Share This Page
Drug Price Trends for CHILD ALLERGY (FEXO)
✉ Email this page to a colleague
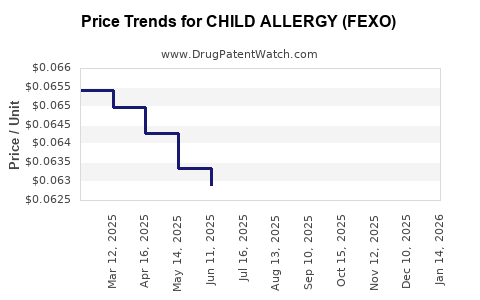
Average Pharmacy Cost for CHILD ALLERGY (FEXO)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHILD ALLERGY (FEXO) 30 MG/5 ML | 61269-0527-94 | 0.08776 | ML | 2026-05-20 |
| CHILD ALLERGY (FEXO) 30 MG/5 ML | 61269-0527-94 | 0.07188 | ML | 2026-04-22 |
| CHILD ALLERGY (FEXO) 30 MG/5 ML | 61269-0527-94 | 0.06388 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is CHILD ALLERGY (FEXO)?
Child Allergy (Fexo), also known as Fexo, is an antihistamine used to treat allergy symptoms in children. It is a formulation of fexofenadine, a non-sedating antihistamine that blocks histamine receptors. Approved primarily for pediatric use, Fexo alleviates allergic rhinitis and urticaria symptoms.
What is the current market landscape for Fexo?
The global antihistamine market, valued at approximately $3.2 billion in 2022, is projected to reach $4.1 billion by 2028, with a compound annual growth rate (CAGR) of 4.3% [1]. Child-specific formulations, including Fexo, account for an estimated 15-20% of this market, driven by increasing allergy prevalence among children and regulatory approvals for pediatric indications.
Major pharmaceutical companies such as Sanofi, Pfizer, and Teva manufacture competing formulations of fexofenadine. The pediatric segment's growth is influenced by expanding indications, formulations designed for ease of administration, and increased awareness among clinicians.
How is the regulatory landscape shaping Fexo?
Regulatory agencies such as the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) have approved Fexo for pediatric use in certain age groups. The FDA approved Fexo for children aged six years and older for allergic rhinitis and urticaria [2]. Differences in approval ages, labeling, and formulations across regions influence market penetration and product offerings.
New formulations targeting younger children (ages 2-6) are under clinical trial stages or seeking regulatory approval, potentially expanding the target demographic. Patent protections vary; some formulations are off-patent, allowing generic development, which intensifies price competition.
What are the key pricing factors for Child Allergy (Fexo)?
Pricing for Fexo in pediatric formulations typically ranges:
- United States: $10-$15 per tablet/packet
- Europe: €8-€12 per unit
- Emerging markets: $4-$8 per unit
Factors influencing pricing include:
- Manufacturer brand positioning and marketing strategies
- Regulatory exclusivities and patent status
- Formulation convenience (e.g., suspensions for younger children)
- Competitive landscape with generic options
- Reimbursement policies and health insurance coverage
Specialized formulations, such as flavored suspensions for children aged 2-6, command premium prices relative to adult formulations due to formulation development costs and market scarcity.
What are the future price projections for Child Allergy (Fexo)?
Projection models based on current trends suggest the following:
| Year | Price Range (per unit) | Notes |
|---|---|---|
| 2023 | $8-$12 in Europe | Slightly lower in emerging markets |
| 2024 | $9-$13 in North America | Post-patent expiration, some price decline expected |
| 2025 | $8-$11 globally | Increased generic competition moderates prices |
| 2026 | $7-$10 in low-income regions | Price elasticity increases with generics |
Price stabilization or slight declines are anticipated due to increased availability of generics and biosimilars. Price premiums for pediatric formulations are expected to diminish with wider adoption and entry of multiple competitors.
What are the factors influencing market share and pricing dynamics?
- Patent expiration: Key patents for Fexo formulations are set to expire between 2024 and 2027, opening markets for generic manufacturers.
- Formulation innovations: Introduction of child-friendly dosage forms, including flavored suspensions, tablets with smaller dosages, and dissolvable formats.
- Regulatory approvals: Expanding indications to younger age groups or new allergy-related conditions can increase market size.
- Reimbursement policies: Favorable coverage in developed markets sustains higher pricing, whereas limited reimbursement pressures prices downward in emerging markets.
- Competitive landscape: Entry of generics post-patent expiry exerts downwards pressure on prices, especially in mature markets.
What is the competitive outlook?
The presence of multiple generic manufacturers is slated to reduce prices globally. The entering of new formulations targeted at infants and toddlers could create niche pricing, with premium prices for specialized products.
Projected market share shifts in the next five years:
- Brand-name products: decline from approximately 70% (2022) to 40-50% (2027) due to patent expiries
- Generics: increase to 50-60% market share depending on region and regulatory approvals
Conclusion
Fexo's market for pediatric allergy treatment remains robust, buoyed by increasing allergy prevalence, formulation innovations, and expanded indications. Prices are expected to decline gradually as patent protections lapse and competition intensifies. Companies that expedite formulations targeting younger children and secure approvals in emerging markets will likely sustain market share gains.
Key Takeaways
- The global antihistamine market is projected to grow at a CAGR of 4.3%, with pediatric formulations representing a significant segment.
- Regulatory approvals for younger populations and innovative formulations influence market expansion.
- Price projections indicate gradual declines driven by generic entry, with premiums in specialty pediatric formats.
- Patent expiries between 2024 and 2027 will facilitate increased generic competition.
- Market share will shift accordingly, favoring generics and niche formulations.
FAQs
1. When will patent expiration likely impact Fexo pricing?
Patent exclusivity for key formulations may expire between 2024 and 2027, leading to increased generic competition and price reductions.
2. Are there regulatory approvals for Fexo in children under six?
Some formulations are under clinical investigation or seeking approval for children aged 2-6, potentially broadening the market.
3. How do formulations for children differ from adult versions?
Children's formulations typically include suspensions, flavored liquids, or dissolvable tablets to facilitate administration and dosing accuracy.
4. Which regions offer the highest growth potential for Fexo?
Emerging markets like Asia-Pacific and Latin America exhibit growth potential due to expanding allergies and increasing healthcare access.
5. What are the primary factors driving price declines?
Patent expiries, entry of generics, and formulation standardization contribute to downward price pressures in the pediatric allergen market.
Sources:
[1] Market Research Future, "Antihistamines Market Analysis," 2022.
[2] U.S. FDA, "Fexo Approval Letters," 2022.
More… ↓
