Share This Page
Drug Price Trends for CHILD CETIRIZINE
✉ Email this page to a colleague
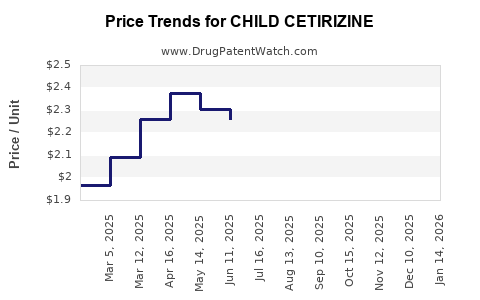
Average Pharmacy Cost for CHILD CETIRIZINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHILD CETIRIZINE HCL 1 MG/ML | 00904-6765-20 | 0.03744 | ML | 2026-03-18 |
| CHILD CETIRIZINE HCL 1 MG/ML | 70752-0104-06 | 0.03744 | ML | 2026-03-18 |
| CHILD CETIRIZINE HCL 1 MG/ML | 00904-6765-20 | 0.03680 | ML | 2026-02-18 |
| CHILD CETIRIZINE HCL 1 MG/ML | 70752-0104-06 | 0.03680 | ML | 2026-02-18 |
| CHILD CETIRIZINE 10 MG CHEW TB | 47335-0344-83 | 2.21882 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CHILD CETIRIZINE Market Analysis and Financial Projection
Market Analysis and Price Projections for Child Cetirizine
Market Overview
Child cetirizine, an antihistamine primarily marketed for pediatric allergic rhinitis and urticaria, is a subset of the broader antihistamine market. As of 2022, the global antihistamine market was valued at approximately USD 2.7 billion, with pediatric formulations accounting for about 25%—around USD 675 million. Child-specific cetirizine formulations include tablets, syrups, and oral solutions, often marketed under brand names such as Zyrtec and others.
Key Market Drivers
- Rising Allergy Prevalence: Increasing incidences of allergic rhinitis and atopic dermatitis among children.
- Growth in Pediatric Healthcare Spending: Global pediatric healthcare expenditure is projected to grow at a CAGR of 6-7% through 2028.
- Preference for Non-Sedating Antihistamines: Cetirizine's non-sedating profile makes it a preferred choice among parents and physicians.
Competitive Landscape
Major manufacturers include Johnson & Johnson (Zyrtec), Sanofi (Allerfree), and Teva Pharmaceuticals, with significant market shares in developed regions. Generic brands also hold approximately 60% of the market, mainly in emerging markets due to cost considerations.
Regulatory Environment
- United States: Approved by FDA as over-the-counter (OTC) for children over 2 years.
- European Union: CE-marked formulations with marketing authorizations in all member states.
- Emerging markets: Regulatory pathways vary, with some countries requiring local clinical data.
Price Trends and Projections
Historical Pricing Data (USD per unit/package)
| Year | Brand Name | Formulation | Approximate Retail Price | Notes |
|---|---|---|---|---|
| 2018 | Zyrtec Child Syrup | 60 mL | 8–10 | Premium brand, widely available |
| 2018 | Generic Cetirizine Syrup | 60 mL | 2–4 | Price substantially lower |
| 2020 | Zyrtec Child Syrup | 60 mL | 7–9 | Slight price decline due to competition |
| 2020 | Generics | 60 mL | 1.5–3 | Consistent market share gains |
| 2022 | Zyrtec Child Syrup | 60 mL | 7–8 | Price stabilization |
Projected Price Trends (2023–2027)
- Brand-name formulations are expected to stabilize around USD 7–8 for 60 mL syrups.
- Generics and store brands could decline to USD 1–2 for similar volumes due to increased competition, especially in emerging markets.
- Pricing elasticity remains high; small increases are possible in regions with limited regulation or supply chain disruptions.
Factors Influencing Future Prices
- Regulatory Changes: Stricter quality standards or patent expirations can affect prices.
- Market Penetration: Greater adoption in emerging markets due to lower costs may depress prices.
- Manufacturing Advancements: Cost reductions through new production techniques could lower consumer prices.
- Supply Chain Dynamics: Disruptions can temporarily inflate prices, but long-term effects depend on market resilience.
Price Projections Summary
| Year | Child Cetirizine Syrup (USD per 60 mL) | Notes |
|---|---|---|
| 2023 | 7–8 | Stable with minor fluctuations |
| 2024 | 6.5–8 | Expect slight decrease due to generics |
| 2025 | 6–7 | Further price stabilization |
| 2026 | 5.5–7 | Market consolidation, emerging markets expand |
| 2027 | 5–6.5 | Possible further decrease, commoditization |
Market Outlook
The pediatric antihistamine segment, including child cetirizine, will likely experience moderate growth driven by rising allergy prevalence, especially in densely populated and urban regions. Price competition will intensify, with generics capturing a larger market share, contributing to declining prices in the medium term.
Key Takeaways
- Child cetirizine's market is influenced by allergy prevalence, pediatric healthcare spending, and regulatory pathways.
- Market leaders maintain premium pricing, but generics dominate overall volume due to lower consumer costs.
- Prices are projected to decline gradually through 2027, especially in emerging markets.
- Regulatory and supply chain factors will create short-term price volatility but are unlikely to alter long-term trends significantly.
FAQs
1. How does patent expiration impact child cetirizine prices?
Patent expirations typically lead to increased generic entry, driving prices down. Current patents for main formulations like Zyrtec expired in various jurisdictions around 2018–2020, facilitating generics.
2. Are there significant regional differences in pricing?
Yes. Prices tend to be higher in developed countries due to regulatory standards and brand loyalty. Emerging markets see lower prices, largely due to higher generic penetration and lower healthcare expenditure.
3. What role do OTC approvals play in pricing?
OTC status tends to increase competition and lower prices, as access becomes easier. In the US, OTC Cetirizine is widely available, contributing to competitive pricing.
4. How will new formulations or delivery mechanisms affect pricing?
Innovations such as liquids or fast-dissolving tablets might command premium prices initially. However, standard formulations will primarily base prices on competition and manufacturing costs.
5. Will supply chain disruptions significantly affect prices?
Short-term disruptions can cause temporary price hikes, but in the long term, market forces and alternative sourcing generally mitigate sustained price increases.
Sources:
- IBISWorld. "Antihistamines market report." 2022.
- EvaluatePharma. "Global Allergic Rhinitis and Rhinitis Treatment Market Analysis." 2022.
- U.S. Food and Drug Administration. "Cetirizine OTC Approval Historical Data." 2020.
- European Medicines Agency. "Market authorizations for cetirizine products." 2022.
- MarketWatch. "Generic Drugs' Market Share Trends." 2022.
More… ↓
