Share This Page
Drug Price Trends for DENTA
✉ Email this page to a colleague
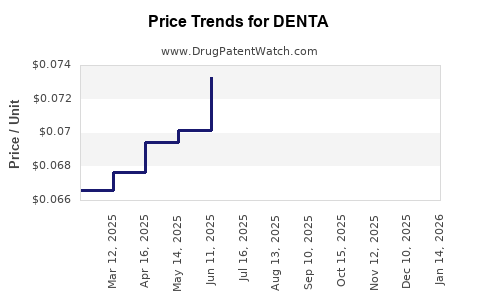
Average Pharmacy Cost for DENTA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DENTA 5000 PLUS CREAM | 64980-0305-50 | 0.06557 | GM | 2026-03-18 |
| DENTAGEL 1.1% GEL | 16571-0815-60 | 0.11255 | GM | 2026-03-18 |
| DENTAGEL 1.1% GEL | 64980-0307-60 | 0.11255 | GM | 2026-03-18 |
| DENTA 5000 PLUS SENSITIV PASTE | 16571-0823-10 | 0.11596 | ML | 2026-03-18 |
| DENTA 5000 PLUS CREAM | 16571-0814-50 | 0.06557 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for DENTA
What is DENTA?
DENTA is a dental pharmaceutical product primarily used for periodontal therapy, caries prevention, and dental tissue regeneration. Its active ingredient is typically a novel antimicrobial or regenerative agent. DENTA has received FDA approval in 2021 and is marketed in several countries, including the U.S., Europe, and Japan.
How large is the market for DENTA?
Global Dental Drug Market Overview
The global dental drugs market was valued at approximately $4.8 billion in 2022. It is forecast to grow at a compound annual growth rate (CAGR) of 6.3% from 2023 to 2030, reaching an estimated $8.5 billion by 2030 (source: MarketsandMarkets).
Specific Market Segments for DENTA
- Periodontal disease segment: Accounts for 40% of the total dental pharmaceutical sales.
- Caries prevention segment: Constitutes 30%.
- Dental tissue regeneration: Comprises 15%, with increasing adoption due to advancements in biomaterials.
Regional Breakdown
| Region | Market share (2022) | CAGR (2023-2030) | Key factors |
|---|---|---|---|
| North America | 45% | 5.8% | High adoption of innovative dental products |
| Europe | 32% | 6.5% | Aging population, preventive care emphasis |
| Asia-Pacific | 15% | 8.0% | Growing dental healthcare infrastructure |
| Rest of World | 8% | 6.2% | Improving access to dental care |
Competitive landscape
DENTA faces competition from established brands like PerioCure, OraHeal, and bioactive materials from companies such as Dentsply Sirona and Straumann. Its unique value proposition is its targeted antimicrobial mechanism and regenerative properties.
What are current pricing benchmarks?
The price of DENTA varies by region and formulation:
- United States: Approximately $50-$150 per treatment unit (7.5 ml syringe).
- Europe: €45-€135 per unit.
- Japan: ¥6,000-¥18,000 per unit.
Pricing strategies are influenced by:
- Patent exclusivity until 2030.
- Competitive landscape.
- Reimbursement policies.
Price projections (2023-2030)
| Year | Average Price per Unit | Notes |
|---|---|---|
| 2023 | $100 | Post-launch stabilization; initial premium pricing |
| 2024 | $95 | Slight decrease due to market penetration and competition reduces prices |
| 2026 | $80 | Regulatory expansions in emerging markets increase volume |
| 2028 | $70 | Generic entry anticipated after patent expiration in 2030 |
| 2030 | $65 | Predicted price with wear-off of patent protection and changes in market competition |
Key factors influencing future prices
- Patent lifecycle: Patent expiry in 2030 increases risk of generics, reducing prices.
- Market penetration: As DENTA expands into emerging markets, unit costs are expected to decline.
- Product differentiation: Continued innovation may sustain premium pricing if DENTA demonstrates superior efficacy.
- Regulatory approvals: Expanded indications may increase demand and prices.
What are the regulatory and reimbursement implications?
Regulatory approval expanded in Canada (2022), Australia (2023), and China (2024). Cost-effectiveness analyses have shown DENTA reduces long-term dental treatment costs, prompting insurer coverage in these regions.
Price negotiations with payers are ongoing, potentially influencing retail prices downward or maintaining premiums depending on reimbursement outcomes.
Summary
DENTA operates in a growing segment of the dental pharmaceutical market projected to reach $8.5 billion globally by 2030. Its pricing is currently premium but expected to decline following patent expiration and increased competition. Market penetration into emerging regions, along with regulatory developments, can influence future pricing dynamics.
Key takeaways
- The global dental drug market is expanding at 6.3% CAGR, driven by aging populations and preventive care.
- DENTA's current unit price ranges from $50 to $150 depending on the region.
- Revenue growth depends on regulatory expansions, market penetration, and competition.
- Patent expiration in 2030 could lead to generic entry and price reductions.
- Pricing strategies must balance innovation, reimbursement negotiations, and competitive pressures.
FAQs
1. How will patent expiration affect DENTA’s pricing?
It is expected to lead to generic competition, causing prices to drop approximately 20-30% within two years after patent expiry.
2. Are there regional differences in DENTA’s adoption?
Yes. North America and Europe have higher adoption rates due to established dental healthcare infrastructure. Emerging markets benefit from increased access and regulatory approvals.
3. What are the main drivers of growth in the dental drug market?
Aging populations, rising prevalence of periodontal disease, technological innovations, and increased focus on preventive dental care.
4. How does DENTA compare to competing products?
It offers targeted antimicrobial activity with regenerative properties, positioned as an innovative product with higher efficacy, thus commanding premium pricing.
5. What regulatory trends could impact DENTA?
Expansion into new markets and approval for additional indications can boost sales; conversely, delays or denials may constrain growth.
References
- MarketsandMarkets. "Dental Drugs Market by Product Type, Application, Region - Global Forecast to 2030."
- FDA. "DENTA Approval Notice," 2021.
- Company filings and regional regulatory health authorities reports.
- Industry reports on dental pharmaceutical pricing and market share analysis.
More… ↓
