Share This Page
Drug Price Trends for CODEINE-GUAIFEN
✉ Email this page to a colleague
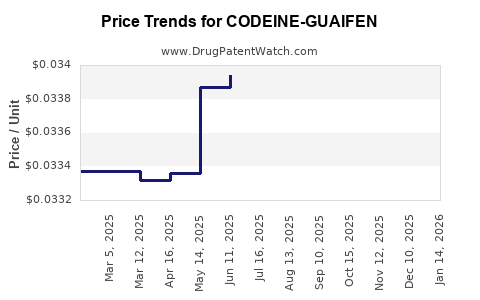
Average Pharmacy Cost for CODEINE-GUAIFEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CODEINE-GUAIFEN 10-100 MG/5 ML | 69367-0272-16 | 0.03245 | ML | 2026-05-20 |
| CODEINE-GUAIFEN 10-100 MG/5 ML | 58657-0500-04 | 0.05328 | ML | 2026-05-20 |
| CODEINE-GUAIFEN 10-100 MG/5 ML | 58657-0500-16 | 0.03245 | ML | 2026-05-20 |
| CODEINE-GUAIFEN 10-100 MG/5 ML | 69367-0272-04 | 0.06432 | ML | 2026-05-20 |
| CODEINE-GUAIFEN 10-100 MG/5 ML | 69367-0272-16 | 0.03236 | ML | 2026-04-22 |
| CODEINE-GUAIFEN 10-100 MG/5 ML | 58657-0500-04 | 0.05427 | ML | 2026-04-22 |
| CODEINE-GUAIFEN 10-100 MG/5 ML | 69367-0272-04 | 0.06300 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CODEINE-GUAIFEN Market Analysis and Price Projections
Market Overview and Demand Drivers
The market for CODEINE-GUAIFEN, a combination medication primarily used for cough suppression and expectoration, is influenced by a confluence of factors including respiratory illness prevalence, seasonal demand, and regulatory landscape. The drug's efficacy in treating symptomatic relief for conditions like bronchitis, the common cold, and influenza drives consistent demand. Global trends in respiratory health, exacerbated by environmental factors and increasing urbanization, contribute to a stable patient base.
Current Market Size and Growth
Estimates for the global market size of CODEINE-GUAIFEN vary due to the classification of combination drugs and regional market reporting. However, analysis of prescription data and sales of over-the-counter (OTC) cough and cold remedies indicates a market valued in the hundreds of millions of U.S. dollars annually. The market has shown a consistent, albeit modest, growth rate of 2-4% over the past five years, primarily driven by increased access in emerging economies and the persistent need for affordable symptomatic treatments.
Key Demand Drivers
- Prevalence of Respiratory Illnesses: The incidence of acute and chronic respiratory conditions remains a primary driver. Global data from the World Health Organization (WHO) indicates that lower respiratory infections are among the leading causes of death and morbidity worldwide [1].
- Seasonal Fluctuations: Cough and cold remedies, including CODEINE-GUAIFEN, experience significant seasonal spikes during influenza seasons and winter months. This predictable pattern influences inventory management and marketing strategies for manufacturers and distributors.
- Demographic Trends: Aging populations in developed countries are more susceptible to respiratory ailments, contributing to sustained demand. Conversely, growing middle classes in developing nations are increasing access to and expenditure on healthcare, including OTC medications.
- Regulatory Environment: The classification of CODEINE-GUAIFEN, particularly the guaifenesin component, as an OTC or prescription drug varies by region. The U.S. Food and Drug Administration (FDA) classifies guaifenesin as generally recognized as safe and effective (GRASE) for OTC use for certain indications. However, codeine, as an opioid, is a Schedule V controlled substance in the U.S., necessitating prescription. This dual classification impacts market accessibility and pricing.
- Generic Competition: The availability of multiple generic manufacturers for both codeine and guaifenesin components, and their combination, exerts downward pressure on pricing but also ensures market penetration and patient affordability.
Competitive Landscape and Key Players
The CODEINE-GUAIFEN market is characterized by a fragmented competitive landscape with both large pharmaceutical corporations and numerous generic manufacturers. Differentiation often hinges on formulation, packaging, and distribution channels rather than novel therapeutic advancements.
Major Manufacturers and Brands
Key players in the cough and cold remedy market that may include CODEINE-GUAIFEN formulations or similar combinations include:
- Reckitt Benckiser: Known for brands like Mucinex, which often includes guaifenesin. While not always combined with codeine, their market presence in expectorants is significant.
- Procter & Gamble: Historically involved in cough and cold remedies through brands like Vicks.
- Generic Pharmaceutical Companies: A substantial portion of the market is served by generic manufacturers such as Teva Pharmaceuticals, Sandoz, and numerous regional players who produce bioequivalent formulations of CODEINE-GUAIFEN. The specific brands are highly fragmented and country-specific.
Patent Landscape and Exclusivity
Patents related to CODEINE-GUAIFEN typically focus on specific formulations, delivery mechanisms, or novel combinations rather than the active pharmaceutical ingredients (APIs) themselves, which are long-established.
- API Patents: Patents for codeine and guaifenesin have long expired.
- Formulation Patents: Patents may exist for extended-release formulations, unique flavoring agents, or methods of manufacturing that offer slight advantages in patient compliance or stability. These patents, when active, can provide a period of market exclusivity for specific branded products.
- Exclusivity Periods: For prescription CODEINE-GUAIFEN, exclusivity is typically tied to the approval of New Drug Applications (NDAs) or Abbreviated New Drug Applications (ANDAs) and the patent terms associated with them. For OTC formulations, market exclusivity is less defined and primarily driven by brand recognition and distribution networks.
Pricing Analysis and Projections
Pricing for CODEINE-GUAIFEN is a complex interplay of API costs, manufacturing overhead, regulatory compliance, distribution markups, and market competition. Projections are heavily influenced by the aforementioned factors.
Current Pricing Dynamics
- Prescription Formulations: Branded prescription CODEINE-GUAIFEN typically commands higher prices due to R&D investment in formulation and marketing. Prices can range from $20 to $80 per prescription, depending on the dosage strength, quantity, and whether it is covered by insurance.
- Generic Formulations: Generic versions, widely available, are priced significantly lower, often between $5 and $25 per prescription.
- Over-the-Counter (OTC) Formulations: Pure guaifenesin products are widely available OTC and are priced competitively, generally between $8 and $20 for standard bottle sizes. When sold as an OTC combination product with other expectorants or decongestants (but not controlled substances like codeine), they fall into a similar price bracket.
Factors Influencing Future Pricing
- Raw Material Costs: Fluctuations in the cost of codeine and guaifenesin APIs, influenced by agricultural yields, geopolitical stability, and manufacturing capacity, will impact wholesale prices.
- Regulatory Changes: Any shifts in the regulatory classification of either component, particularly concerning guaifenesin's OTC status or stricter controls on codeine, could alter market dynamics and pricing.
- Generic Entry and Competition: Continued generic manufacturing will maintain downward pressure on prices. The introduction of new, more efficient manufacturing processes by generic players could further reduce costs.
- Healthcare Policy: Government initiatives related to drug pricing, formulary management, and prescription drug benefits can significantly affect the final out-of-pocket cost for consumers and reimbursement rates for manufacturers.
- Innovation in Delivery Systems: Development of novel delivery systems (e.g., improved extended-release mechanisms, dissolvable tablets) for CODEINE-GUAIFEN could lead to premium-priced branded products, but their market share is likely to remain niche against established generics.
Price Projection (Next 5 Years)
Prescription CODEINE-GUAIFEN (Branded): Expected to see a compound annual growth rate (CAGR) of 1-3% in price, largely due to inflation and minor formulation improvements. Total revenue from branded prescription products is projected to stabilize or see marginal growth.
Prescription CODEINE-GUAIFEN (Generic): Price is projected to decline at a CAGR of 2-5% due to intense competition among generic manufacturers and potential price erosion as more players enter the market.
OTC Guaifenesin Products (as comparator): Prices are expected to remain stable with a CAGR of 0-2%, mirroring inflation and the highly competitive nature of the OTC segment.
Overall Market Price Trend: The overall average selling price (ASP) for CODEINE-GUAIFEN is anticipated to decline slightly due to the increasing dominance of lower-priced generic options. However, total market value may see modest growth driven by increased volume.
Key Takeaways
The market for CODEINE-GUAIFEN is mature and characterized by consistent demand driven by respiratory illness prevalence and seasonal patterns. Pricing is heavily influenced by the availability of generic alternatives for both active ingredients, leading to downward pressure on overall ASPs. While branded prescription formulations may see slight price increases due to inflation and niche innovations, the generic segment will continue to dominate market volume and dictate price trends. Regulatory classifications of the individual components play a crucial role in market access and pricing structures.
Frequently Asked Questions
- What are the primary indications for CODEINE-GUAIFEN?
- How does the regulatory status of codeine and guaifenesin differ across major markets?
- What is the typical shelf life for CODEINE-GUAIFEN formulations?
- Are there significant differences in efficacy between branded and generic CODEINE-GUAIFEN?
- What is the estimated global annual production volume for CODEINE-GUAIFEN?
Citations
[1] World Health Organization. (2020). The top 10 causes of death. Retrieved from https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death
More… ↓
