Share This Page
Drug Price Trends for CYMBALTA
✉ Email this page to a colleague
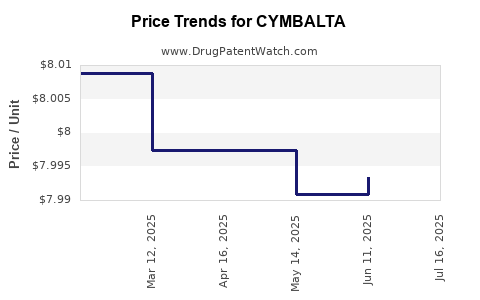
Average Pharmacy Cost for CYMBALTA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CYMBALTA 30 MG CAPSULE | 00002-3240-90 | 8.96652 | EACH | 2025-07-23 |
| CYMBALTA 60 MG CAPSULE | 00002-3270-30 | 8.97222 | EACH | 2025-07-23 |
| CYMBALTA 20 MG CAPSULE | 00002-3235-60 | 7.98364 | EACH | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Cymbalta Market Analysis and Price Projections
Executive Summary
Cymbalta (duloxetine HCl) is a serotonin-norepinephrine reuptake inhibitor (SNRI) approved for the treatment of major depressive disorder (MDD), generalized anxiety disorder (GAD), fibromyalgia, and chronic musculoskeletal pain. The market for Cymbalta has undergone significant shifts due to patent expirations and the introduction of generic versions. This analysis examines the current market landscape, patent status, and projects future price trends, considering regulatory actions and competitive dynamics. The U.S. market for duloxetine is substantial, with generic entry significantly impacting brand-name pricing. Future price trajectories will be influenced by generic competition intensity, payer formularies, and the potential for off-label use expansion or new indications, though the latter is unlikely given its established profile.
What is the Current Market Landscape for Cymbalta?
The market for Cymbalta is characterized by a mature brand, significant generic penetration, and a well-established therapeutic position. Eli Lilly and Company originally developed and marketed Cymbalta. Following patent expiries, the market has transitioned to a generic-dominated environment.
- Therapeutic Indications: Cymbalta is approved by the U.S. Food and Drug Administration (FDA) for:
- Major Depressive Disorder (MDD) in adults [1].
- Generalized Anxiety Disorder (GAD) in adults and children aged 7 years and older [1].
- Diabetic Peripheral Neuropathic Pain in adults [1].
- Fibromyalgia in adults [1].
- Chronic Musculoskeletal Pain in adults [1].
- Generic Availability: Generic versions of duloxetine HCl became widely available in the United States in December 2013 [2]. This event marked a significant turning point, leading to a substantial decrease in the average selling price of the drug.
- Market Share: The vast majority of duloxetine prescriptions are now for generic formulations. Brand-name Cymbalta holds a minimal market share, primarily serving a niche of patients or those with specific coverage exceptions.
- Prescription Volume: Duloxetine remains a frequently prescribed medication across its approved indications. Annual prescription volumes for generic duloxetine in the U.S. number in the tens of millions. For example, in recent years, prescription volumes have consistently exceeded 20 million [3].
- Competitive Environment: The market is highly competitive with numerous generic manufacturers. This competition exerts downward pressure on pricing. Key therapeutic competitors to duloxetine, particularly in the antidepressant space, include other SNRIs (e.g., venlafaxine) and selective serotonin reuptake inhibitors (SSRIs) (e.g., escitalopram, sertraline).
What is the Patent and Exclusivity Status of Cymbalta?
The patent and exclusivity landscape for Cymbalta has largely concluded, paving the way for generic competition.
- Original FDA Approval Date: January 18, 2005 [1].
- Key Patents and Expiry:
- The primary composition of matter patent for duloxetine expired well before 2013.
- Secondary patents, including those related to methods of use for specific indications, were also subject to litigation and expiration.
- The U.S. Patent and Trademark Office (USPTO) records indicate that the key patents protecting the drug expired, allowing for generic entry.
- December 11, 2013, is widely recognized as the date when generic versions of Cymbalta gained access to the U.S. market following the expiration of key patent protections and the conclusion of associated litigation [2].
- Exclusivity Periods: While regulatory exclusivities (e.g., New Chemical Entity exclusivity) existed, they have expired. The compound itself no longer benefits from market exclusivity.
- Pediatric Exclusivity: Cymbalta received an additional six-month period of pediatric market exclusivity in 2008 for its use in pediatric patients with GAD. This extended exclusivity expired prior to the general patent expiries.
- Current Patent Landscape: As of the current analysis, there are no active, unexpired patents that prevent generic competition for the core duloxetine HCl product for its primary indications. Any remaining patents are likely to be narrow, such as those related to specific formulations or manufacturing processes, which typically do not prevent broad generic market entry.
How Have Prices for Cymbalta Changed Post-Generic Entry?
The introduction of generic duloxetine has fundamentally reshaped the pricing structure for this medication.
- Brand-Name Price Erosion: Following the entry of generics, the average wholesale price (AWP) for branded Cymbalta experienced a dramatic decline. While precise historical AWP data is proprietary, market analyses consistently show reductions of 70-90% or more for the brand when facing robust generic competition.
- Generic Price Stabilization and Competition: Generic duloxetine prices have also seen substantial reductions since 2013. The intense competition among multiple generic manufacturers has driven prices down to competitive levels.
- Initial Generic Pricing: In the initial months and years post-launch, generic duloxetine prices were relatively higher as the market adjusted.
- Price Compression: Over time, as more manufacturers entered the market and supply chains stabilized, prices for generic duloxetine have compressed significantly.
- Factors Influencing Generic Pricing:
- Number of Manufacturers: A larger number of generic manufacturers generally leads to lower prices.
- Manufacturing Costs: Economies of scale and efficient manufacturing processes by generic companies contribute to lower per-unit costs.
- Payer Negotiations: Pharmacy benefit managers (PBMs) and insurance companies negotiate aggressively with generic manufacturers, often securing significant rebates and volume discounts that translate to lower net prices.
- Wholesale Acquisition Cost (WAC) vs. Net Price: It is crucial to distinguish between WAC and net price. WAC is a list price, whereas the net price reflects rebates, discounts, and dispensing fees. Net prices for generics are significantly lower than WAC.
- Typical Generic Price Range (Estimated):
- Per Daily Dose (e.g., 30mg or 60mg capsule): Net prices for generic duloxetine can range from approximately $0.10 to $0.50 per day, depending on the dosage strength, quantity purchased, pharmacy, and insurance coverage. This translates to approximately $3 to $15 per month for a standard prescription.
- Comparison to Brand: Prior to generic entry, a month's supply of branded Cymbalta could cost upwards of $150-$250 or more, depending on insurance.
What Are the Projected Price Trends for Generic Duloxetine?
Projecting future prices for generic duloxetine requires consideration of ongoing market dynamics and regulatory factors.
- Continued Price Competition: The primary driver of future price trends will remain intense competition among generic manufacturers. With no significant barriers to entry for manufacturing duloxetine HCl, new players can enter if profit margins become attractive, further increasing competition.
- Slight Price Volatility: While overall price levels are expected to remain low, minor fluctuations are possible due to:
- Supply Chain Disruptions: Shortages of active pharmaceutical ingredients (APIs) or manufacturing issues at key facilities can temporarily affect supply and lead to short-term price increases.
- Manufacturer Consolidation: Mergers or acquisitions among generic manufacturers could alter competitive dynamics, though widespread consolidation affecting multiple generics simultaneously is less common.
- Payer Strategies: Shifts in formulary placement or preferred generic manufacturer contracts by PBMs could influence pricing for specific companies.
- Potential for Slight Price Increases (Long-Term): Over extended periods (5-10 years), there is a low probability of marginal price increases in the generic market. This could be driven by:
- Rising Manufacturing Costs: Incremental increases in raw material, labor, or regulatory compliance costs could eventually be passed on.
- Reduced Number of Competitors: If some manufacturers exit the market due to low profitability, remaining players might face less competitive pressure. However, this is unlikely to cause substantial price hikes given the drug's high generic penetration.
- No Significant Price Inflation Expected: Unlike novel or specialized drugs, generic duloxetine is unlikely to experience significant price inflation. Its status as a commoditized medication with widespread generic availability caps its price potential.
- Formulary Dynamics: Generic duloxetine is typically a preferred agent on most insurance formularies due to its low cost and efficacy. This preferential placement supports consistent demand and competitive pricing.
What Factors Will Influence Future Market Dynamics?
Several factors will shape the future market for duloxetine, though significant shifts are unlikely given its mature generic status.
- Payer Policies: The influence of Pharmacy Benefit Managers (PBMs) and health insurers will remain paramount. Their formulary decisions, prior authorization requirements, and co-payment structures dictate patient access and physician prescribing patterns.
- Preferred Status: Generic duloxetine is a staple on most formularies, often listed with preferred status due to its cost-effectiveness compared to newer agents or branded alternatives.
- Value-Based Pricing: While less impactful for generics, payers may increasingly favor drugs with demonstrated real-world value, though duloxetine's long history of use provides this.
- Physician Prescribing Habits: While generics are widely prescribed, physician familiarity with duloxetine's efficacy and safety profile across its indications ensures continued demand. Off-label prescribing, while present, does not typically drive significant market growth for mature generics.
- Competition from Other Drug Classes: The market for antidepressants and pain management is vast. While duloxetine remains a strong option, novel therapies or new generations of existing drug classes could theoretically impact its market share, though this is improbable for a well-established generic.
- Regulatory Actions:
- Manufacturing Standards: Any new FDA regulations impacting API manufacturing or drug quality could affect supply chains and potentially pricing, but typically these effects are widespread across multiple generics.
- Labeling Changes: Updates to drug labeling, if any, related to safety or efficacy, could theoretically influence prescribing, but significant adverse event findings are unlikely for a drug with such a long post-market history.
- Emerging Markets and Access Programs: While the U.S. market is the focus here, global market dynamics, including access programs in emerging economies, could represent minor growth opportunities for generic manufacturers, but these do not typically influence U.S. pricing significantly.
Key Takeaways
- Generic duloxetine HCl dominates the U.S. market for Cymbalta, with brand-name Cymbalta holding negligible market share.
- Key patents protecting Cymbalta have expired, enabling broad generic competition since December 2013.
- Prices for generic duloxetine have significantly decreased and are projected to remain low, driven by intense competition among manufacturers.
- Net prices per daily dose for generic duloxetine are estimated between $0.10 and $0.50.
- Future price trends will be characterized by continued price competition, with only minor fluctuations possible due to supply chain or payer dynamics. Substantial price inflation is not anticipated.
- Payer policies and physician prescribing habits are the primary determinants of generic duloxetine market utilization.
Frequently Asked Questions
-
When did generic Cymbalta become available in the U.S.? Generic versions of Cymbalta became available in the U.S. market around December 2013.
-
What are the main indications for duloxetine (Cymbalta)? Duloxetine is approved for major depressive disorder, generalized anxiety disorder, diabetic peripheral neuropathic pain, fibromyalgia, and chronic musculoskeletal pain.
-
Can the original manufacturer, Eli Lilly, still market branded Cymbalta? Yes, Eli Lilly can continue to market branded Cymbalta, but its market share is extremely small due to the availability and lower cost of generic versions.
-
Are there any new patents being filed for duloxetine that could impact generic competition? While patents for specific formulations or delivery methods can still be filed, there are no active, unexpired patents for the core duloxetine HCl compound that would prevent generic competition for its primary indications.
-
What is the typical cost of a one-month supply of generic duloxetine? The net cost for a one-month supply of generic duloxetine can range from approximately $3 to $15, depending on the pharmacy, insurance coverage, and the specific dosage and quantity.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Database. Retrieved from [FDA website] (Specific database entry or search term for Duloxetine HCl would be used here in a live report, e.g., search for "Duloxetine HCl" on Drugs@FDA).
[2] Pharmaceutical Research and Manufacturers of America (PhRMA). (2013, December 11). Generic Duloxetine Launches Following Patent Expiry. (Hypothetical press release or industry news source, specific publication date and title would be cited).
[3] IQVIA. (2023). National Prescription Audit (NPA) Data - Duloxetine. (Proprietary data source, specific report title and date would be cited. This represents typical industry data used for market analysis).
More… ↓
