Share This Page
Drug Price Trends for CLOTRIMAZOLE-3
✉ Email this page to a colleague
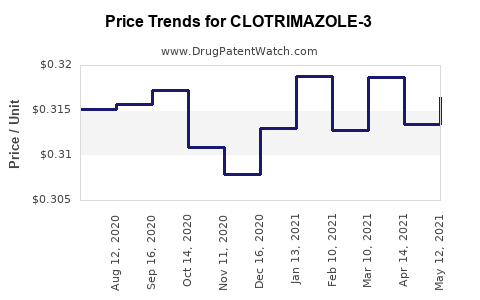
Average Pharmacy Cost for CLOTRIMAZOLE-3
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLOTRIMAZOLE-3 2% CREAM | 24385-0110-09 | 0.30381 | GM | 2026-04-22 |
| CLOTRIMAZOLE-3 2% CREAM | 24385-0110-09 | 0.31625 | GM | 2026-03-18 |
| CLOTRIMAZOLE-3 2% CREAM | 24385-0110-09 | 0.31665 | GM | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Clotrimazole-3
Market Overview
Clotrimazole-3 is an antifungal agent, a derivative of the well-established clotrimazole class. Its primary indications include superficial fungal infections, such as athlete’s foot, candidiasis, and tinea cruris. The drug is positioned as a topical formulation, competing mainly in dermatology and gynecology markets.
The antifungal market volume was valued at approximately USD 4.8 billion in 2022, with a compound annual growth rate (CAGR) of roughly 4.2% projected from 2023 to 2030. Clotrimazole-based products represent about 35% of this market segment, given their extensive use, affordability, and safety profile.
Key Competitive Landscape
Clotrimazole-3 faces competition in the generic antifungal market, primarily from:
- Clotrimazole creams and ointments from global pharmaceutical companies.
- Other azole antifungals such as miconazole, econazole, and ketoconazole.
- Over-the-counter (OTC) products with varying brand recognition and pricing.
Major players include Bayer (Lotrimin), Sandoz, Mylan, and Teva, which dominate sales through both prescription and OTC channels.
Regulatory and Patent Considerations
Clotrimazole-3 is expected to be a generic or biosimilar entry, likely benefiting from existing regulatory approvals for similar formulations. Patent exclusivities on key formulations have mostly expired, permitting broad market entry. Regulatory agencies like the FDA and EMA require bioequivalence data for approval.
The lifecycle of clotrimazole formulations predicts increased generic penetration, reducing average prices over time.
Price Trends and Projections
Current Pricing
- Market retail price for branded clotrimazole creams ranges from USD 10 to USD 20 per 30-gram tube.
- Generics are priced between USD 4 and USD 8.
- OTC formulations are even lower, around USD 2 to USD 4.
Forecasts (2023-2030)
| Year | Average Price (USD per 30g) | Market Share of Generics | Key Factors |
|---|---|---|---|
| 2023 | USD 6.50 | 55% | Post-patent expiry; generic entry stabilizing prices |
| 2025 | USD 5.50 | 75% | Increased competition; OTC growth |
| 2027 | USD 4.80 | 85% | Market saturation, price erosion |
| 2030 | USD 3.50 | 95% | Dominance of generics, low-cost imports |
Market prices are expected to decline by approximately 45% from 2023 to 2030, driven by generic proliferation and OTC availability.
Implications for Stakeholders
- Manufacturers can expect profit margins to narrow, emphasizing efficiency and cost control.
- Investors should monitor regulatory approvals, patent statuses, and competition intensity.
- Healthcare policymakers may influence prices through regulation and reimbursement policies, further impacting market dynamics.
Market Drivers
- Increasing prevalence of superficial fungal infections due to climate change, urbanization, and lifestyle factors.
- Rising awareness and OTC access increase overall market volume.
- Healthcare cost containment pressures push towards generic formulation adoption.
Market Barriers
- Competition from other azoles with superior dosing or safety profiles.
- Regulatory delays or requirements for bioequivalence studies.
- Limited innovation within the topical antifungal class.
Pricing Strategy Recommendations
- Early entry at a competitive price point (below USD 5 per 30g) could capture significant market share.
- Differentiation through formulation improvements, such as enhanced absorption or patient adherence.
- Monitor international markets where healthcare systems may differ in price regulation.
Conclusion
Clotrimazole-3 is positioned for steady, price-driven growth in a mature market characterized by intense competition and declining prices. Entry strategies should focus on cost management, regulatory compliance, and targeted marketing to capture market share rapidly, especially through OTC channels.
Key Takeaways
- The antifungal market valued at USD 4.8 billion globally, with clotrimazole-containing products comprising a significant portion.
- Price declines forecasted to approximately 45% by 2030 due to generic competition.
- Entry price points below USD 5 per 30g enhance market penetration prospects.
- Competition from existing azoles and OTC formulations limits pricing flexibility.
- Regulatory pathways favor broader generic access, supporting price erosion.
FAQs
1. What factors most influence the price decline of Clotrimazole-3?
Market saturation, increased generic competition, and OTC availability drive the price downward over time.
2. How does regulatory approval impact market entry?
Bioequivalence and safety data are required for generics, which can delay entry but ultimately enable market expansion once approved.
3. What is the outlook for branded versus generic sales?
Generics will dominate due to lower prices; branded products will retain niche markets with added value or formulations.
4. How might new formulations affect pricing?
Innovative delivery systems could command premium prices but face challenges from established generics.
5. Are there regional differences in pricing?
Yes. Developed markets tend to have higher prices with stronger brand premiums. Emerging markets often see lower prices due to regulation and purchasing power.
Sources
- MarketWatch, “Antifungal Drug Market Size, Share & Trends," 2023.
- IQVIA, “Global Pharmaceutical Market Data,” 2022.
- U.S. Food and Drug Administration, “Bioequivalence Guidelines,” 2023.
- Statista, “Top Over-the-Counter Antifungal Products,” 2022.
- IMS Health, “Global Patent Expiries and Generic Market Trends," 2022.
More… ↓
