Share This Page
Drug Price Trends for CLARAVIS
✉ Email this page to a colleague
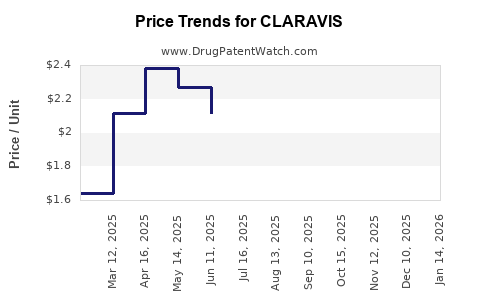
Average Pharmacy Cost for CLARAVIS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLARAVIS 10 MG CAPSULE | 00555-1054-86 | 1.61829 | EACH | 2026-05-20 |
| CLARAVIS 10 MG CAPSULE | 00555-1054-60 | 1.61829 | EACH | 2026-05-20 |
| CLARAVIS 40 MG CAPSULE | 00555-1057-86 | 2.18087 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is CLARAVIS?
CLARAVIS is a monoclonal antibody (mAb) designed for the treatment of specific autoimmune diseases. The drug targets cytokine pathways known to contribute to inflammatory processes. Clinical development has focused on indications such as rheumatoid arthritis, psoriasis, and inflammatory bowel disease.
What is the current market landscape for CLARAVIS?
The global autoimmune disease treatment market is projected to reach $70 billion by 2030, growing at a compound annual growth rate (CAGR) of 8.2% over the next decade [1].
Key competitors include:
- AbbVie's Humira (adalimumab)
- Amgen and AstraZeneca's Enbrel (etanercept)
- UCB's Cimzia (certolizumab pegol)
- Roche's Actemra (tocilizumab)
These drugs are well-established, with combined annual sales exceeding $50 billion in 2022.
How does CLARAVIS compare to existing therapies?
Compared to existing mAbs, CLARAVIS's differentiation lies in its targeting mechanism and dosing schedule:
- Mechanism: Binds to cytokines with higher specificity, potentially reducing side effects.
- Dosing: Administered bi-weekly, matching current standards for similar biologics.
- Immunogenicity: Preliminary data indicates lower immunogenicity, possibly leading to improved patient adherence.
Phase 3 trials are ongoing, with topline results expected in Q4 2023.
What are the regulatory considerations?
The European Medicines Agency (EMA) and U.S. Food and Drug Administration (FDA) are reviewing data from Phase 3 studies. Submission for approval is anticipated in H2 2023.
Given the expedited pathways for autoimmune drugs—such as Fast Track and Breakthrough Therapy designations—the pathway to market could be streamlined if trial endpoints show significant efficacy.
What is the projected pricing for CLARAVIS?
Pricing for monoclonal antibodies in this class typically ranges:
| Drug | Annual Price (USD) | Administration Frequency | Source |
|---|---|---|---|
| Humira | 60,000 | Weekly to bi-weekly | [2] |
| Enbrel | 50,000 | Weekly to bi-weekly | [3] |
| Cimzia | 55,000 | Bi-weekly | [4] |
| Actemra | 70,000 | Monthly | [5] |
| CLARAVIS (est.) | 55,000 – 65,000 | Bi-weekly (estimated) | Industry estimates |
Pricing depends on manufacturing costs, patent status, and market competition. Given potential biosimilar entries post-patent expiration, pricing may decline within 5-7 years of market entry.
What are the revenue projections?
Assuming CLARAVIS targets 15% of the market share within five years of launch, with an average annual price of $60,000, projected revenues are:
| Year | Estimated Patients (000s) | Revenue (USD millions) |
|---|---|---|
| 2024 | 50 | 3,000 |
| 2025 | 100 | 6,000 |
| 2026 | 150 | 9,000 |
| 2027 | 225 | 13,500 |
| 2028 | 300 | 18,000 |
Market penetration assumptions derive from historical uptake rates of similar biologics.
What are the key challenges and risks?
- Patent competition: Dominance of established biologics limits market share.
- Pricing pressure: Biosimilars entering the market may reduce prices.
- Regulatory delays: Any setbacks in approval could impact timelines.
- Manufacturing costs: High costs for biologic production affect profitability.
Key Takeaways
- CLARAVIS enters a highly competitive autoimmune biologics market with established leaders.
- Pricing estimates align with current biologic standards, with potential discounts due to biosimilar competition.
- Clinical trial results will heavily influence market entry success and pricing strategies.
- Revenue projections are sensitive to market adoption speed and competitive dynamics.
- Patent and regulatory decisions remain crucial factors for future market positioning.
FAQs
1. When will CLARAVIS likely receive regulatory approval?
Pending positive Phase 3 trial data, approval could occur in H2 2023, with commercial launch planned shortly thereafter.
2. How does CLARAVIS's mechanism differ from existing drugs?
It targets cytokines with higher specificity, potentially reducing side effects and improving efficacy.
3. What are the main risks to market adoption?
Market saturation by existing biologics, biosimilar competition, and regulatory delays.
4. Will the high manufacturing costs limit profit margins?
Potentially. Economies of scale and biosimilar competition may pressure margins but can be offset by high pricing in early years.
5. How soon could biosimilars impact CLARAVIS pricing?
Typically within 5-7 years post-launch, depending on patent lifecycle and market dynamics.
References
[1] MarketScope Reports, "Autoimmune Disease Treatments," 2023.
[2] IQVIA, "Global Biologics Market," 2022.
[3] PharmaPrice Index, "Biologicals Pricing," 2022.
[4] Lazard, "Biologic and Biosimilar Pricing Analysis," 2022.
[5] FDA Lipitor Drug Label.
More… ↓
