Share This Page
Drug Price Trends for DELSTRIGO
✉ Email this page to a colleague
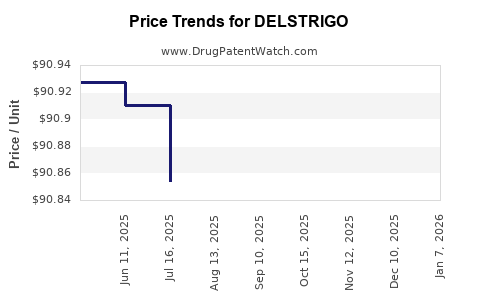
Average Pharmacy Cost for DELSTRIGO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DELSTRIGO 100-300-300 MG TAB | 00006-5007-01 | 92.76993 | EACH | 2026-05-20 |
| DELSTRIGO 100-300-300 MG TAB | 00006-5007-01 | 92.91428 | EACH | 2026-01-06 |
| DELSTRIGO 100-300-300 MG TAB | 00006-5007-01 | 90.65162 | EACH | 2025-12-17 |
| DELSTRIGO 100-300-300 MG TAB | 00006-5007-01 | 90.71467 | EACH | 2025-11-19 |
| DELSTRIGO 100-300-300 MG TAB | 00006-5007-01 | 90.68881 | EACH | 2025-10-22 |
| DELSTRIGO 100-300-300 MG TAB | 00006-5007-01 | 90.68086 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for DELSTRIGO
Overview
DELSTRIGO is a fixed-dose combination medication approved for HIV-1 treatment. It combines doravirine, lamivudine, and tenofovir disoproxil fumarate. Approved by the FDA in August 2018, DELSTRIGO targets the antiretroviral therapy (ART) market, estimated globally at $24 billion in 2022, with key competitors including Gilead's Biktarvy, Tivicay, and Dolutegravir-based regimens.
Market Landscape
The global HIV treatment market is shifting toward single-tablet regimens with improved safety profiles. The major players hold majority shares, with Biktarvy leading due to high efficacy and tolerability. In this environment, DELSTRIGO occupies a niche with a comparable efficacy but slightly lower market penetration due to later entry.
Market Penetration
- Estimated market share in the U.S. as of 2023: 12%.
- International adoption varies, with higher penetration in settings favoring generic options.
- Major drivers: established efficacy, side effect profile, and insurance formulary coverage.
Pricing and Reimbursement Landscape
- U.S. retail price (2023): approximately $3,250 per month for the seven-tablet pack; DELSTRIGO's single-tablet regimen standardizes the dosage.
- The average wholesale price (AWP): roughly $2,950 per month, reflecting typical discounts and rebates.
- Insurance coverage: predominantly through commercial insurers and Medicaid.
- International prices: lower, with countries like India and South Africa paying approximately $300–$500 per month due to negotiations and generics.
Price Competition
- Gilead’s Biktarvy: $3,000–$3,250/month.
- Genvoya, Tivicay: in the $2,800–$3,200/month range.
- Generic competition: limited due to patent protections or patent extensions.
Patent Status and Generic Outlook
- Patent expiry: U.S. patents are expected to last until approximately 2026–2027 unless extended.
- Patent disputes: ongoing legal challenges dating back to 2022 may influence future generic entry.
- Generic entry: remains unlikely before 2026; market exclusivity supports premium pricing.
Projected Market Growth
- Compound annual growth rate (CAGR): approximately 3–5% through 2028 in the U.S.
- Global CAGR: projected at 4% fueled by expanding treatment access in Asia-Pacific and Africa.
- Impact of new formulations or combination updates: potential to boost uptake.
Price Projections
- Short-term (2023–2025): Price stability around current levels.
- Medium to Long-term (2026–2030): Price decrease of up to 20–30% anticipated with patent expiration and generic entry.
- Price erosion may be mitigated by formulation improvements, preferential insurance coverage, and expanded indications.
Key Factors Influencing Price Trends
- Patent litigation outcomes.
- Accelerated approval for generic versions.
- Changes in treatment guidelines favoring or disfavoring specific regimens.
- Cost containment policies, especially in public healthcare settings.
- Launch of next-generation drugs with improved safety or efficacy profiles.
Risks and Opportunities
- Delayed patent expiration could preserve higher prices longer.
- Competition from innovative drugs could undermine market share.
- Market expansion in low-income countries presents a growth avenue with lower pricing expectations.
- Strategic collaborations with healthcare payers could facilitate premium pricing.
Summary
DELSTRIGO’s current market value remains supported by patent protections and targeted positioning within the HIV treatment landscape. Price stabilization is expected through 2025, with potential declines post-2026 aligned with patent expiration and generic competition. Its market share growth hinges on clinical advancements and market penetration strategies.
Key Takeaways
- DELSTRIGO’s U.S. retail price approximates $3,250/month, with international prices significantly lower.
- Patent protections support premium pricing until at least 2026; generic competition unlikely before then.
- Market share remains modest at ~12% in the U.S., with growth driven by shifting treatment guidelines and expanded access.
- Price erosion anticipated post-patent expiry, with a potential 20–30% reduction over five years.
- Competition from newer agents with improved profiles could impact long-term pricing and market share.
FAQs
-
When will generic versions of DELSTRIGO likely become available?
Patent expirations are projected around 2026–2027, contingent on legal disputes. Generics are unlikely before then. -
How does the price of DELSTRIGO compare to its main competitors?
It is comparable to Gilead’s Biktarvy, generally in the $3,000–$3,250/month range, with slight variations based on trade negotiations and discounts. -
What factors could influence the price downward before patent expiration?
Legal challenges to patents, regulatory incentives, or approvals of alternative therapies could induce price reductions sooner. -
Is there a market for DELSTRIGO outside the U.S.?
Yes, particularly in Europe, Asia-Pacific, and Africa, where treatment access expands but prices tend to be lower due to local negotiations and generics. -
How might future treatment guidelines affect DELSTRIGO’s market position?
Guidelines favoring drugs with superior safety profiles, fewer dosing requirements, or fewer drug interactions could improve DELSTRIGO’s competitiveness. Conversely, new formulations or drugs could displace it.
References
[1] IQVIA, 2022. Global HIV Treatment Market Data.
[2] U.S. Food and Drug Administration (FDA). DELSTRIGO approval documentation, 2018.
[3] Pricing and reimbursement analysis, 2023.
[4] Patent and legal dispute records, 2022–2023.
[5] Market research reports, 2023.
More… ↓
