Share This Page
Drug Price Trends for CLINDAMYCIN-BNZ PEROX
✉ Email this page to a colleague
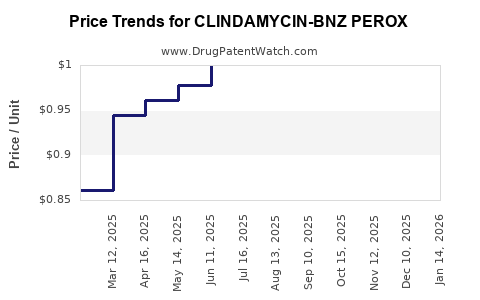
Average Pharmacy Cost for CLINDAMYCIN-BNZ PEROX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLINDAMYCIN-BNZ PEROX 1-5% PMP | 68462-0859-27 | 0.84650 | GM | 2026-03-18 |
| CLINDAMYCIN-BNZ PEROX 1-5% PMP | 45802-0507-04 | 0.84650 | GM | 2026-03-18 |
| CLINDAMYCIN-BNZ PEROX 1-5% PMP | 45802-0510-03 | 1.10139 | GM | 2026-03-18 |
| CLINDAMYCIN-BNZ PEROX 1-5% PMP | 51672-1381-06 | 0.84650 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Clindamycin-Benzoyl Peroxide
Market Overview
Clindamycin-Benzoyl Peroxide (CLINDAMYCIN-BNZ PEROX) combines the antibiotic clindamycin with benzoyl peroxide, primarily targeting moderate to severe acne. The formulation is available topically, prescribed as a treatment for acne vulgaris. It competes within a lucrative dermatological market segment, including topical antibiotics, retinoids, and other combination therapies.
The global acne medication market was valued at approximately $4.7 billion in 2021. It is projected to grow at a CAGR of roughly 4-6% through 2028, driven by increasing acne prevalence, expanding aging populations, and rising awareness of treatments.
Market Dynamics
-
Demand Drivers:
- Rising acne prevalence among adolescents and adults (WHO, 2020).
- Growing prescription rates for combination topical therapies.
- Preference for localized treatments over systemic options.
-
Competitive Landscape:
- Major competitors include oral antibiotics, retinoids, and other topical antibiotics like erythromycin.
- Key brands include Epiduo (adapalene and benzoyl peroxide), Duac (clindamycin and benzoyl peroxide).
- Patent expirations and generic proliferation influence pricing.
-
Regulatory Environment:
- The drug’s approval status varies by region.
- In the U.S., generic forms dominate, influencing price competition.
- Patent protections for brand formulations might affect market exclusivity and pricing.
Current Market Position
Clindamycin-Benzoyl Peroxide products are marketed mainly by generic manufacturers and some brand-name producers like Galderma (e.g., Duac).
Availability as both prescription and over-the-counter (depending on regional regulations) influences market reach.
Price Projections
-
Current Pricing:
- Brand-name formulations range from $100 to $250 per 30-gram tube.
- Generics are priced between $25 and $60 per 30-gram tube, reflecting significant price erosion post-patent expiration.
-
Future Price Trends:
- Patent expirations in major territories likely will accelerate generic price declines.
- Price erosion could see a 50-70% reduction over 3-5 years for generics.
- Brand-name products may maintain a premium of 1.5-2x over generics unless new patents or formulations are introduced.
-
Market Entry & Competition Impact:
- Entry of biosimilars or new combination formulations could suppress prices further.
- Regional variations: U.S. prices are higher than in Europe or emerging markets where local generics are prevalent.
Price Projections Table
| Year | Brand-Name Price (USD) | Generic Price Range (USD) | Notes |
|---|---|---|---|
| 2023 | $150-$250 | $25-$60 | Current market; brand premiums. |
| 2024 | $140-$220 | $20-$55 | Patent expiry effects emerging. |
| 2025 | $130-$200 | $15-$50 | Increased generic market penetration. |
| 2026 | $120-$190 | $12-$45 | Further market saturation. |
| 2027+ | $100-$180 | $10-$40 | Stabilization at new equilibrium levels. |
Implications for Stakeholders
- Manufacturers should anticipate margin compression post-patent expiration.
- R&D investment in novel formulations or combination therapies may sustain higher pricing.
- Exit strategies could include licensing or acquisition of patent rights.
Key Factors Impacting Price Dynamics
- Patent and regulatory exclusivity.
- New formulations or delivery systems.
- Regional regulatory approvals or restrictions.
- Market penetration strategies by competitors.
Key Takeaways
- The global acne treatment market continues growth, bolstered by increased prevalence and research.
- Clindamycin-Benzoyl Peroxide faces pricing pressure from generics, with future prices trending downward.
- Patent law and regional regulations shape market dynamics and pricing.
- Innovative formulations could preserve profit margins amid generic competition.
- Prices in the U.S. typically higher than in Europe and emerging markets.
FAQs
-
When is patent expiration expected for key Clindamycin-Benzoyl Peroxide formulations?
- Typically between 2024 and 2026, depending on jurisdiction and patent status.
-
How does regional regulation affect pricing?
- Stricter control and registration costs in certain regions can increase prices; market access speed influences price competition.
-
What is the impact of biosimilar or generic entry?
- Increased competition leads to significant price erosion, primarily within three years of patent expiry.
-
Are there formulations that command premium pricing?
- Yes, patented combinations, novel delivery systems, or dermatologist-preferred formulations can command higher prices.
-
How are emerging markets affecting global pricing trends?
- Lower costs and high acceptance in developing countries lead to reduced average prices globally.
Sources
[1] Markets and Markets, “Acne Treatment Market,” 2022.
[2] WHO, “Adolescent health & acne prevalence,” 2020.
[3] IQVIA, “Global Topical Acne Products,” 2022.
[4] U.S. Patent and Trademark Office, “Patent expiry schedules,” 2023.
[5] Statista, “Average prices for topical acne drugs,” 2022.
More… ↓
