Share This Page
Drug Price Trends for CLEARLAX POWDER
✉ Email this page to a colleague
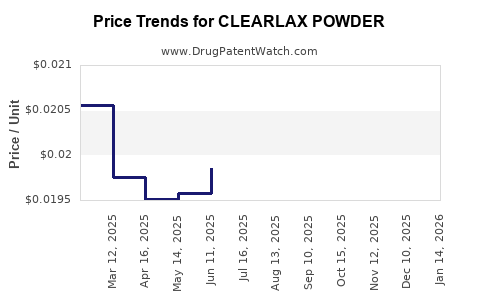
Average Pharmacy Cost for CLEARLAX POWDER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLEARLAX POWDER | 70000-0704-01 | 0.02405 | GM | 2026-03-18 |
| CLEARLAX POWDER | 70000-0415-01 | 0.02959 | GM | 2026-03-18 |
| CLEARLAX POWDER | 70000-0415-03 | 0.02039 | GM | 2026-03-18 |
| CLEARLAX POWDER | 46122-0014-31 | 0.02959 | GM | 2026-03-18 |
| CLEARLAX POWDER | 70000-0415-02 | 0.02405 | GM | 2026-03-18 |
| CLEARLAX POWDER | 46122-0014-33 | 0.02405 | GM | 2026-03-18 |
| CLEARLAX POWDER | 46122-0014-71 | 0.02039 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CLEARLAX POWDER Market Analysis and Financial Projection
What is the Market Size and Demand for CLEARLAX POWDER?
CLEARLAX POWDER is primarily used for the treatment of occasional constipation, mainly in pediatric populations and the elderly. The global laxatives market was valued at approximately USD 5.4 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 3.5% through 2030[1].
In the U.S., the pediatric segment accounts for an estimated 25% of the total laxatives market, with pediatric constipation treatments valued around USD 1.4 billion in 2022. The demand correlates with increased awareness and diagnosis of pediatric constipation, alongside a preference for osmotic laxatives like polyethylene glycol (PEG) formulations, including powders like CLEARLAX.
European markets show similar patterns, with OTC laxatives constituting about 60% of sales and pediatric formulations representing a significant share. Growing awareness about non-prescription options drives market expansion.
How Does CLEARLAX POWDER Compare to Competitors?
CLEARLAX POWDER, which contains polyethylene glycol 3350 (PEG 3350), competes primarily with similar osmotic laxatives approved for over-the-counter use:
-
Miralax (U.S.) / Glycolax (U.K.): The most comparable product, marketed widely for pediatric and adult use. As of 2022, Miralax sales in the U.S. approximate USD 315 million annually[2].
-
Lax-A-Day (Canada): PEG powder aimed at pediatric use, with less market penetration but growing availability.
-
Other OTC powders: Includes products like Fleet Pediatric Enema, although mainly used in clinical settings or for specific populations.
Market share among these products depends on brand recognition, regulatory approvals, and consumer preferences. CLEARLAX's competitive positioning hinges on dosing efficacy, safety profile, taste, and packaging.
What Are the Regulatory Conditions and Patent Landscape?
CLEARLAX is generally marketed as an OTC product with approval from the FDA in the U.S. and regulatory agencies across Europe. PEG-based laxatives typically hold FDA monographs as laxatives, not requiring pre-approval, which can allow rapid market entry.
Patents for PEG formulations expired or are nearing expiration, generally after 2004 for the main PEG APIs. This situation opens pathways for generic competition, affecting pricing strategies[3].
What Are Price Trends and Future Projections?
Current retail prices for OTC PEG powders like CLEARLAX range from USD 8 to USD 15 for a container with approximately 30 doses, or USD 0.25-0.50 per dose.
In a highly competitive environment, price erosion is expected as generics enter the market once patents expire or are invalidated. Historical data indicates a 4-6% annual price decline for PEG laxatives in the U.S. after patent expiry[4].
Forecasting for 2023-2030 suggests that the global OTC laxatives market will experience a gradual decrease in per-unit prices, with a compound annual decline of 2-3% in mature markets like North America and Europe.
Price competition could be intensified by the entry of private-label products and international generic manufacturers, reducing profit margins for brand-name producers.
What Are Strategic Considerations for Pricing and Market Penetration?
Producers of CLEARLAX should consider:
-
Product Differentiation: Offering pediatric-specific formulations with flavoring or dosing devices.
-
Market Expansion: Entering emerging markets where OTC laxatives are less regulated and demand is rising.
-
Partnerships: Collaborating with pharmacies and pediatric clinics to improve brand visibility.
-
Pricing Strategy: Maintaining competitive pricing initially while leveraging brand loyalty to slow erosion post-patent expiry.
Closing Summary: Price and Market Outlook
The global OTC laxatives market exhibits steady growth, driven by increasing prevalence of constipation, especially among children and seniors. Competitive pressures and patent expirations are likely to lead to price declines, with a projected annual decrease of 2-3%. CLEARLAX must adapt to this environment through product innovation, strategic positioning, and cost optimization.
Key Takeaways
- The global laxatives market was valued at USD 5.4 billion in 2022, with a CAGR of 3.5% forecasted through 2030.
- Pediatric demographics underpin a significant share, with OTC PEG powders like CLEARLAX vying against Miralax and generics.
- Current retail prices are USD 8-15 per container; expected to decline by 2-3% annually after patent expirations.
- Competition from generics will heighten, influencing pricing strategies and profit margins.
- Market expansion into emerging economies offers growth opportunities amid evolving regulatory landscapes.
FAQs
1. When will patent expiration likely affect CLEARLAX pricing?
Patents for PEG formulations generally expired around 2004. If CLEARLAX has additional proprietary formulations or packaging patents, these could extend exclusivity until their expiration, commonly 10-15 years after filing.
2. How significant are regulatory barriers for entering new markets?
Most OTC PEG powders require compliance with local OTC monographs or registration procedures. European markets often align with EMA standards, while emerging markets may have less stringent requirements, facilitating easier entry.
3. What are the main factors influencing consumer choice?
Efficacy, safety profile, taste, ease of dosing, and brand reputation are primary drivers. Pediatric formulations with flavoring and dosing aids tend to increase adherence.
4. How do emerging therapies impact the laxatives market?
Novel agents, such as prokinetics or microbiome-targeting drugs, could alter treatment paradigms, potentially reducing reliance on traditional osmotic laxatives over time.
5. What is the outlook for private-label PEG laxatives?
Private-label products account for approximately 50-60% of OTC laxatives in North America and Europe, exerting downward pressure on prices and challenging brand-name market share.
References:
[1] MarketsandMarkets, "Laxatives Market by Type, Application, and Region," 2023.
[2] Statista, "Miralax Annual Sales," 2022.
[3] FDA, "Generic Drug Facts," 2022.
[4] IQVIA, "Pharmaceutical Price Trends," 2022.
More… ↓
