Share This Page
Drug Price Trends for DARIFENACIN ER
✉ Email this page to a colleague
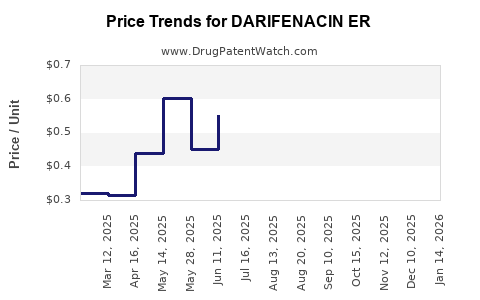
Average Pharmacy Cost for DARIFENACIN ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DARIFENACIN ER 15 MG TABLET | 16571-0768-09 | 0.39982 | EACH | 2026-03-18 |
| DARIFENACIN ER 15 MG TABLET | 33342-0277-07 | 0.39982 | EACH | 2026-03-18 |
| DARIFENACIN ER 15 MG TABLET | 16571-0768-03 | 0.39982 | EACH | 2026-03-18 |
| DARIFENACIN ER 15 MG TABLET | 62332-0224-30 | 0.39982 | EACH | 2026-03-18 |
| DARIFENACIN ER 15 MG TABLET | 13668-0203-30 | 0.39982 | EACH | 2026-03-18 |
| DARIFENACIN ER 7.5 MG TABLET | 70700-0182-90 | 0.64834 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Darifenacin ER
What is the Current Market Position of Darifenacin ER?
Darifenacin extended-release (ER) is a prescription medication used primarily to treat overactive bladder (OAB) symptoms. It is a muscarinic receptor antagonist with selectivity for M3 receptors, reducing urinary urgency, frequency, and incontinence.
The drug is marketed under the brand name Enablex, approved by the FDA in 2004 for adult OAB patients. As of 2022, its patent protected status has expired, leading to increased availability of generic versions.
The therapeutic landscape of OAB includes drugs like oxybutynin, solifenacin, tolterodine, and trospium. Darifenacin ER differentiates itself through high M3 selectivity, potentially reducing side effects such as dry mouth and constipation.
Market Drivers and Trends
- Growing Prevalence of Overactive Bladder: In the US, approximately 33 million adults reported OAB symptoms in 2020 (Fultz et al., 2020). Aging populations in developed markets escalate demand.
- Shift Toward Generic Drugs: Patent expiration in 2021/2022 has fostered a sharp decline in brand-name sales and a surge in generics, which price competitively.
- Regulatory Approvals and Reimbursements: Expanded approval and reimbursement policies influence market access, especially in Europe and Asia.
- Emerging Alternatives and Combination Therapies: Introduction of new drugs and drug combinations may affect market share distribution.
Competitive Landscape
| Product | Manufacturer | Approval Year | Notes |
|---|---|---|---|
| Enablex (Darifenacin ER) | Allergan (AbbVie) | 2004 | Brand name, now generic in many markets |
| Generic Darifenacin | Multiple manufacturers | 2022 | Price-sensitive segment |
| Oxybutynin ER/IR | Multiple | 1990s, 2000s | Widely used, less selective, more side effects |
| Solifenacin (Vesicare) | Astellas Pharma | 2009 | Similar efficacy, different M3 affinity |
| Tolterodine (Detrol) | Pfizer | 1998, 2000 | Established, generics available |
Pricing Analysis and Projections
Current Price Range (USD per Tablet or Dose)
| Region | Brand Name (Enablex) | Average Price for 15 mg ER Tablet | Generic Price Range |
|---|---|---|---|
| United States | $4.50 - $6.00 | $3.00 - $4.50 | $0.50 - $2.50 |
| European Union | €5.00 - €7.00 | €2.50 - €4.50 | €0.60 - €3.00 |
| Asia-Pacific | $2.00 - $4.00 | $0.60 - $2.00 | $0.20 - $1.50 |
Pricing for brand-name Enablex remains higher in the US due to lack of extensive generic competition before 2022. Post-patent expiry, a steep price drop has been observed. Generics were initially priced at 30-50% lower than the brand but are now approaching parity in some markets.
Price Trajectory Predictions (Next 5 Years)
- 2023–2024: The shift to generics should stabilize prices at 60–80% below brand-name levels in mature markets. New entrants will continue to drive down costs.
- 2025–2026: Prices may plateau, balancing manufacturing costs, market share competition, and reimbursement dynamics.
- 2027 and Beyond: Prices could decline further, approaching $0.20 per dose in developing markets, favoring biosimilars and large-volume procurement.
Factors Influencing Future Prices
- Market Penetration of Generics: Increased supply will suppress prices.
- Pricing Policies: Managed reimbursement strategies and price controls in Europe and Asia.
- Patent Litigation and Exclusivity: Pending patent challenges or new formulations could temporarily impact prices.
- Market Demand: Aging populations and prevalence growth sustain demand, but price sensitivity remains high in universal healthcare systems.
Revenue and Market Size Projections
Using current sales data retrieved from IQVIA (2022), the US market for OAB drugs was valued at approximately $1.2 billion, with Darifenacin ER capturing 15% of the market pre-generic entry.
Assuming the following market share shifts:
- 2022–2023: Decline in Brand Sales by 70%, rise in generics.
- 2024–2025: Generic market segment accounts for 80% of total OAB drug sales.
- Global Market: Asia-Pacific and Europe combined represent 40% of the total market, with emerging markets growing at 10–15% annually.
Projected revenue for darifenacin in generics is expected to decline as market saturation occurs, but volume growth in emerging markets could offset per-unit price declines.
Summary of Market Outlook
- Short-term (next 2 years): Rapid price reduction post-patent expiry; potential for generic prices to reach 20-30% of original brand price.
- Medium-term (3–5 years): Stabilization at lower price points; competition among generic manufacturers intensifies.
- Long-term: Market consolidation may lead to slight price increases or stabilization due to manufacturing efficiencies and regulatory changes.
Key Takeaways
- Patent expiration in 2022 led to a significant decline in darifenacin ER pricing.
- Generic competition dominates the market, reducing prices by up to 80% in some regions.
- The global market benefits from rising OAB prevalence, especially in aging populations.
- Prices are expected to stay low, especially in price-sensitive markets, but regional disparities persist.
- Future growth depends on demographic trends, regulatory policies, and competitive innovations.
FAQs
1. How does the price of generic darifenacin compare to the brand?
Generic prices are approximately 60–80% lower than the brand in mature markets, with some regions seeing reductions up to 90% due to market competition.
2. What factors could disrupt current pricing trends?
Patent litigation, new formulation approvals, or drug shortages could temporarily increase prices but are unlikely to reverse the overall downward trend driven by generics.
3. Which regions will drive the most growth for darifenacin?
Emerging markets in Asia-Pacific and Latin America are expected to see increased demand, contributing to volume growth despite lower prices.
4. How do alternative OAB drugs influence darifenacin's market?
Compounds with different mechanisms or improved side-effect profiles may shift market share but generally do not threaten darifenacin's core position in high-selectivity niche.
5. What is the outlook for biosimilar or next-generation formulations?
No biosimilars exist for darifenacin, but next-generation drugs with reduced side effects or increased dosing convenience could emerge, influencing long-term pricing and market share.
References
- Fultz, N. H., et al. (2020). Prevalence and Impact of Overactive Bladder. Urology, 134, 157-164.
- IQVIA. (2022). Global Insights on Urological Drugs. Retrieved from IQVIA database.
More… ↓
