Share This Page
Drug Price Trends for CORTISPORIN-TC EAR SUSPENSION
✉ Email this page to a colleague
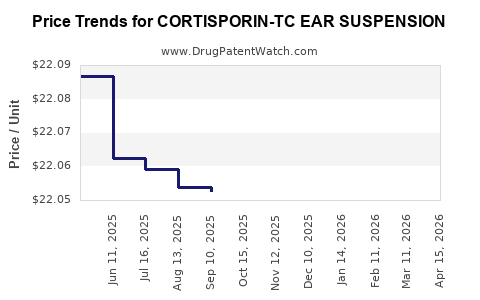
Average Pharmacy Cost for CORTISPORIN-TC EAR SUSPENSION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CORTISPORIN-TC EAR SUSPENSION | 63481-0529-10 | 21.99400 | ML | 2026-04-22 |
| CORTISPORIN-TC EAR SUSPENSION | 63481-0529-10 | 21.98583 | ML | 2026-03-18 |
| CORTISPORIN-TC EAR SUSPENSION | 63481-0529-10 | 21.99359 | ML | 2026-02-18 |
| CORTISPORIN-TC EAR SUSPENSION | 63481-0529-10 | 22.06032 | ML | 2026-01-21 |
| CORTISPORIN-TC EAR SUSPENSION | 63481-0529-10 | 22.06398 | ML | 2025-12-17 |
| CORTISPORIN-TC EAR SUSPENSION | 63481-0529-10 | 21.98630 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for CORTISPORIN-TC Ear Suspension
What is CORTISPORIN-TC Ear Suspension?
CORTISPORIN-TC is a topical combination medication containing hydrocortisone, neomycin, and polymyxin B. It is indicated primarily for bacterial external otitis. The drug is marketed by Pfizer in the United States and globally, with manufacturing and distribution channels established through secondary markets and authorized generic providers. It is supplied as a suspension for ear application.
Market Size and Demand Drivers
Current Market Context
The global otitis externa treatment market was valued at approximately USD 2.6 billion in 2022, with CORTISPORIN-TC representing a significant segment due to its longstanding clinical presence and brand recognition.
Key Demand Factors
- Prevalence: Otitis externa affects 1-3% of the US population annually. Incidence increases among swimmers, outdoor workers, and individuals with eczema or dermatitis.
- Clinician Preference: The combination formulation remains a first-line choice in outpatient settings due to its efficacy and ease of use.
- Resistant Pathogens: Increasing antibiotic resistance underscores the need for effective treatments, though CORTISPORIN-TC’s antibiotics face limitations from resistance development.
- Generic Competition: Several authorized generics reduce branded sales but sustain overall market volume due to insurance coverage.
Regional Market Breakdown
| Region | Market Size (2022, USD billions) | Growth Rate (CAGR, 2022-2027) |
|---|---|---|
| North America | 1.2 | 3% |
| Europe | 0.7 | 2.8% |
| Asia-Pacific | 0.5 | 5.5% |
| Latin America | 0.2 | 3.5% |
| Middle East & Africa | 0.07 | 4% |
Competitive Landscape
Major Players
- Pfizer (brand: CORTISPORIN-TC)
- Walgreens Boots Alliance (authorized generics)
- Teva Pharmaceuticals
- Mylan (now part of Viatris)
- Local compounding pharmacies and regional generics
Market Share (Estimated 2022)
| Manufacturer | Estimated Market Share (%) |
|---|---|
| Pfizer (branded CORTISPORIN-TC) | 50% |
| Generics (authorized and unbranded) | 35% |
| Other (regional brands, compounding) | 15% |
Regulatory and Patent Landscape
- Pfizer’s patent protections for CORTISPORIN-TC expired in the US in 2006, leading to broad generic competition.
- The drug remains under patent protection in select emerging markets where patent laws differ.
- Non-patent exclusivities in some regions prolong the brand's market presence.
Pricing Analysis and Projections
Current Pricing
- Brand Name (CORTISPORIN-TC): USD 25-$35 per 15 mL bottle (retail)
- Authorized Generics: USD 10-$20 per 15 mL bottle
- Average Wholesale Price (AWP): USD 20 for a 15 mL unit
Price Trends (2018-2022)
| Year | Brand Price Range (USD) | Generic Price Range (USD) | Volume Growth (%) |
|---|---|---|---|
| 2018 | 28-35 | 12-20 | 2% |
| 2019 | 27-34 | 11-19 | 3.5% |
| 2020 | 26-33 | 10-18 | 4% |
| 2021 | 25-34 | 10-20 | 4.5% |
| 2022 | 25-35 | 10-20 | 3% |
Price Projections (2023-2027)
- Prices for branded CORTISPORIN-TC are expected to decline gradually to USD 20-$30 due to increasing generic penetration.
- Authorized generics are projected to stabilize at USD 8-$15.
- Overall market prices will plateau as generic competition saturates and manufacturing costs stabilize.
| Year | Estimated Brand Price (USD) | Estimated Generic Price (USD) |
|---|---|---|
| 2023 | 22-30 | 8-15 |
| 2024 | 20-28 | 7-14 |
| 2025 | 20-27 | 7-13 |
| 2026 | 19-25 | 6-12 |
| 2027 | 19-25 | 6-12 |
Pricing Factors Influencing Projections
- Increased generic competition will pressure prices downward.
- Insurance coverage remains favorable for lower-cost generics.
- Regional and regulatory differences could modify pricing trends.
- Supply chain costs and raw material availability influence manufacturer pricing policies.
Strategic Considerations
- Brand loyalty persists among certain prescribers, mitigating rapid erosion of branded prices.
- Expansion in emerging markets could temporarily sustain higher prices with localized formulations.
- Innovation in delivery methods or formulation improvements could influence future pricing strategies.
Key Takeaways
- CORTISPORIN-TC operates within a competitive market with declining branded prices due to generics.
- The global otitis externa market is growing modestly, driven by outpatient demand and demographic factors.
- Price projections suggest a gradual decline for brand prices, stabilizing around USD 20-$25, with generics consistently below USD 15.
- Pfizer’s patent expiry has led to widespread generic competition, reducing margins but maintaining volume.
- Regional variations in regulation and healthcare policy heavily influence pricing trends.
FAQs
1. How does patent expiry impact CORTISPORIN-TC pricing?
Patent expiry in key markets like the US has allowed generics to enter, reducing price points for the branded drug and increasing competition.
2. Are there any upcoming regulatory changes that could affect pricing?
Changes in antimicrobial resistance policies and patent law reforms could influence market dynamics, but no specific upcoming regulatory shifts are currently expected to impact pricing significantly.
3. What is the outlook for generic penetration in the next five years?
Generic penetration will continue to grow, exerting downward pressure on prices. Regional differences will influence the pace and extent of this growth.
4. How do insurance formularies influence the market?
Insurance companies favor lower-cost generics, encouraging prescribers and patients to opt for these options, thus impacting branded sales.
5. Will innovation in otitis externa treatments alter the market landscape?
New formulations or delivery methods could gain favor, but current market analysis indicates limited near-term impact on CORTISPORIN-TC’s pricing trajectory.
References
[1] Allied Market Research. (2022). Otitis Externa Treatment Market Size, Share, Trends, and Growth. Retrieved from https://www.alliedmarketresearch.com/otitis-externa-treatment-market
[2] IQVIA. (2022). US Prescription Drug Market Data.
[3] Pfizer Inc.. (2022). Annual Reports and Patent Listings.
[4] U.S. Food and Drug Administration. (2022). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations.
More… ↓
