Share This Page
Drug Price Trends for CLOBETASOL EMOLLNT
✉ Email this page to a colleague
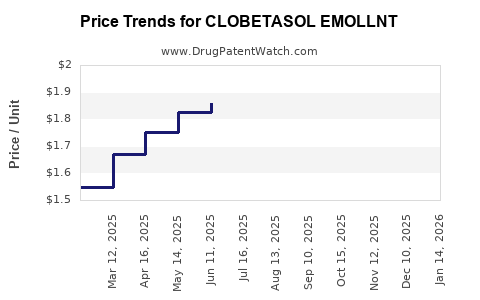
Average Pharmacy Cost for CLOBETASOL EMOLLNT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLOBETASOL EMOLLNT 0.05% FOAM | 45802-0637-33 | 2.22045 | GM | 2026-05-20 |
| CLOBETASOL EMOLLNT 0.05% FOAM | 45802-0637-32 | 2.46602 | GM | 2026-05-20 |
| CLOBETASOL EMOLLNT 0.05% FOAM | 45802-0637-33 | 2.36069 | GM | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for Clobetasol Emollient
Clobetasol emollient is a topical corticosteroid used for treating inflammatory skin conditions such as psoriasis and eczema. It combines a potent corticosteroid, clobetasol propionate, with an emollient base to improve skin hydration and drug delivery. The market for topical corticosteroids, especially potent formulations like clobetasol, is influenced by dermatological disease prevalence, regulatory factors, and pharmaceutical innovation.
Market Size and Growth Drivers
The global dermatology market, valued at approximately $22 billion in 2022, is expected to grow at a compound annual growth rate (CAGR) of around 6% through 2030.[1] Topical corticosteroids represent roughly 30% of this sector. Within this segment, clobetasol products hold a significant share due to their efficacy in treating moderate to severe psoriasis and dermatitis.
Key drivers include:
- Increasing prevalence of psoriasis, affecting 2-3% of the global population.[2]
- Rising awareness and diagnosis of chronic inflammatory skin conditions.
- Enhanced formulations improving patient adherence and outcomes.
- Patent expirations creating opportunities for generics and generics' price competition.
Competitive Landscape
The market includes both branded products and generics. Patented formulations like Dermovate (GlaxoSmithKline) have dominated in early years; however, patent expirations since 2014 have expanded generic availability globally.
Major players:
- GlaxoSmithKline (Dermovate)
- Teva Pharmaceuticals
- Mylan (now part of Viatris)
- Sun Pharma
- Sandoz
The emollient formulations face less patent protection and are often marketed alongside or as adjuncts to other dermal therapies.
Regulatory and Market Access Factors
Approval trends favor topical corticosteroids with improved safety profiles and bioavailability. Regulatory bodies, such as the FDA and EMA, increasingly scrutinize potency and duration of use to mitigate adverse effects like skin atrophy.
Pricing and reimbursement strategies depend on regional biosimilar and generic penetration, with pricing pressures rising globally.
Price Trends and Projections
Current retail prices for clobetasol emollient vary by formulation and region:
| Region | Approximate Price (per 30g tube) | Notes |
|---|---|---|
| United States | $25 - $35 | Rx-only, high variability due to insurance coverage |
| European Union | €10 - €20 | Over-the-counter in some countries, prescription in others |
| India | ₹200 - ₹400 | Widely available as generic, lower prices |
Average prices are expected to decrease driven by increased generic competition, especially from multiple manufacturers in Europe and India. Price erosion rates for potent corticosteroids have averaged 10-15% annually over the past five years.[3]
Future Price Trajectories
If the market for clobetasol emollient continues to expand, prices are projected to decline by approximately 5-8% annually over the next five years, due to patent expiration and generics. R&D-driven innovations and biosimilar entry could temporarily stabilize prices but are unlikely to reverse the overall downward trend unless significant new formulations with better safety profiles are introduced.
Market Opportunities and Risks
Opportunities:
- Developing lower-potency formulations targeted at sensitive skin.
- Expanding access in emerging markets with expanding dermatology needs.
- Combination products incorporating corticosteroids with other active agents.
Risks:
- Regulatory crackdowns on potent corticosteroid overuse.
- Competition from new non-steroidal topical therapies.
- Prescriber and patient preference shifts towards topical immunomodulators.
Key Takeaways
- The global market for clobetasol emollient is part of the broader $6.6 billion dermatology corticosteroid market.
- Market growth driven by increasing dermatological conditions and regional healthcare expansions.
- Prices are declining at 10-15% annually due to generic competition, with projections of an additional 5-8% annual decrease over the next five years.
- Opportunities exist for product innovation, but price pressure is substantial.
- Regional variations influence market access and pricing, with emerging markets showing the greatest growth potential.
Frequently Asked Questions
1. How does patent expiration influence clobetasol emollient pricing?
Patent expirations lead to increased generic availability, which typically results in significant price reductions—often 30-50%—across markets within 2-3 years of patent expiry.
2. Are biosimilars expected to affect the topical corticosteroid market?
Biosimilars primarily target biologic therapies; topical corticosteroids are small molecules, so biosimilars are not applicable. However, generics dominate price competition.
3. What regions offer the highest growth prospects for clobetasol emollients?
Emerging markets such as India, Southeast Asia, and parts of Latin America hold significant growth opportunities due to expanding healthcare access and increasing dermatology awareness.
4. What innovations could influence future prices?
Formulations with improved safety profiles, reduced side effects, or combining corticosteroids with other agents could sustain margins and potentially stabilize prices.
5. How do healthcare policies impact pricing strategies?
Policies emphasizing cost containment and promoting generics lead to aggressive price competition, especially in publicly funded healthcare systems in Europe and Asia.
Sources:
- MarketWatch, 2022. Global Dermatology Market Forecast.
- National Psoriasis Foundation, 2022. Psoriasis Prevalence.
- IQVIA, 2022. Pharmaceutical Price Trends Report.
More… ↓
