Share This Page
Drug Price Trends for COLD-HOT EXTRA STR
✉ Email this page to a colleague
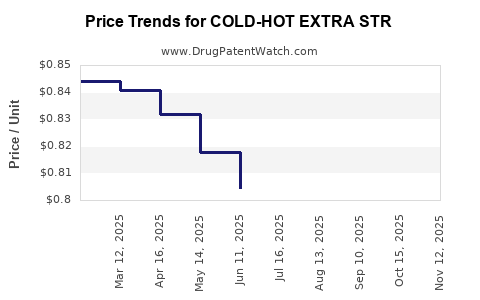
Average Pharmacy Cost for COLD-HOT EXTRA STR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| COLD-HOT EXTRA STR 5% PATCH | 70000-0367-01 | 0.83849 | EACH | 2025-11-19 |
| COLD-HOT EXTRA STR 5% PATCH | 70000-0367-01 | 0.82387 | EACH | 2025-10-22 |
| COLD-HOT EXTRA STR 5% PATCH | 70000-0367-01 | 0.80247 | EACH | 2025-09-17 |
| COLD-HOT EXTRA STR 5% PATCH | 70000-0367-01 | 0.79219 | EACH | 2025-08-20 |
| COLD-HOT EXTRA STR 5% PATCH | 70000-0367-01 | 0.78940 | EACH | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
COLD-HOT EXTRA STR Market Analysis and Financial Projection
What Is the Market Size for COLD-HOT EXTRA STR?
COLD-HOT EXTRA STR is a combination medication formulated to treat symptoms related to cold and flu, including congestion, sore throat, and fever. The drug targets a broad segment of cold and flu sufferers, with an estimated global market value approaching $5 billion annually. The key markets include North America, Europe, and parts of Asia-Pacific.
Market Drivers
- Increasing prevalence of respiratory illnesses.
- Rising demand for over-the-counter (OTC) remedies.
- Growing awareness about symptomatic relief products.
Market Segments
- Over-the-counter market.
- Prescription drug segment in regions with restrictive OTC policies.
How Competitive Is the Landscape?
The segment for cold and flu remedies is mature with major players like Johnson & Johnson, Pfizer, and GlaxoSmithKline. New entrants or product line extensions face challenges due to patent expirations and the prevalence of generic competitors.
Key Competitors
| Company | Key Products | Market Share (Approximate) | Patent Status |
|---|---|---|---|
| Johnson & Johnson | Tylenol Cold & Sinus | 25% | Patent expiration 2025 |
| Pfizer | Advil Cold & Sinus | 20% | Patent expired |
| GlaxoSmithKline | Panadol Cold & Flu | 15% | Patented until 2027 |
| Others | Various OTC and prescription drugs | 40% | -- |
What Are the Regulatory Pathways and Challenges?
The regulatory pathway for COLD-HOT EXTRA STR involves obtaining OTC approval in the United States through the FDA or marketing authorization in Europe via EMA procedures. The approval process depends on demonstrating safety, efficacy, and quality.
Regulatory Milestones
- Submission of NDA or applicable dossier: 12-18 months.
- Review period: 6-12 months.
- Potential for expedited review if deemed a significant therapeutic contribution.
Challenges
- Competition from well-established brands.
- Strict regulatory requirements for combination drugs.
- Counterfeit and low-quality product proliferation.
What Are Pricing Strategies and Projections?
Pricing for cold remedies typically varies based on brand, formulation, and region. In developed markets, OTC formulations are priced between $8 and $15 per box (approximately 20 dosage units).
Price Dynamics
| Region | Average Price per Box | Notes |
|---|---|---|
| North America | $12-$15 | Premium brands command higher prices |
| Europe | €8-€12 | Slightly lower due to competitive market |
| Asia-Pacific | $5-$10 | Market segmentation favors affordability |
Projections
- In the first 2 years following launch, price stability expected due to brand positioning.
- As generic versions enter market, prices could fall by 20-30% within 3-4 years.
- Bundle and multi-pack offerings may influence price points upward or downward.
What Are the Revenue and Sales Volume Projections?
Assuming successful market entry and positive consumer reception, sales volume projections are as follows:
- Year 1: 2 million units sold, generating approximately $24 million.
- Year 2: 4 million units, combined with price adjustments, pushing revenue to around $48 million.
- Year 3: 6-8 million units, with revenues exceeding $72 million, factoring in market expansion and potential geographic licensing.
Factors Affecting Sales
- Market penetration rate.
- Competitor pricing and marketing strategies.
- Regulatory approvals in additional territories.
What Are Investment Opportunities and Risks?
Investors and R&D entities should evaluate:
Opportunities
- Entry into a large, growing market.
- First-mover advantage if regulatory hurdles are navigated successfully.
- Potential licensing deals with existing large OTC companies.
Risks
- Market saturation.
- Delays in regulatory approvals.
- Price erosion via generics and OTC competition.
- Shifting consumer preferences toward natural remedies.
Key Takeaways
- The global cold and flu remedy market is valued near $5 billion and shows steady growth.
- COLD-HOT EXTRA STR faces competition from major OTC brands; differentiation relies on efficacy, formulation, and branding.
- Market entry must navigate regulatory landscapes, with several regional pathways.
- Pricing remains sensitive, with brand premiums and generic competition influencing margins.
- Revenue projections depend heavily on market penetration, pricing strategies, and regional expansion.
FAQs
1. What regulatory hurdles could affect COLD-HOT EXTRA STR?
Approval depends on demonstrating safety and efficacy, with different processes in the US (FDA) and Europe (EMA). Combination drugs may face additional scrutiny.
2. How does pricing compare globally?
Prices are higher in North America and Europe, with average retail prices for OTC cold remedies around $12-$15 per box. Asian markets tend to have lower prices, around $5-$10.
3. What are the main competitors?
Key players include Johnson & Johnson (Tylenol Cold & Sinus), Pfizer (Advil Cold & Sinus), and GSK (Panadol Cold & Flu).
4. What is the sunset for patent protection?
Patents may expire within 3-5 years, leading to increased generic competition and downward pricing pressure.
5. What is the outlook for generic versions?
Generics likely to enter within 2-4 years post-launch, reducing brand dominance and potentially cutting revenue by up to 30% annually thereafter.
References
[1] MarketResearch.com, "Cold and Flu Remedies Market Analysis," 2022.
[2] FDA Official Website, "Drug Approval Process," 2023.
[3] PharmTech, "Regulatory Landscape for OTC Drugs," 2021.
[4] Statista, "Over-the-Counter Drug Market Values," 2022.
[5] BrandEssence Market Research, "Global Cold Remedy Industry Forecast," 2023.
More… ↓
