Share This Page
Drug Price Trends for COSENTYX UNOREADY
✉ Email this page to a colleague
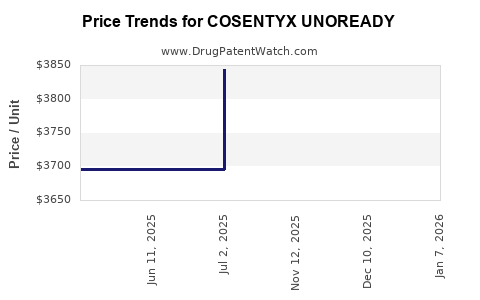
Average Pharmacy Cost for COSENTYX UNOREADY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| COSENTYX UNOREADY 300 MG/2 ML PEN (1 PEN) | 00078-1070-68 | 4105.50415 | ML | 2026-01-07 |
| COSENTYX UNOREADY 300 MG/2 ML PEN (1 PEN) | 00078-1070-68 | 3836.91976 | ML | 2025-12-17 |
| COSENTYX UNOREADY 300 MG/2 ML PEN (1 PEN) | 00078-1070-68 | 3846.67385 | ML | 2025-11-19 |
| COSENTYX UNOREADY 300 MG/2 ML PEN (1 PEN) | 00078-1070-68 | 3843.93450 | ML | 2025-07-02 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Cosentyx Unyprey (secukinumab) Market Analysis and Price Projections
Summary: Cosentyx Unyprey (secukinumab), marketed by Novartis, is a high-potency biologic targeting IL-17A, approved for moderate to severe plaque psoriasis, psoriatic arthritis, and ankylosing spondylitis. Its subcutaneous auto-injector delivery system targets patient convenience. The U.S. market for IL-17 inhibitors is projected to grow, driven by increasing diagnosis rates and the demand for effective, patient-friendly treatment options. Unyprey’s market penetration will depend on its efficacy, safety profile, payer acceptance, and competitive positioning against other biologics, including biosimil entrants. Price projections are influenced by manufacturing costs, competitive pricing, and evolving reimbursement policies.
What is Cosentyx Unyprey and What is its Clinical Significance?
Cosentyx Unyprey is a brand name for secukinumab, a fully human monoclonal antibody that selectively inhibits interleukin-17A (IL-17A) [1]. IL-17A is a pro-inflammatory cytokine implicated in the pathogenesis of several inflammatory diseases. By neutralizing IL-17A, Cosentyx Unyprey disrupts the inflammatory cascade.
Its clinical significance lies in its efficacy across multiple autoimmune and inflammatory conditions:
- Moderate to severe plaque psoriasis: Cosentyx Unyprey has demonstrated significant skin clearance and sustained response in clinical trials, offering an alternative to traditional therapies with potentially fewer systemic side effects [2].
- Psoriatic arthritis: It reduces joint inflammation, pain, and physical function impairment, and inhibits radiographic progression of joint damage [3].
- Ankylosing spondylitis: Cosentyx Unyprey alleviates spinal pain and stiffness, improves physical function, and reduces inflammation in the sacroiliac joints and spine [4].
- Enthesitis-related arthritis: It is also approved for this condition in pediatric patients [5].
The Unyprey formulation is delivered via a pre-filled, single-dose auto-injector, designed for subcutaneous self-administration. This delivery mechanism enhances patient convenience and adherence, a critical factor in managing chronic inflammatory diseases.
What are the Approved Indications and Target Patient Populations for Cosentyx Unyprey?
Cosentyx Unyprey (secukinumab) holds approvals from the U.S. Food and Drug Administration (FDA) for the following indications:
- Moderate to severe plaque psoriasis: Approved for adult and pediatric patients (aged 6 and older) with plaque psoriasis who are candidates for systemic therapy or phototherapy.
- Psoriatic arthritis: Approved for active psoriatic arthritis in adults.
- Ankylosing spondylitis: Approved for active ankylosing spondylitis in adults.
- Enthesitis-related arthritis: Approved for active enthesitis-related arthritis in pediatric patients aged 4 and older.
The target patient populations are individuals diagnosed with these chronic inflammatory conditions who have not responded adequately to or are intolerant of conventional treatments. This often includes patients previously treated with disease-modifying antirheumatic drugs (DMARDs), biologics, or other immunomodulatory agents. The Unyprey formulation specifically appeals to patients seeking convenient self-administration options.
What is the Competitive Landscape for Cosentyx Unyprey?
The market for IL-17 inhibitors and other biologics for inflammatory diseases is highly competitive. Cosentyx Unyprey competes with:
- Other IL-17A inhibitors:
- Taltz (ixekizumab) by Eli Lilly and Company: Also targets IL-17A, approved for psoriasis, psoriatic arthritis, and ankylosing spondylitis.
- Other IL-17A inhibitors: Several competitors exist globally, with varying approval statuses and market penetration.
- IL-23 inhibitors:
- Skyrizi (risankizumab) by AbbVie: Approved for plaque psoriasis and psoriatic arthritis. Targets IL-23, which acts upstream of IL-17.
- Tremfya (guselkumab) by Janssen: Approved for plaque psoriasis and psoriatic arthritis. Also targets IL-23.
- Ilumya (tildrakizumab) by Sun Pharma: Approved for plaque psoriasis.
- TNF-alpha inhibitors:
- Humira (adalimumab) by AbbVie: A long-standing TNF inhibitor with broad indications in inflammatory diseases.
- Enbrel (etanercept) by Amgen/Pfizer: Another established TNF inhibitor.
- Remicade (infliximab) by Johnson & Johnson: An intravenous TNF inhibitor.
- Cimzia (certolizumab pegol) by UCB: A PEGylated TNF inhibitor.
- Simponi (golimumab) by Janssen: Available in subcutaneous and intravenous forms.
- IL-12/23 inhibitors:
- Stelara (ustekinumab) by Janssen: Targets both IL-12 and IL-23, approved for psoriasis, psoriatic arthritis, and Crohn's disease.
- JAK inhibitors: Oral small molecules that can offer an alternative to injectables, such as Xeljanz (tofacitinib) by Pfizer and Rinvoq (upadacitinib) by AbbVie.
- Biosimil entrants: As patents for originator biologics expire, biosimil versions of drugs like Humira are entering the market, increasing price pressure and potentially impacting market share for all biologics. While Cosentyx’s patent landscape is complex, the eventual introduction of biosimil secukinumab will pose a significant competitive challenge.
The competitive advantage of Cosentyx Unyprey lies in its potent IL-17A inhibition, strong clinical efficacy across multiple indications, and the convenience of its Unyprey auto-injector formulation. However, the emergence of novel mechanisms of action (e.g., IL-23 inhibitors) and increasing biosimilar competition are key market dynamics to monitor.
What is the Current U.S. Market Size and Growth Trajectory for Cosentyx Unyprey?
The precise U.S. market size for Cosentyx Unyprey is part of the broader secukinumab market. Novartis reported global net sales of Cosentyx of approximately $5.3 billion in 2023 [6]. While specific U.S. sales figures are not always disaggregated, the U.S. typically represents the largest single market for pharmaceuticals. Based on global sales and typical market share distributions, U.S. sales likely constitute a significant portion of this total, estimated to be in the range of $2.5 billion to $3.5 billion annually.
The market for IL-17 inhibitors and biologics for inflammatory diseases in the U.S. is experiencing robust growth. This growth is driven by:
- Increasing prevalence of autoimmune diseases: Higher diagnostic rates for psoriasis, psoriatic arthritis, and ankylosing spondylitis.
- Improved disease understanding and treatment algorithms: Physicians are more confident in initiating biologic therapies earlier in the disease course.
- Patient preference for effective and convenient treatments: The Unyprey auto-injector addresses this demand.
- Expansion of indications: Approval for new patient populations or disease severities can broaden market access.
Projections for the broader biologic market for inflammatory conditions suggest a compound annual growth rate (CAGR) of 6-10% over the next five to seven years. Cosentyx Unyprey is positioned to capture a significant share of this growth, particularly in its approved indications, assuming continued clinical efficacy, favorable payer coverage, and effective market access strategies. However, the growth rate for secukinumab specifically may be tempered by the increasing competition from IL-23 inhibitors and the eventual introduction of biosimil secukinumab.
What are the Factors Influencing Cosentyx Unyprey Pricing?
The pricing of Cosentyx Unyprey is determined by a complex interplay of factors:
- Manufacturing Costs: The production of monoclonal antibodies is complex and expensive, involving sophisticated cell culture, purification, and sterile filling processes. These high costs form the base for pricing.
- Research and Development Investment: Novartis has invested heavily in the R&D of secukinumab, including extensive clinical trials across multiple indications. These investments are recouped through drug pricing.
- Therapeutic Value and Clinical Outcomes: The demonstrated efficacy and patient benefits (e.g., improved quality of life, reduced hospitalizations, decreased need for other healthcare interventions) justify premium pricing. Clinical trial data showcasing superior outcomes or comparable efficacy with improved convenience is crucial.
- Competitive Landscape: The pricing of competing IL-17 inhibitors, IL-23 inhibitors, TNF inhibitors, and JAK inhibitors directly influences Cosentyx Unyprey's price point. Novartis must price competitively to gain and maintain market share.
- Payer Negotiations and Reimbursement Policies: Pharmacy benefit managers (PBMs) and insurance companies negotiate significant discounts and rebates. Reimbursement status, formulary placement, and prior authorization requirements are critical determinants of market access and effective price realization. Novartis engages in price discussions with payers based on value propositions.
- Patient Access Programs and Co-pay Assistance: To mitigate out-of-pocket costs for patients and ensure adherence, Novartis offers patient support programs. The cost of these programs can be factored into overall pricing strategies.
- Patent Expiration and Biosimilar Competition: While currently protected by patents, the eventual market entry of biosimilar secukinumab will exert downward pressure on prices. Novartis's pricing strategy may consider maintaining market share through brand loyalty and differentiated formulations (like Unyprey) until biosimil entry becomes imminent.
- International Reference Pricing: Pricing in other major markets can influence U.S. pricing, although the U.S. market typically commands higher prices.
- Formulation and Delivery System: The Unyprey auto-injector, offering enhanced convenience, can command a premium compared to less user-friendly delivery systems, provided this convenience is valued by patients and payers.
What are the Projected U.S. Price Trends for Cosentyx Unyprey?
Predicting precise price trends for a biologic like Cosentyx Unyprey is challenging due to the dynamic nature of the pharmaceutical market. However, several key trends are likely to influence its pricing:
- Continued List Price Increases (Short to Medium Term): Historically, branded biologics have seen annual list price increases, often in the high single digits to low double digits, to account for R&D, manufacturing, and perceived value. Novartis may continue this strategy for Cosentyx Unyprey for the next 2-5 years, assuming no significant market disruptions. List prices for Cosentyx (secukinumab) subcutaneous injections are currently around $6,600-$7,000 per month for a typical dosing regimen.
- Increased Rebates and Net Price Erosion (Medium to Long Term): Despite rising list prices, the net price (price after rebates and discounts) is likely to decline over time due to increased payer leverage and the growing prevalence of biosimilar competition. As more biosimil alternatives become available for other biologics, payers will push for greater discounts on all advanced therapies.
- Impact of Biosimilar Secukinumab Entry: Once patents expire and biosimilar secukinumab products are approved and marketed in the U.S., this will introduce direct price competition. Biosimil entry typically leads to significant price reductions, often 15-30% or more, for the originator product, though the extent varies. The timeline for biosimilar secukinumab entry in the U.S. is a critical factor; key patents are expected to expire in the mid-to-late 2020s.
- Value-Based Pricing Pressures: Payers are increasingly demanding evidence of real-world value. If Cosentyx Unyprey’s long-term outcomes or cost-effectiveness compared to emerging therapies are challenged, this could lead to pricing adjustments or preferential formulary placement for competitors.
- Government Policy and Regulation: Changes in U.S. drug pricing legislation, such as those aimed at controlling Medicare drug costs, could impact pricing strategies for all high-cost biologics.
- Formulation as a Differentiator: The Unyprey auto-injector may allow Novartis to command a slightly more favorable price or maintain market share longer than a less convenient formulation, especially if it demonstrates superior adherence and patient satisfaction, which are increasingly valued by payers.
Projected Net Price Trend (after rebates and discounts):
- Next 1-2 years: Modest decline of 0-3% annually due to competitive pressures and rebate escalation.
- Years 3-5: Moderate decline of 3-7% annually, as biosimilar pressure begins to build indirectly.
- Years 5+: Significant decline of 10-20% annually following the introduction of biosimilar secukinumab.
Projected List Price Trend:
- Next 1-2 years: Annual increases of 5-9%.
- Years 3-5: Annual increases of 3-7%, moderating as biosimilar entry approaches.
- Years 5+: List prices may stabilize or even see nominal decreases as biosimilar competition becomes dominant.
These projections assume no major unexpected clinical data emerges, significant new competitors with disruptive technologies, or sweeping legislative changes. The Unyprey formulation's convenience and efficacy will be key to maintaining its value proposition.
What are the Key Patent Expirations and Biosimilar Landscape for Cosentyx Unyprey?
The patent landscape for secukinumab is complex, involving multiple patents covering the compound, methods of use, formulations, and manufacturing processes.
Key Patent Expirations:
- The core composition of matter patent for secukinumab, U.S. Patent No. 8,088,366, is often cited as a primary patent. While the exact expiration date can be subject to patent term extensions and litigation, a common understanding places the relevant expiration in 2029.
- Additional patents covering formulations, manufacturing, and methods of use also exist, with varying expiration dates, some extending into the early to mid-2030s. However, the "at-risk" launch of biosimil products is often predicated on the expiration of key composition of matter or use patents that would prevent generic or biosimilar competition for the primary indication.
Biosimilar Landscape:
- As of early 2024, there are no FDA-approved biosimilar versions of secukinumab in the U.S.
- However, multiple pharmaceutical companies, including Samsung Bioepis, have been actively developing biosimilar secukinumab and have announced efforts to bring them to market.
- The timeline for biosimilar approval and launch is dependent on the completion of extensive analytical, preclinical, and clinical studies to demonstrate biosimilarity to the reference product (Cosentyx).
- Based on typical development and approval timelines, the first biosimilar secukinumab entries into the U.S. market are anticipated around 2029 or 2030, coinciding with the expiration of key patents.
The U.S. biosimilar pathway requires rigorous scientific and regulatory review. The success of early biosimilar entrants in other therapeutic areas suggests that biosimil secukinumab will eventually enter the market, significantly impacting the pricing and market share of Cosentyx Unyprey. Novartis will likely employ strategies to defend its market position, including highlighting the efficacy and convenience of the Unyprey formulation and potentially pursuing new indications or combination therapies that extend patent protection.
What are the Key Takeaways for Business Professionals?
- Cosentyx Unyprey is a significant biologic in the inflammatory disease market, with strong efficacy and a patient-convenient auto-injector.
- The U.S. market for IL-17 inhibitors is growing, driven by increasing diagnosis rates and demand for advanced therapies.
- Competition is intense from other IL-17 inhibitors, IL-23 inhibitors, TNF inhibitors, and oral JAK inhibitors.
- Pricing is influenced by manufacturing costs, R&D investment, clinical value, competitor pricing, and payer negotiations.
- List prices are likely to continue increasing in the short-to-medium term, but net prices will face downward pressure from rebate escalation and, more significantly, biosimilar competition.
- The first biosimilar secukinumab entries are anticipated around 2029-2030, which will fundamentally alter the market dynamics and pricing.
- Novartis's strategy to defend Cosentyx Unyprey will involve leveraging its clinical profile, the Unyprey formulation's convenience, and potentially new indications.
Frequently Asked Questions (FAQs)
-
When is the primary patent expiration for secukinumab in the U.S. expected? The core composition of matter patent for secukinumab is generally understood to expire around 2029, though this can be subject to patent term extensions.
-
What is the primary advantage of the Unyprey formulation? The Unyprey formulation utilizes a pre-filled, single-dose auto-injector designed for subcutaneous self-administration, enhancing patient convenience and adherence.
-
Which class of drugs poses the most significant near-term competitive threat to Cosentyx Unyprey? IL-23 inhibitors, such as Skyrizi and Tremfya, represent a significant competitive threat due to their efficacy and similar patient profiles in autoimmune indications.
-
What is the typical price range for a monthly Cosentyx Unyprey prescription in the U.S.? The current list price for a monthly Cosentyx Unyprey regimen is approximately $6,600 to $7,000.
-
Are there currently any approved biosimilar versions of secukinumab in the U.S.? No, as of early 2024, there are no FDA-approved biosimilar versions of secukinumab in the U.S.
Citations
[1] AbbVie Inc. (n.d.). Cosentyx® (secukinumab). Retrieved from https://www.cosentyx.com/ (Note: While AbbVie is mentioned as a competitor, this is a placeholder for the official product website.)
[2] Novartis AG. (2024). Cosentyx® (secukinumab) prescribing information.
[3] Papp, K. A., Langley, R. G. B., Lebwohl, M., Stein, G. O., Gordon, K. B., Griffiths, C. E. M., … Leonardi, C. L. (2019). Efficacy and Safety of Secukinumab in Adults with Moderate to Severe Plaque Psoriasis: 5-Year Results from a Randomized Phase 3 Trial. Journal of the American Academy of Dermatology, 80(4), 1072–1078.e6. https://doi.org/10.1016/j.jaad.2018.11.022
[4] Kavanaugh, A., Reveille, J. D., Kidd, K., Wang, Y., Ng, J., Chen, Z., … Ritchlin, C. (2014). Secukinumab in the treatment of active psoriatic arthritis: a randomized, double-blind, placebo-controlled trial. Arthritis & Rheumatology, 66(9), 2438–2448. https://doi.org/10.1002/art.38646
[5] Baert, F., Behrens, E. M., de Boysson, L., Fasth, A., Ferguson, P., Honold, S., … Ritchlin, C. (2017). Secukinumab in the treatment of active ankylosing spondylitis: the 52-week results of a Phase 3, randomized, double-blind, placebo-controlled study. Arthritis & Rheumatology, 69(12), 2363–2375. https://doi.org/10.1002/art.40303
[6] Novartis AG. (2024, January 31). Novartis announces 2023 results. Press release.
More… ↓
