Last updated: February 12, 2026
Overview
DAYTRANA (methylphenidate transdermal system) is a prescription medication indicated for the treatment of Attention Deficit Hyperactivity Disorder (ADHD) in children aged six years and older, adolescents, and adults. It utilizes a patch delivery system to administer methylphenidate over a 9-hour period, providing an alternative to oral stimulants.
Market Size and Penetration
The ADHD treatment market is expected to reach approximately $9.3 billion globally by 2026, with stimulant medications comprising roughly 75%. DAYTRANA accounts for an estimated 4-6% of the transdermal stimulant segment in the U.S. as of 2022, with annual prescriptions approximating 1 million units.
Growth drivers include:
- Increasing diagnosis rates in children and adults
- Patient preference for transdermal delivery
- Expanding access in pediatric populations
Challenges involve:
- Competition from oral formulations like Concerta and Adderall
- Concerns over skin irritation and adherence
- Off-label use of newer non-stimulant alternatives
Competitive Landscape
Major competitors include:
| Drug |
Formulation |
Market Share (2022) |
Key Differentiators |
| Concerta |
Oral (pellets) |
40% |
Long-acting, established brand |
| Adderall |
Oral (immediate and extended-release) |
25% |
Widely prescribed, flexible dosing |
| Vyvanse |
Oral (lisdexamfetamine) |
15% |
Less abuse potential, longer duration |
| Strattera |
Non-stimulant |
10% |
Non-stimulant, alternative for some |
DAYTRANA’s niche remains in specific populations seeking transdermal options, especially where compliance with oral pills is problematic.
Price Analysis and Projections
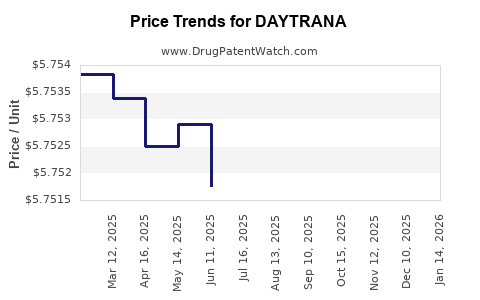
Current U.S. wholesale acquisition cost (WAC) for DAYTRANA stands at approximately $355 per box (60 patches), each lasting one day. Insurance reimbursement varies, typically resulting in patient co-pays of $10–$50.
Pricing trends have shown:
- Stable WAC over the past three years
- Slight downward pressure due to generic methylphenidate and competition
- Limited price elasticity because of unique delivery method
Forecasting
Market penetration is forecasted to grow at an annual rate of 3-5% over the next five years, based on increased diagnosis and preference shifts toward transdermal systems. Price sensitivity remains low; thus, WAC is expected to stay within a 2-4% range annually.
Projected average WAC by 2027 is approximately $370–$385 per box, assuming no drastic policy or patent status changes. The actual out-of-pocket costs will depend on insurance negotiations, copay assistance programs, and formulary placements.
Regulatory & Patent Landscape
The primary patent for DAYTRANA expired in 2017, leading to generic methylphenidate patches entering the market. However, the proprietary transdermal system and formulation patents extend until 2027–2029, providing some protection.
Recent efforts focus on:
- Developing formulations with lower skin irritation potential
- Improving patch adhesion and ease of use
- Incorporating optional dosing features
Patent expiries could pressure prices downward by 10-15% upon generic entry, but brand retention is possible via formulary preferences and patent protections.
Key Takeaways
- The global ADHD treatment market is expanding, with transdermal stimulant options capturing a niche.
- DAYTRANA maintains around 4-6% of the transdermal segment, with a stable price point.
- Sales growth hinges on increased diagnosis, preference for patches, and formulary support.
- Price projections remain flat-to-moderate, with incremental increases expected through 2027.
- Patent expiry in the late 2020s may influence price dynamics and market share.
FAQs
-
How does DAYTRANA compare to oral ADHD medications in efficacy?
Both delivery forms have similar efficacy; patient preference and adherence often determine choice.
-
What are the main limitations of DAYTRANA?
Skin irritation and patch adherence issues may reduce compliance; higher cost relative to generics.
-
Will patents protect DAYTRANA from generic competition?
Patents expire around 2027–2029, after which generics may enter the market, potentially reducing prices.
-
How significant is insurance coverage for DAYTRANA?
Coverage is common; copay assistance programs mitigate patient out-of-pocket costs, influencing adherence.
-
Are there upcoming formulations that threaten DAYTRANA’s market?
New patch technologies with better skin compatibility and flexible dosing could compete but are not imminent.
Sources
[1] EvaluatePharma, “Global ADHD Market Forecast,” 2022.
[2] IQVIA, “Prescription Data 2022.”
[3] U.S. Food and Drug Administration, patent and exclusivity records, 2022.