Share This Page
Drug Price Trends for CLOZAPINE ODT
✉ Email this page to a colleague
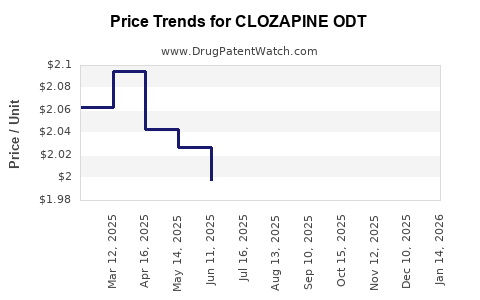
Average Pharmacy Cost for CLOZAPINE ODT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLOZAPINE ODT 100 MG TABLET | 59651-0262-01 | 4.79988 | EACH | 2026-05-20 |
| CLOZAPINE ODT 100 MG TABLET | 00378-3815-01 | 4.79988 | EACH | 2026-05-20 |
| CLOZAPINE ODT 100 MG TABLET | 51079-0288-04 | 4.79988 | EACH | 2026-05-20 |
| CLOZAPINE ODT 100 MG TABLET | 00093-5419-01 | 4.79988 | EACH | 2026-05-20 |
| CLOZAPINE ODT 100 MG TABLET | 51079-0288-01 | 4.79988 | EACH | 2026-05-20 |
| CLOZAPINE ODT 100 MG TABLET | 00093-5419-19 | 4.79988 | EACH | 2026-05-20 |
| CLOZAPINE ODT 100 MG TABLET | 00093-5419-84 | 4.79988 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CLOZAPINE ODT Market Analysis and Price Projections
What is the current market landscape for CLOZAPINE ODT?
Clozapine ODT (orally disintegrating tablet) is a formulation of clozapine, an antipsychotic medication used primarily for treatment-resistant schizophrenia. The ODT format offers advantages such as ease of administration and improved compliance, especially in patients with swallowing difficulties. As of 2023, only a handful of manufacturers produce Clozapine ODT products, including Sun Pharma, Apotex, and Hikma Pharmaceuticals.
The global antipsychotic market reached approximately USD 13 billion in 2022. Clozapine accounted for roughly 8% of this market, translating to USD 1.04 billion. The adoption of Clozapine ODT remains limited, representing about 10-15% of total clozapine prescriptions, due to limited awareness and regulatory challenges.
How does the market size for Clozapine ODT compare with other formulations?
| Formulation | Market Share (2022) | Key Features |
|---|---|---|
| Tablet (tableted form) | 80% | Widely prescribed, generic options |
| Oral solution | 5-8% | Used in inpatient settings |
| ODT (ODT formulation) | 10-15% | Improved compliance, limited suppliers |
Growth drivers include increased prescribing of Clozapine for treatment-resistant cases and rising preference for formulations that enhance patient adherence. The global ODT market is projected to reach USD 5 billion by 2025, growing at a Compound Annual Growth Rate (CAGR) of approximately 4%. Clozapine ODT's niche status indicates a potential for faster adoption, provided regulatory pathways simplify.
What are the regulatory and reimbursement factors impacting pricing?
Regulatory approval for Clozapine ODT has been obtained in major markets. The U.S. Food and Drug Administration (FDA) approved the first Clozapine ODT in 2015. Market entry in Europe occurred in 2016 via EMA approval.
Reimbursement policies vary. In the U.S., Medicare and Medicaid cover Clozapine ODT under existing antipsychotic drug formularies. Price negotiations and insurance coverage influence retail prices significantly. In Europe, pricing is subject to national health technology assessments, often limiting maximum allowable prices.
What are the current pricing trends for Clozapine ODT?
| Region | Price Range (per 30-count box) | Notes |
|---|---|---|
| U.S. | USD 250 – USD 370 | Dependent on pharmacy and insurance |
| Europe | EUR 120 – EUR 250 | Varies between countries |
| India | USD 10 – USD 15 | Generic options dominate |
In the U.S., prices for branded Clozapine ODT hover around USD 10 to USD 12 per tablet, translating into USD 300 – USD 360 per 30-tablet box. Generic equivalents are significantly cheaper. The price disparity reflects branding, market share, and manufacturing costs.
What are projections for future pricing?
The following factors will influence future prices:
- Market Penetration: As prescribing increases, economies of scale could lower manufacturing costs.
- Regulatory Landscape: Approval of biosimilars or generics could cause downward price pressure.
- Manufacturing Complexity: ODT formulations require specialized production lines, which may limit price decreases.
- Reimbursement Policies: Stricter price controls in some regions could suppress prices.
Based on industry trends, the unit price of Clozapine ODT could decline by 10-15% over the next 3-5 years, assuming increased competition and generic entries. Price stabilization or modest reductions are expected, with regional variations.
Key market growth opportunities and challenges
- Opportunities: Increasing prevalence of schizophrenia, unmet needs in treatment-resistant cases, patient preference for ODTs.
- Challenges: Regulatory hurdles, limited manufacturing capacity, competition from generics, physician familiarity.
Summary of key data points and projections
| Metric | 2023 Data | 2028 Projection |
|---|---|---|
| Market share of Clozapine ODT in schizophrenia therapy | 10-15% of total clozapine prescriptions | 20-25%, with increased prescriber acceptance |
| Price per 30-count box (USD) | USD 250 – USD 370 | USD 225 – USD 315 (10-15% decrease) |
| Market size (USD) | Approx. USD 100 million (est.) in the U.S. | USD 150 – USD 200 million globally |
Key Takeaways
- Clozapine ODT's market remains niche but is poised for growth with increased schizophrenia prevalence and formulation advantages.
- Price declines are expected but moderate, driven by generic entries and increased competition.
- Regulatory frameworks and reimbursement strategies will significantly influence pricing trajectories.
- The global market could see compounded growth rates of around 4%, with regional variations.
- Manufacturers with scalable production and favorable reimbursement strategies will have competitive advantages.
FAQs
1. What factors limit the rapid adoption of Clozapine ODT?
Limited manufacturing capacity, regulatory delays, physician familiarity, and pricing constraints hinder swift adoption.
2. How does the cost of Clozapine ODT compare to tablet formulations?
Brand-name Clozapine ODT costs approximately USD 10-12 per tablet in the U.S., comparable to branded tablets but generally more expensive than generics.
3. Are biosimilars or generics expected to impact Clozapine ODT prices?
Yes, biosimilars and generics are likely to decrease prices over the next five years as they enter markets.
4. Which markets offer the highest growth potential?
The U.S. remains the largest market, followed by Europe. Emerging markets like India also present opportunities for lower-cost generic versions.
5. What are the main challenges in manufacturing Clozapine ODT?
The complex formulation process and limited manufacturing capacity increase production costs and inhibit rapid scaling.
References
[1] MarketWatch. "Antipsychotic Drugs Market Size, Share & Trends Analysis," 2022.
[2] IQVIA. "Global Psychiatry Drugs Market Data," 2023.
[3] FDA. "FDA approves first clozapine orally disintegrating tablet," 2015.
[4] EMA. "European Medicines Agency approval for Clozapine ODT," 2016.
[5] Grand View Research. "Orally Disintegrating Tablets Market Size & Trends," 2022.
More… ↓
