the next decade?
A: As patents expire and generics enter markets, prices are expected to decline significantly, with potential reductions of up to 50% or more.
References
- MarketWatch. "Global Oral Contraceptives Market Size & Growth." 2022.
- IQVIA. "Pharmaceutical Market Data." 2022.
- DrugPatentWatch. "Patent and Market Data for Hormonal Contraceptives." 2022.
- FDA. "Approved Drug Products." 2022.
- European Medicines Agency. "Product Approvals." 2022.
More… ↓
