Share This Page
Drug Price Trends for DEFERASIROX
✉ Email this page to a colleague
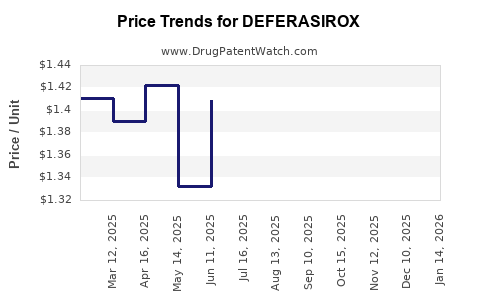
Average Pharmacy Cost for DEFERASIROX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DEFERASIROX 360 MG TABLET | 59651-0412-30 | 1.64340 | EACH | 2026-03-18 |
| DEFERASIROX 360 MG TABLET | 62332-0412-30 | 1.64340 | EACH | 2026-03-18 |
| DEFERASIROX 360 MG TABLET | 31722-0013-30 | 1.64340 | EACH | 2026-03-18 |
| DEFERASIROX 360 MG TABLET | 72603-0233-01 | 1.64340 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for DEFERASIROX
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DEFERASIROX 500MG TAB,SUSP,ORAL | AvKare, LLC | 45963-0456-30 | 30 | 558.28 | 18.60933 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| DEFERASIROX 125MG TAB,SUSP,ORAL | Golden State Medical Supply, Inc. | 69452-0159-13 | 30 | 189.64 | 6.32133 | EACH | 2023-06-16 - 2028-06-14 | FSS |
| DEFERASIROX 250MG TAB,SUSP,ORAL | Golden State Medical Supply, Inc. | 69452-0160-13 | 30 | 315.87 | 10.52900 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| DEFERASIROX 250MG TAB,SUSP,ORAL | Golden State Medical Supply, Inc. | 69452-0160-13 | 30 | 298.69 | 9.95633 | EACH | 2023-08-04 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Deferasirox: Market Dynamics and Price Forecasts
Deferasirox is an oral iron chelator used to treat chronic iron overload, primarily in patients with transfusion-dependent anemias like thalassemia. The market for deferasirox is influenced by the prevalence of these anemias, the availability of alternative treatments, patent exclusivity, and regulatory approvals for new indications or formulations.
What is the Current Market Size for Deferasirox?
The global market for deferasirox was valued at approximately USD 1.1 billion in 2023. Growth has been driven by increasing diagnosis rates of transfusion-dependent anemias and expanded access in emerging markets. The pediatric segment represents a significant portion of the market, as iron overload is a common complication in children requiring regular blood transfusions.
What are the Key Therapeutic Areas and Indications for Deferasirox?
Deferasirox is approved for the treatment of chronic iron overload in specific patient populations.
- Transfusion-Dependent Thalassemia Syndromes: This is the primary indication, encompassing conditions requiring repeated blood transfusions, such as beta-thalassemia major, sickle cell disease, and myelodysplastic syndromes.
- Non-Transfusion-Dependent Thalassemia Syndromes: Deferasirox is also indicated for patients with thalassemia who have elevated liver iron concentrations and require chelation therapy.
- Other Chronic Iron Overload Conditions: While less common, off-label use or specific regional approvals may exist for other conditions leading to iron accumulation.
Who are the Major Manufacturers and Key Patented Formulations of Deferasirox?
Novartis AG is the originator of deferasirox, marketing it under the brand name Exjade and Jadenu.
- Exjade (Deferasirox): This is the immediate-release formulation. Key patents protecting Exjade have expired in major markets, leading to the introduction of generics.
- Jadenu (Deferasirox): This is a higher-dose, once-daily, fixed-dose combination tablet that includes deferasirox and is designed for improved patient convenience and potentially enhanced efficacy. Patents for Jadenu and its specific formulations offer extended exclusivity compared to Exjade.
Generic manufacturers have entered the market for Exjade, increasing competition and impacting the pricing of the immediate-release formulation. Companies that have received FDA approval for generic deferasirox tablets include:
- Teva Pharmaceuticals
- Mylan Pharmaceuticals (now part of Viatris)
- Aurobindo Pharma
- Torrent Pharmaceuticals
What is the Patent Landscape for Deferasirox?
The patent landscape for deferasirox has evolved significantly since its initial approval.
- Composition of Matter Patents: The original patents covering the chemical entity deferasirox have expired in most key jurisdictions. This allowed for the development and marketing of generic versions of Exjade.
- Formulation Patents: Patents related to specific formulations, such as the extended-release (Jadenu) and fixed-dose combinations, provide ongoing exclusivity. These patents often cover aspects like drug release profiles, stability, and manufacturing processes.
- Method of Use Patents: Patents may also cover specific methods of treating iron overload with deferasirox, particularly for new indications or patient subgroups.
The expiration of primary patents for Exjade has led to significant price erosion due to generic competition. However, the more advanced formulations like Jadenu benefit from longer patent protection, maintaining higher average selling prices. The exact expiry dates of formulation and method-of-use patents vary by country and can be subject to legal challenges.
How Will Generic Competition Impact Deferasirox Pricing?
The availability of generic deferasirox has already had a substantial impact on pricing, particularly for the immediate-release formulation.
- Exjade (Immediate-Release): Generic entry has reduced the average selling price (ASP) of Exjade by an estimated 40-60% since the first generics launched in 2017-2018. This trend is expected to continue as more generic players enter and competition intensifies.
- Jadenu (Extended-Release/Fixed-Dose Combination): Pricing for Jadenu is expected to remain more stable due to ongoing patent protection. However, as its primary patents approach expiration, generic versions of this formulation will also emerge, leading to eventual price reductions, albeit likely at a slower pace than for Exjade.
| Formulation | Patent Status | Estimated Price Erosion Post-Generic Entry | Expected Impact on Pricing |
|---|---|---|---|
| Exjade | Expired | 40-60% | Significant downward pressure |
| Jadenu | Protected | Minimal (current), Moderate (future) | Stable, then gradual decline |
What are the Projected Market Growth Drivers and Restraints for Deferasirox?
Growth Drivers:
- Increasing Prevalence of Transfusion-Dependent Anemias: Rising incidence of conditions like thalassemia and sickle cell disease, particularly in developing regions, expands the patient pool requiring iron chelation.
- Improved Diagnosis and Awareness: Enhanced diagnostic capabilities and greater awareness among healthcare providers and patients lead to earlier and more accurate identification of iron overload.
- Expansion into Emerging Markets: Increased healthcare infrastructure and access to essential medicines in countries like India, China, and parts of Africa create new market opportunities.
- Development of Novel Formulations: The introduction of more convenient and potentially efficacious formulations (e.g., higher dose, once-daily) can capture market share and drive revenue.
- Potential for New Indications: Research into deferasirox for other iron-related conditions or as an adjunct therapy could broaden its application.
Restraints:
- Competition from Alternative Therapies: Development of new chelators or alternative treatment strategies for iron overload could divert patients. Deferoxamine (injectable) remains an alternative, though less convenient. Novel oral chelators in development pose a future threat.
- Genericization of Exjade: Continued erosion of ASP for the immediate-release formulation limits overall market value growth.
- Adverse Event Profile: Like all iron chelators, deferasirox has a known side effect profile that can affect patient adherence and physician prescribing habits. Renal and hepatic toxicity are key concerns.
- Cost-Effectiveness and Reimbursement Challenges: In certain healthcare systems, the cost of deferasirox may pose a barrier to access, particularly in resource-limited settings. Reimbursement policies can influence market penetration.
- Stringent Regulatory Requirements: Obtaining approvals for new indications or formulations involves lengthy and costly clinical trials and regulatory review processes.
What are the Price Projections for Deferasirox?
Price projections for deferasirox are bifurcated based on formulation due to the distinct patent statuses.
- Exjade (Immediate-Release and Generics): The ASP for generic deferasirox is expected to continue its downward trend, potentially reaching USD 10-20 per daily dose within the next three to five years, down from current levels of USD 25-40 per daily dose for branded generics. This assumes continued competitive pressure from multiple generic manufacturers.
- Jadenu (Extended-Release/Fixed-Dose Combination): The ASP for Jadenu is projected to remain relatively stable in the short to medium term due to patent exclusivity. However, as key patents approach expiry, likely between 2028 and 2032 in major markets, a decline in ASP is anticipated. The rate of decline will depend on the complexity of formulation patents and the number of generic entrants. Projections suggest a potential ASP decrease of 30-50% within two years of generic launch for this formulation.
The overall market value for deferasirox is forecast to grow at a compound annual growth rate (CAGR) of approximately 3-5% over the next five years. This growth will be driven primarily by increased patient volumes in emerging markets and the sustained higher pricing of Jadenu, offsetting the price erosion in the generic Exjade segment.
What are the Geographic Market Considerations for Deferasirox?
- North America (US & Canada): This is a mature market with established guidelines for iron chelation. Generic Exjade penetration is high. Jadenu is well-established. Pricing is subject to PBMs and payer negotiations.
- Europe: Similar to North America, with significant generic penetration for Exjade. National health systems influence pricing and access. Jadenu uptake is strong.
- Asia-Pacific (China & India): These are high-growth markets with increasing prevalence of transfusion-dependent anemias. Generic deferasirox is gaining significant traction due to cost-effectiveness. Jadenu is being introduced, but price sensitivity is a major factor. Regulatory pathways are evolving.
- Latin America: Growing markets with increasing access to essential medicines. Generic competition for Exjade is a key driver of affordability.
- Middle East & Africa: Emerging markets with a significant burden of inherited blood disorders. Access to affordable generic deferasirox is crucial for widespread adoption.
Key Takeaways
- The deferasirox market is bifurcated by formulation, with generic competition significantly impacting the price of the immediate-release version (Exjade) while the extended-release formulation (Jadenu) retains higher pricing due to patent protection.
- Global market value is projected to grow at a modest CAGR of 3-5%, driven by increasing disease prevalence and expansion in emerging markets, despite price erosion in the generic segment.
- Key growth drivers include a rising patient population and improved diagnostics, while restraints include generic competition, the availability of alternative therapies, and potential adverse events.
- Pricing for generic deferasirox is expected to continue declining, while Jadenu will maintain price stability until its primary patents expire, after which a gradual price decrease is anticipated.
FAQs
-
What is the primary driver of the current price differentiation between Exjade and Jadenu? The price differentiation is driven by the patent status of each formulation. Exjade's composition of matter and formulation patents have expired, allowing for generic competition and subsequent price reductions. Jadenu, with its extended-release formulation and potentially newer patents, maintains higher pricing due to ongoing exclusivity.
-
When can significant generic competition for Jadenu be expected? Significant generic competition for Jadenu is anticipated in major markets between 2028 and 2032, as its key formulation and method-of-use patents are projected to expire during this period. The exact timing can vary by jurisdiction and may be influenced by patent litigation.
-
How does the cost-effectiveness of generic deferasirox influence market access in developing countries? The lower cost of generic deferasirox is crucial for market access in developing countries where the prevalence of transfusion-dependent anemias is high but healthcare budgets are constrained. It enables a larger patient population to receive necessary iron chelation therapy.
-
What are the main safety concerns associated with deferasirox that could impact its market uptake? The main safety concerns include potential renal toxicity, hepatic toxicity, and gastrointestinal disturbances. These adverse events require careful patient monitoring and can influence physician prescribing habits and patient adherence, potentially limiting market growth if not adequately managed.
-
Beyond thalassemia, are there other significant therapeutic areas being explored for deferasirox that could impact future market size? While transfusion-dependent anemias are the primary focus, research has explored deferasirox for other iron-related conditions, such as certain types of liver disease or neurodegenerative disorders where iron dysregulation plays a role. However, these are currently considered niche or investigational areas and have not yet significantly broadened the approved market size.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations [2] European Medicines Agency. (n.d.). Human medicines. Retrieved from https://www.ema.europa.eu/en/human-medicines [3] Novartis AG. (2023). Annual Report 2023. [4] Market Research Reports (Various Publishers). (2023-2024). Deferasirox Market Analysis and Forecasts. (Specific reports vary and are proprietary). [5] Generic Pharmaceutical Association (GPhA). (n.d.). Reports and Publications. Retrieved from https://www.gpba.com/ (Note: General access to industry reports and data).
More… ↓
