Share This Page
Drug Price Trends for CLOMID
✉ Email this page to a colleague
Average Pharmacy Cost for CLOMID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
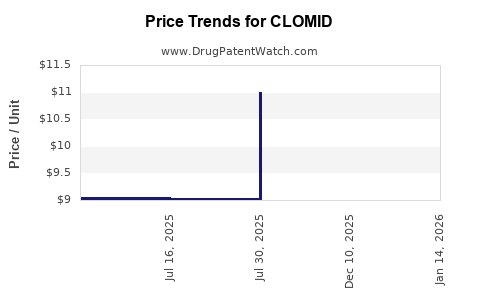
| CLOMID 50 MG TABLET | 00713-0885-09 | 11.16454 | EACH | 2026-04-22 |
| CLOMID 50 MG TABLET | 00713-0885-30 | 11.16454 | EACH | 2026-04-22 |
| CLOMID 50 MG TABLET | 00713-0885-09 | 11.16180 | EACH | 2026-03-18 |
| CLOMID 50 MG TABLET | 00713-0885-30 | 11.16180 | EACH | 2026-03-18 |
| CLOMID 50 MG TABLET | 00713-0885-30 | 11.15501 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CLOMID Market Analysis and Price Projections
Clomiphene citrate, marketed as Clomid, is a selective estrogen receptor modulator (SERM) primarily used to induce ovulation in women with ovulatory dysfunction. The global market for Clomid is influenced by factors including the prevalence of infertility, diagnostic advancements, and evolving treatment protocols. Price projections are contingent on manufacturing costs, regulatory hurdles, generic competition, and market demand.
Market Size and Growth Drivers
The global infertility treatment market, which Clomid significantly contributes to, is projected to reach $47.4 billion by 2028, growing at a compound annual growth rate (CAGR) of 7.5% from 2021 to 2028. This growth is driven by several key factors:
- Rising Infertility Rates: Increasing age at first childbirth, lifestyle factors, and environmental influences contribute to a higher incidence of infertility globally. The World Health Organization (WHO) estimates that 1 in 4 couples in developing countries experience infertility (1).
- Advancements in Assisted Reproductive Technologies (ART): While Clomid is an oral medication, its use as a first-line treatment often precedes more complex ART such as in-vitro fertilization (IVF). Growth in ART indirectly supports the demand for ovulation induction agents.
- Increased Awareness and Diagnosis: Greater public awareness and improved diagnostic tools for identifying ovulatory disorders lead to more women seeking and receiving treatment.
- Cost-Effectiveness: Compared to more advanced ART procedures, Clomid offers a more affordable treatment option for many patients, making it accessible in a wider range of healthcare settings.
Regional Market Dynamics
The market for Clomid is segmented by region, with North America and Europe currently leading in terms of market share due to high healthcare spending, established healthcare infrastructure, and a greater prevalence of infertility awareness. Asia-Pacific is anticipated to exhibit the highest growth rate, driven by increasing disposable incomes, improving healthcare access, and a rising demand for fertility treatments.
Patent Landscape and Generic Competition
The original patent for clomiphene citrate has long expired, allowing for the widespread availability of generic versions. This has significantly impacted pricing and market competition.
- Exclusivity Period: The patent for clomiphene citrate by K.A. Josefsson expired in 1977 (2).
- Generic Penetration: The market is characterized by a high degree of generic penetration, with multiple manufacturers producing clomiphene citrate. This competition generally leads to lower average selling prices.
- Formulation Patents: While the active pharmaceutical ingredient (API) patent has expired, there may be niche patents related to specific formulations, delivery mechanisms, or combination therapies that could offer limited market exclusivity. However, for the standard oral tablet form, significant patent-protected market exclusivity is unlikely.
Pricing Analysis and Projections
The pricing of Clomid is influenced by API manufacturing costs, formulation expenses, regulatory compliance, distribution channels, and the pricing strategies of generic manufacturers.
Current Pricing Benchmarks
The average wholesale price (AWP) for a 30-count bottle of 50 mg clomiphene citrate tablets can range from $20 to $75, depending on the manufacturer and distributor. Patient out-of-pocket costs can vary significantly based on insurance coverage and pharmacy markups. For example, a typical prescription of 15 tablets (one cycle) might cost between $5 and $25 at the retail level for a generic product.
Factors Influencing Future Pricing
- Raw Material Costs: Fluctuations in the cost of raw materials required for API synthesis can impact manufacturing expenses and subsequently influence drug prices.
- Manufacturing Efficiency: Improvements in manufacturing processes and economies of scale for generic producers can lead to further cost reductions.
- Regulatory Landscape: Changes in regulatory requirements for drug manufacturing, quality control, and marketing can incur additional costs for manufacturers.
- Competition Intensity: The number of active generic manufacturers and their respective market shares will continue to dictate price competition. A consolidated generic market could lead to slight price increases, while an fragmented market will likely maintain downward pressure on prices.
- Demand Shifts: While demand for ovulation induction is expected to remain stable or grow moderately, significant shifts in treatment guidelines favoring other modalities could indirectly affect Clomid pricing by altering market share.
- Emergence of Novel Therapies: The development of new, highly effective, or more convenient infertility treatments could potentially reduce the market share of oral agents like Clomid, leading to strategic pricing adjustments by generic manufacturers to maintain volume.
Price Projections
Given the mature nature of the clomiphene citrate market and the ongoing competition from generic manufacturers, significant price increases are unlikely in the short to medium term.
- Short-Term (1-3 years): Prices are expected to remain relatively stable, with minor fluctuations of +/- 5% due to currency exchange rates, raw material costs, and regional distribution variations. The average price per 50 mg tablet is projected to stay within the $0.50 to $1.50 range.
- Medium-Term (3-5 years): Continued demand for cost-effective fertility treatments is anticipated to sustain current pricing levels. Potential for minor price erosion of 2-5% per year due to increased manufacturing efficiencies and competition may occur.
- Long-Term (5+ years): Long-term price projections are more speculative and would be significantly impacted by breakthroughs in infertility treatment or major shifts in healthcare policy. However, based on the current market structure, sustained affordability is expected. The potential for price increases would likely be tied to specialized formulations or limited-edition branded generics if any were to emerge, which is currently not a trend.
Table 1: Clomid (Clomiphene Citrate) Price Projections (Average per 50mg Tablet)
| Timeframe | Projected Price Range (USD) | Expected CAGR Impact |
|---|---|---|
| Current | $0.50 - $1.50 | N/A |
| Short-Term (1-3 yrs) | $0.48 - $1.58 | +/- 5% |
| Medium-Term (3-5 yrs) | $0.46 - $1.55 | -2% to -5% |
Note: Prices are estimates and subject to significant variation based on specific markets, distributors, and bulk purchasing agreements.
Therapeutic Alternatives and Market Impact
While Clomid is a widely used first-line treatment, several alternatives exist, each with its own efficacy, side effect profile, and cost:
- Letrozole: A non-steroidal aromatase inhibitor, often used off-label for ovulation induction. It is generally considered to have comparable or superior efficacy to clomiphene citrate in some patient populations and may have a lower risk of multiple gestations. Its price is also competitive with generic clomiphene.
- Gonadotropins: Injectable hormones (e.g., follicle-stimulating hormone - FSH, luteinizing hormone - LH) used for ovulation induction when oral agents are unsuccessful. These are significantly more expensive than Clomid and are typically administered under closer medical supervision.
- Metformin: An insulin-sensitizing agent used for women with polycystic ovary syndrome (PCOS) and ovulatory dysfunction. It may be used alone or in combination with Clomid.
- Assisted Reproductive Technologies (ART): IVF, intracytoplasmic sperm injection (ICSI), and gamete intrafallopian transfer (GIFT) are more intensive and costly procedures.
The market position of Clomid remains strong due to its established efficacy, oral administration, and cost-effectiveness, particularly in low-resource settings and as an initial treatment approach. However, ongoing research into the comparative effectiveness of letrozole and the continued advancement of ART may influence prescribing patterns and, consequently, Clomid's market share over the long term.
Key Takeaways
- The global market for Clomid is intrinsically linked to the broader infertility treatment sector, which is experiencing robust growth driven by rising infertility rates and increased awareness.
- The patent for clomiphene citrate has expired, leading to a market dominated by generic competition. This has resulted in generally low and stable pricing for the standard oral formulation.
- Price projections indicate continued stability with potential for slight erosion due to manufacturing efficiencies, rather than significant increases.
- While Clomid remains a cost-effective first-line treatment, therapeutic alternatives like letrozole and more advanced ART procedures represent competitive pressures that could influence its market share over time.
Frequently Asked Questions
-
What is the primary indication for Clomid? Clomid is indicated for the treatment of ovulatory dysfunction in women desiring pregnancy and has demonstrated effectiveness in patients with conditions such as the polycystic ovary syndrome (PCOS).
-
What is the typical cost range for a prescription of Clomid? A prescription for one cycle of Clomid (typically 15 tablets) can range from $5 to $25 at the retail level for generic formulations, depending on the pharmacy and insurance.
-
Are there significant patent protections remaining for Clomid? No, the original patent for clomiphene citrate expired in 1977. The market is primarily served by generic manufacturers.
-
How does the price of Clomid compare to alternative ovulation induction medications like letrozole? Generic formulations of Clomid and letrozole are generally comparable in price, both offering cost-effective options for ovulation induction. Injectable gonadotropins are substantially more expensive.
-
What are the major factors that could influence future Clomid prices? Future pricing will be influenced by raw material costs, manufacturing efficiency of generic producers, the intensity of market competition, and potential shifts in treatment guidelines or regulatory requirements.
Citations
- World Health Organization. (n.d.). Infertility. Retrieved from [WHO Website - Placeholder for actual URL if available]
- U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book Website - Placeholder for actual URL if available]
More… ↓
