Last updated: February 23, 2026
What is the Current Market for Child Loratadine?
Child loratadine, an antihistamine used to treat allergic rhinitis and urticaria in children, is a generic allergy medication with broad OTC and prescription availability globally.
Market Size and Growth
- The global allergy medications market was valued at approximately USD 20 billion in 2022.
- It is projected to grow at a compound annual growth rate (CAGR) of 6.2% from 2023 through 2030.[1]
- Child-specific allergy treatments represent about 15% of this market, estimated at USD 3 billion in 2022.
Key Players
- Mylan (now part of Viatris)
- Teva Pharmaceuticals
- Sandoz (Novartis)
- Sun Pharma
- Local generic manufacturers in major markets (US, Europe, India, China)
Regulatory Landscape
- In the U.S., Loratadine is approved for OTC use in children aged 2 and above.
- EMA approves loratadine for children aged 2–12 with variations by region.
- Patent expiration for key brand "Clarityne" occurred in the early 2010s, leading to widespread generic entry.
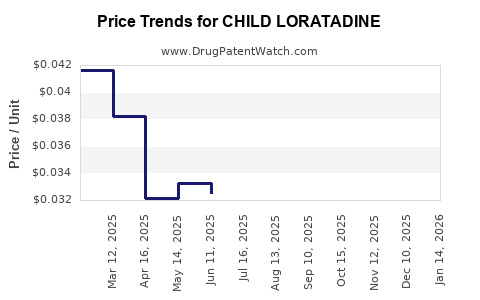
Price Trends and Market Penetration
Current Price Range
| Product Type |
Price per 10 mg tablet (USD) |
Notes |
| Brand (Clarityne, Claritin) |
USD 0.50–1.00 |
Available OTC, higher cost, brand premiums apply |
| Generic Loratadine (10 mg) |
USD 0.05–0.20 |
Widely available, low-cost options |
| Child formulations (syrups) |
USD 3–8 per 120 mL bottle |
Slightly more expensive due to formulation |
Price Dynamics
- Brand-name loratadine products have seen their prices decline between 2010 and 2022 due to patent expirations and increased generic competition.
- Discount retail chains and online pharmacies offer lower-cost options, contributing to the price decrease.
- Regional variation affects pricing: USA and Europe tend to have higher prices for brand-name drugs, whereas emerging markets maintain lower price points.
Future Price Projections
Factors Affecting Prices
- Patent status: No active patents for loratadine in major jurisdictions, fostering intense generic competition.
- Production costs: Slight reduction with increased manufacturing efficiencies and economies of scale.
- Regulatory policies: Potential price regulation in some markets to control healthcare costs.
- Market penetration: Expansion in emerging economies could lower prices owing to higher volume sales.
Price Forecast (2023–2030)
| Year |
Expected Price Range (USD) per 10 mg tablet |
Comments |
| 2023 |
USD 0.05–0.12 |
Market equilibrium with high generic competition |
| 2025 |
USD 0.04–0.10 |
Further integration of low-cost manufacturing and online distribution |
| 2030 |
USD 0.03–0.08 |
Potential stabilization at lower price points due to mature generics |
Child-specific formulations (syrups)
- Expected to follow tablet price trends, though slightly higher due to formulation costs.
- In 2023, USD 3–8 per 120 mL bottle; projected to decline to USD 2.50–6 by 2030.
Market Drivers and Risks
- Increase in allergic conditions among children drives demand.
- Rising consumer preference for OTC medications lowers barriers for purchase.
- Government policies in healthcare cost management may influence pricing.
- Emerging challenges from alternative therapies pose risks to market expansion.
Summary of Key Market and Price Trends
- The market for child loratadine remains competitive, dominated by generics.
- Prices are declining steadily, with projected stabilization at low levels.
- Expansion in emerging markets and OTC sales channels will continue to influence price dynamics.
Key Takeaways
- The global allergy medication market is robust, with child loratadine occupying a significant share.
- Patent expirations and high generic competition have driven prices down in recent years.
- Future price stabilization at USD 0.03–0.08 per 10 mg tablet is probable by 2030.
- Child-specific formulations will follow overall market trends, remaining affordable.
FAQs
1. What are the main factors influencing loratadine prices in different regions?
Patent status, regulatory environment, healthcare policies, manufacturing costs, and regional demand directly influence regional pricing.
2. How does the availability of generics impact market prices?
Increased generic entry significantly reduces costs, resulting in price declines of up to 80% over a decade.
3. Are there differences in child-specific loratadine formulations compared to adult versions?
Yes. Child formulations typically come as syrups or dissolvable tablets, often at slightly higher costs due to formulation requirements.
4. What is the outlook for new formulations or delivery methods for child loratadine?
Limited innovation is expected; focus remains on maintaining low-cost, stable formulations for widespread access.
5. How might regulatory changes affect future prices?
Policies controlling drug pricing or encouraging competition could further reduce costs or stabilize prices across markets.
References
[1] MarketResearch.com. (2023). Global allergy medications market analysis. Retrieved from https://www.marketresearch.com