Last updated: February 12, 2026
What Is Clobazam and Its Market Overview?
Clobazam is an anti-epileptic drug (AED) classified as a benzodiazepine. It is primarily prescribed for Lennox-Gastaut syndrome and other forms of seizures. It is marketed under brand names such as Onfi (US) and Frisium (Europe), with generic versions available.
The global market for Clobazam was valued at approximately $300 million in 2022. It is utilized mainly in the US, Europe, and parts of Asia where epilepsy prevalence is high. Off-label use includes anxiety and other neurological disorders, although these are less common applications.
Market growth results from increased epilepsy diagnoses, off-label use, and new formulation developments. Generic versions have increased market penetration, reducing overall drug prices and expanding access.
How Is the Market for Clobazam Projected to Evolve?
Market Size and Growth
- 2022: Estimated at $300 million
- 2027: Projected to reach $430 million, compound annual growth rate (CAGR) of approximately 8.3% (Fortune Business Insights, 2022)
Regional Market Shares
| Region |
2022 Market Size |
Projected 2027 Market Size |
Notable Factors |
| United States |
$150 million |
$210 million |
Dominates due to high epilepsy prevalence and favorable reimbursement policies. |
| Europe |
$80 million |
$120 million |
Market growth driven by increasing diagnoses and approvals. |
| Asia-Pacific |
$50 million |
$70 million |
Emerging markets, expanding healthcare infrastructure. |
| Others |
$20 million |
$30 million |
Smaller markets with growth potential. |
Key Drivers
- Rising epilepsy incidence (WHO estimates 50 million affected globally)
- Increased off-label prescriptions for anxiety
- Generic drug availability reducing costs
- Approval of new formulations (e.g., sustained-release tablets)
Challenges
- Competitive landscape with other AEDs (e.g., Valproate, Lamotrigine)
- Regulatory hurdles in certain markets
- Concerns over dependence and side effects common to benzodiazepines
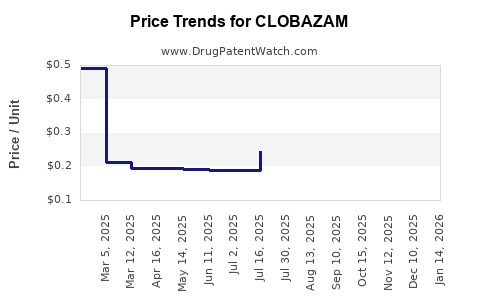
What Are the Price Trends for Clobazam?
Historical Pricing
- Brand-name Clobazam (Onfi): Approximate wholesale price in the US was around $900 per month in 2022.
- Generic versions: Prices decreased to around $500–$700 per month due to market entry.
Projected Price Movements
- With increased generic penetration, prices are expected to decline further, reaching approximately $300–$400 per month by 2027.
- Emerging markets may see lower prices, around $150–$250 per month.
Factors Influencing Price Trends
- Patent expirations lead to increased generics, pressuring prices downward.
- Price negotiations with payers may further reduce costs.
- Development of fixed-dose combinations and formulations may stabilize or alter pricing structures.
What Are the Competitive Dynamics and Patent Status?
Patent Landscape
- Original patent for Onfi expired in the US in 2019.
- Several generic manufacturers have since entered the market.
- Patent protections remain in some jurisdictions for specific formulations or delivery methods.
Key Competitors
- Generic manufacturers securing approval and distribution rights
- Other benzodiazepine AEDs (e.g., clonazepam)
- Newer antiseizure medications with improved safety profiles
Regulatory Environment
- US FDA approval for generics obtained in 2019, increasing supply and lowering prices.
- European Medicines Agency (EMA) approvals align with US regulatory timelines.
What Are the Investment and R&D Opportunities?
- Developing new formulations (e.g., long-acting, liquid forms) could address unmet needs.
- Combination therapies may enhance efficacy or reduce dosages.
- Patent applications related to delivery methods or formulations could extend market exclusivity.
Key Takeaways
- The Clobazam market is expanding at a CAGR of around 8.3% to reach approximately $430 million by 2027.
- The US dominates the market, with increasing penetration in other regions.
- Prices are declining due to generics but may stabilize with new formulation innovations.
- Patent expirations have led to increased competition, lowering prices.
- Opportunities exist in formulation development, combination therapies, and geographic expansion.
FAQs
Q1. When did the patent for Onfi expire, and how has that impacted the market?
A1. The patent for Onfi expired in the US in 2019, leading to increased generic manufacturing, competition, and price reductions.
Q2. Are there any approved new formulations of Clobazam?
A2. Yes, some formulations like extended-release tablets are under development or approval, aiming to improve patient adherence and reduce side effects.
Q3. What are the main side effects associated with Clobazam?
A3. Common side effects include sedation, fatigue, and dizziness. Long-term use risks include dependence and withdrawal issues.
Q4. How does Clobazam compare to other benzodiazepines in terms of efficacy?
A4. Clobazam has a longer half-life and is considered effective for Lennox-Gastaut syndrome, with a favorable safety profile compared to other benzodiazepines.
Q5. Which markets offer the highest growth potential for Clobazam?
A5. Emerging markets in Asia-Pacific and Latin America present significant growth opportunities due to increasing epilepsy diagnosis and expanding healthcare infrastructure.
References
- Fortune Business Insights. (2022). Global Clobazam Market Size, Share & Industry Analysis.
- World Health Organization. (2021). Epilepsy Fact Sheet.
- U.S. Food and Drug Administration. (2019). Onfi patent and approval information.
- IQVIA. (2022). Prescription Trends for AEDs.
- European Medicines Agency. (2022). EMA Approval Data on Benzodiazepine Drugs.