Share This Page
Drug Price Trends for CHILD PAIN-FEVER
✉ Email this page to a colleague
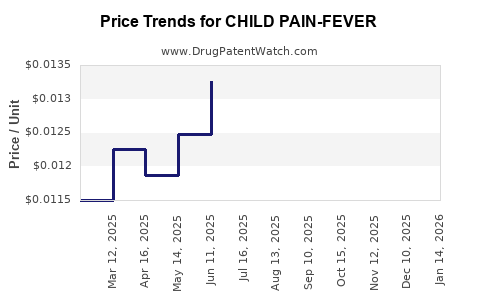
Average Pharmacy Cost for CHILD PAIN-FEVER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHILD PAIN-FEVER 160 MG/5 ML | 46122-0211-26 | 0.02077 | ML | 2026-04-22 |
| CHILD PAIN-FEVER 160 MG/5 ML | 46122-0210-26 | 0.02077 | ML | 2026-04-22 |
| CHILD PAIN-FEVER 160 MG/5 ML | 83720-0500-16 | 0.01284 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for CHILD PAIN-FEVER Drugs
What is the Current Market Landscape for Child Pain-Fever Medications?
The pediatric analgesic and antipyretic market includes products primarily used to treat pain and fever in children. Key categories encompass acetaminophen-based formulations, ibuprofen, and other non-steroidal anti-inflammatory drugs (NSAIDs). The market has experienced steady growth driven by increased awareness of pediatric health, expanding pharmaceutical portfolios, and rising demand for over-the-counter (OTC) remedies.
Market Size and Growth
- Global Pediatric Pain and Fever Market: Valued at approximately USD 2.4 billion in 2022 (Grand View Research).
- Compound Annual Growth Rate (CAGR): Estimated at 4.2% from 2023 to 2030.
- Region Breakdown: North America leads with around 45% of the market, followed by Europe (25%) and Asia-Pacific (20%).
Major Players and Products
| Company | Key Products | Market Share (Estimate) | Distribution Channels |
|---|---|---|---|
| Johnson & Johnson | Tylenol (Acetaminophen) | 35% | OTC, pharmacies, online |
| Pfizer | Advil (Ibuprofen) | 20% | OTC, pharmacies, online |
| Sanofi | Tempo (Paracetamol variants) | 10% | OTC, pharmacies |
| Other Companies | Various brands | 35% | OTC, local markets |
What Are the Regulatory Dynamics Impacting Pricing?
Regulatory authorities impose standards on safety, efficacy, and manufacturing practices for pediatric medications. The US Food and Drug Administration (FDA) and the European Medicines Agency (EMA) require:
- Standardized labeling for pediatric use.
- Evidence of safety in children.
- Good Manufacturing Practices (GMP).
Recent regulatory focus has been on labeling for dosing accuracy and risk warnings, influencing development costs. Regulatory delays can impact product launches and influence price stability.
What Pricing Trends Are Observed?
Current Retail Prices
- Acetaminophen (e.g., Tylenol, OTC): USD 2–5 per 100-count bottle of liquid or chewable tablets.
- Ibuprofen (e.g., Advil, OTC): USD 3–6 per 100-count bottle.
- Prescription Pediatric Pain-Fever Drugs: Range from USD 100–300 per course, depending on formulation, brand, and indication.
Price Determinants
- Brand vs. Generic: Generics sell at 40–60% lower prices than branded products.
- Formulation: Suspension, chewables, and dissolvables command higher prices due to added convenience.
- Regulatory Approvals: New formulations or indications often have higher pricing due to development costs.
Reimbursement and Insurance Impact
Insurance policies often cover OTC analgesics with minimal copayments. Prescription formulations are subject to insurance negotiations, impacting net prices. Price transparency and market consolidation influence overall cost trends.
How Will Product Innovations Influence Market and Pricing?
Development trends include:
- Enhanced formulations: Long-acting or combination products intended to reduce dosing frequency.
- Novel delivery systems: Dissolvable strips, flavor improvements, and low-dose products tailored for different age groups.
- Biological innovations: Not currently prominent for pain/fever but possible future developments.
Innovations tend to command higher prices initially but may shift downward post-generic entry.
Price Projections Through 2028
| Year | Retail Price Range (USD per 100 units) | Expectations |
|---|---|---|
| 2023 | USD 2–6 | Stable with minor variations based on brand and formulation |
| 2025 | USD 2.50–7 | Slight increases due to inflation and innovation costs |
| 2028 | USD 2.45–6.50 | Prices expected to decline with generic market entry |
The overall trend indicates marginal price stabilization or reduction as generics penetrate markets globally.
Key Market Risks and Opportunities
- Risks: Regulatory delays, generic competition, safety concerns impacting formulation approvals.
- Opportunities: Increasing global pediatric population, rising health awareness, potential for new delivery systems, growth in emerging markets.
Key Takeaways
- The pediatric pain/fever drug market was USD 2.4 billion in 2022, with a 4.2% CAGR forecast to 2030.
- Leading products include Tylenol and Advil, with generics capturing significant market share.
- Prices vary by formulation, brand, and region; OTC drugs cost USD 2–6 per 100 units.
- Regulatory standards influence development costs and pricing, especially for new formulations or indications.
- Price projections suggest minor increases through 2025, followed by stabilization or slight decline by 2028 due to generic competition.
FAQs
1. What factors most influence pediatric pain and fever drug prices?
Brand name status, formulation complexity, regulatory approval costs, and market competition primarily influence prices.
2. How does the entry of generics affect pricing trends?
Generics typically reduce overall market prices by 40–60%, leading to increased affordability and market share shifts.
3. Are pediatric-specific formulations more expensive than adult versions?
Yes, due to additional flavoring, dosing accuracy, and safety features, pediatric formulations often cost more per dose.
4. What role do regulatory agencies play?
They establish safety and efficacy standards that can extend to development costs, influence approval timelines, and impact pricing strategies.
5. How might innovations impact future prices?
New delivery systems and formulations can command premium prices initially, but widespread adoption and generic competition tend to reduce costs over time.
References
- Grand View Research. (2023). Pediatric pain management market size, share & trends analysis report.
- U.S. Food and Drug Administration. (2022). Pediatric drug safety guidelines.
- European Medicines Agency. (2022). Standard requirements for pediatric medicines.
More… ↓
