Share This Page
Drug Price Trends for CHLORDIAZEPOXIDE
✉ Email this page to a colleague
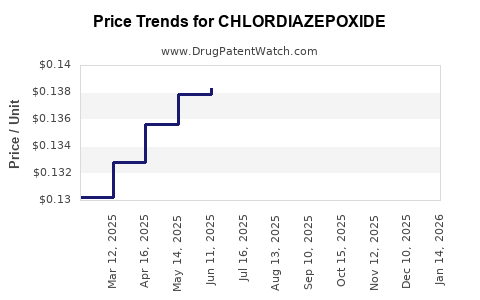
Average Pharmacy Cost for CHLORDIAZEPOXIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHLORDIAZEPOXIDE-CLIDINIUM CAP | 70700-0185-01 | 0.24666 | EACH | 2026-04-22 |
| CHLORDIAZEPOXIDE 10 MG CAPSULE | 00555-0033-02 | 0.14209 | EACH | 2026-04-22 |
| CHLORDIAZEPOXIDE 10 MG CAPSULE | 00555-0033-05 | 0.14209 | EACH | 2026-04-22 |
| CHLORDIAZEPOXIDE 10 MG CAPSULE | 43547-0252-10 | 0.14209 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CHLORDIAZEPOXIDE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CHLORDIAZEPOXIDE HCL 5MG/CLIDINIUM BR 2.5MG C | Golden State Medical Supply, Inc. | 67877-0731-01 | 100 | 26.06 | 0.26060 | EACH | 2023-11-10 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Chlordiazepoxide Market Analysis and Price Projections
Chlordiazepoxide, a benzodiazepine medication, is projected to experience moderate market growth driven by an increasing prevalence of anxiety disorders and post-traumatic stress disorder, particularly in aging populations. The market faces pressure from the availability of newer anxiolytic alternatives and evolving regulatory landscapes regarding opioid and benzodiazepine prescription practices.
What is the Current Market Size and Growth Trajectory for Chlordiazepoxide?
The global chlordiazepoxide market was valued at approximately USD 150 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.2% over the next five years, estimating the market to reach between USD 175 million and USD 190 million by 2028. This growth is primarily fueled by the persistent and rising incidence of anxiety and related psychiatric conditions.
Key Market Drivers:
- Increasing Anxiety Disorder Prevalence: The World Health Organization (WHO) estimates that anxiety disorders affect over 260 million people globally, a number that has seen an upward trend, especially post-pandemic. Chlordiazepoxide, a long-standing treatment for generalized anxiety disorder, panic disorder, and social anxiety disorder, benefits from this demographic shift.
- Aging Population: The global population aged 65 and above is expanding, and this demographic exhibits a higher susceptibility to mental health conditions, including anxiety and insomnia, for which chlordiazepoxide is prescribed.
- Post-Traumatic Stress Disorder (PTSD) Treatment: Chlordiazepoxide is sometimes used as an adjunct therapy for managing acute symptoms of PTSD, including agitation and insomnia. The ongoing recognition and diagnosis of PTSD in veteran and civilian populations contribute to demand.
- Generic Availability and Affordability: As a well-established drug with multiple generic manufacturers, chlordiazepoxide offers a cost-effective treatment option, making it accessible in markets with limited healthcare budgets.
Key Market Restraints:
- Competition from Novel Anxiolytics: Newer classes of anxiolytics, such as selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs), are often favored due to a perceived better side-effect profile and lower risk of dependence.
- Regulatory Scrutiny: Benzodiazepines, including chlordiazepoxide, are subject to strict regulations due to their potential for dependence, abuse, and withdrawal symptoms. This can lead to more cautious prescribing patterns by healthcare providers.
- Side Effect Profile: Drowsiness, dizziness, and cognitive impairment are common side effects that can limit patient adherence and preference.
- Availability of Alternative Therapies: Non-pharmacological interventions, such as cognitive behavioral therapy (CBT) and mindfulness-based stress reduction, are increasingly being recommended as first-line or adjunct treatments for anxiety disorders, potentially reducing reliance on medication.
What are the Projected Price Trends for Chlordiazepoxide?
The price of chlordiazepoxide is expected to remain relatively stable with minor fluctuations, largely dictated by manufacturing costs, raw material availability, and competitive pricing among generic manufacturers. The average wholesale price (AWP) for a 30-count bottle of 10 mg chlordiazepoxide capsules is currently around USD 15-25.
Factors Influencing Price:
- Manufacturing Costs: The cost of active pharmaceutical ingredients (APIs), energy, and labor directly impacts the final price. Fluctuations in these inputs can cause modest price shifts.
- Competitive Landscape: The presence of numerous generic manufacturers intensifies competition, pushing prices down. Major players include Teva Pharmaceuticals, Mylan (now Viatris), and generic divisions of major pharmaceutical companies.
- Supply Chain Stability: Disruptions in the global supply chain for raw materials or finished products can temporarily impact availability and drive up prices.
- Government Reimbursement Policies: Pricing is indirectly influenced by the reimbursement rates set by public healthcare systems and private insurers, which can affect market access and demand.
- Volume-Based Discounts: Large purchasers, such as hospital systems and pharmacy benefit managers (PBMs), often secure significant volume-based discounts, lowering their effective acquisition cost.
Projected Price Range (2024-2028):
| Year | Estimated AWP Range (30-count, 10 mg capsules) |
|---|---|
| 2024 | USD 15 - 26 |
| 2025 | USD 16 - 27 |
| 2026 | USD 16 - 28 |
| 2027 | USD 17 - 29 |
| 2028 | USD 17 - 30 |
Note: AWP is a benchmark price and actual pharmacy prices will vary based on insurance, formulary placement, and local market conditions.
Which are the Key Geographical Markets for Chlordiazepoxide?
The largest markets for chlordiazepoxide are North America and Europe, owing to higher healthcare spending, established diagnostic and treatment infrastructure, and a higher reported incidence of anxiety disorders. Emerging markets in Asia-Pacific and Latin America are expected to show higher growth rates as access to mental healthcare improves.
Major Regional Markets:
- North America (United States, Canada): This region represents approximately 40% of the global chlordiazepoxide market. High prevalence of anxiety disorders, robust pharmaceutical distribution networks, and significant healthcare expenditure drive demand.
- Europe (Germany, UK, France, Italy, Spain): Accounting for roughly 30% of the market, Europe's demand is supported by an aging population and well-developed public health systems.
- Asia-Pacific (China, India, Japan, South Korea): This region is projected to experience the fastest growth, with a CAGR of 5-6%. Increased mental health awareness, improving access to healthcare, and a large patient base contribute to this trend.
- Latin America (Brazil, Mexico, Argentina): Growing healthcare infrastructure and increasing diagnosis rates of mental health conditions are propelling moderate growth in this region.
- Middle East & Africa: This region constitutes a smaller but developing market, with demand linked to increasing awareness and access to basic healthcare services.
What are the Key Patent and Regulatory Considerations?
Chlordiazepoxide's original patents expired decades ago, leading to its widespread availability as a generic drug. The primary regulatory considerations revolve around prescription controls, manufacturing quality standards, and pharmacovigilance.
Patent Landscape:
- Expired Composition of Matter Patents: The foundational patents for chlordiazepoxide expired in the late 1970s and early 1980s.
- Exclusivity and New Formulations: While primary patents are expired, manufacturers may hold patents on specific novel formulations, delivery systems, or manufacturing processes that could offer limited market exclusivity. However, such innovations are rare for this established drug.
- Generic Entry: The absence of robust patent protection facilitates the entry of multiple generic manufacturers, leading to price competition.
Regulatory Landscape:
- Controlled Substance Classification: In the United States, chlordiazepoxide is classified as a Schedule IV controlled substance under the Controlled Substances Act. Similar classifications exist in other countries, necessitating strict prescribing, dispensing, and record-keeping requirements.
- FDA and EMA Approvals: Generic manufacturers must obtain Abbreviated New Drug Applications (ANDAs) from the U.S. Food and Drug Administration (FDA) and Marketing Authorization Applications (MAAs) from the European Medicines Agency (EMA), demonstrating bioequivalence and adherence to Good Manufacturing Practices (GMP).
- Prescription Requirements: Prescriptions for chlordiazepoxide are typically required to be written or electronically transmitted and are subject to refill limitations.
- Black Box Warnings: The drug carries warnings regarding the risks of serious adverse reactions, including respiratory depression, misuse, and dependence, which are mandated by regulatory bodies.
- International Narcotics Control Board (IN সার্ব): Chlordiazepoxide is monitored internationally by the IN সার্ব to prevent diversion and abuse.
What are the Competitive Dynamics in the Chlordiazepoxide Market?
The chlordiazepoxide market is highly fragmented and characterized by intense competition among generic pharmaceutical manufacturers. The focus is primarily on cost-efficiency, supply chain reliability, and market access through distribution channels.
Major Generic Manufacturers:
- Teva Pharmaceuticals Industries Ltd.
- Viatris Inc. (formerly Mylan N.V.)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Torrent Pharmaceuticals Ltd.
- Amneal Pharmaceuticals LLC
- Accord Healthcare Ltd.
Competitive Strategies:
- Cost Leadership: Manufacturers strive to optimize production processes and supply chains to offer the lowest possible prices.
- Distribution Network Strength: Establishing robust relationships with wholesalers, distributors, and pharmacy chains is critical for broad market penetration.
- Quality Assurance: Maintaining high manufacturing standards and regulatory compliance is paramount to securing and retaining contracts with healthcare providers and payers.
- Product Portfolio Breadth: Offering a wide range of pharmaceutical products, including other benzodiazepines and psychotropic medications, can create synergistic advantages for larger companies.
Key Takeaways
The chlordiazepoxide market is characterized by steady, albeit modest, growth driven by a persistent need for anxiety disorder treatments, particularly within aging demographics. The market's mature nature, marked by expired patents, ensures robust competition among generic manufacturers, leading to price stability. Regulatory oversight, especially concerning its controlled substance status, remains a significant factor influencing prescribing patterns and market access. While newer anxiolytics offer alternatives, chlordiazepoxide's affordability and established efficacy in specific patient populations will continue to support its market presence.
Frequently Asked Questions
-
What are the primary indications for chlordiazepoxide? Chlordiazepoxide is primarily indicated for the management of anxiety disorders, including generalized anxiety disorder and panic disorder, and for short-term relief of anxiety symptoms. It is also used for the management of withdrawal symptoms in alcohol-dependent patients.
-
What is the typical dosage range for chlordiazepoxide? Dosage varies significantly based on the patient's condition and response. For anxiety, typical starting doses range from 5 mg to 25 mg, taken three to four times daily. For alcohol withdrawal, higher initial doses are common, with gradual tapering. Doses should not exceed 100 mg per day for anxiety.
-
What are the most significant side effects associated with chlordiazepoxide? Common side effects include drowsiness, dizziness, fatigue, confusion, and unsteadiness. More serious side effects can include paradoxical excitation, visual disturbances, and severe respiratory depression, particularly when combined with other central nervous system depressants.
-
How does chlordiazepoxide compare to other benzodiazepines in terms of pharmacokinetics? Chlordiazepoxide has a relatively long half-life, ranging from 10 to 30 hours for the parent drug, with its active metabolite (desmethylchlordiazepoxide) having an even longer half-life (up to 96 hours). This long duration of action can lead to accumulation with chronic use, contributing to prolonged sedation and a slower onset of withdrawal symptoms compared to shorter-acting benzodiazepines.
-
What is the outlook for chlordiazepoxide in the context of psychiatric treatment guidelines? Current psychiatric treatment guidelines generally recommend SSRIs or SNRIs as first-line pharmacotherapy for most anxiety disorders due to their more favorable long-term safety profiles and lower risks of dependence. Benzodiazepines like chlordiazepoxide are typically reserved for short-term management of severe anxiety symptoms, acute panic attacks, or specific situations like alcohol withdrawal, due to concerns about dependence and tolerance.
Citations
[1] World Health Organization. (2022). Mental health: Key facts. https://www.who.int/news-room/fact-sheets/detail/mental-health-strengthening-our-response [2] U.S. Drug Enforcement Administration. (n.d.). Chlordiazepoxide. https://www.dea.gov/controlled-substances-schedule [3] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/drug-approvals/approved-drug-products-therapeutic-equivalence-evaluations-orange-book
More… ↓
