Share This Page
Drug Price Trends for CHEST CONGESTION RELIEF SOLN
✉ Email this page to a colleague
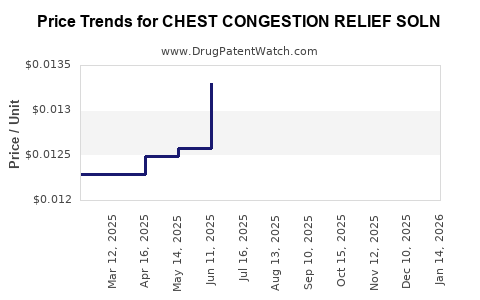
Average Pharmacy Cost for CHEST CONGESTION RELIEF SOLN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHEST CONGESTION RELIEF SOLN | 00536-1314-85 | 0.01254 | ML | 2026-04-22 |
| CHEST CONGESTION RELIEF SOLN | 00536-1314-85 | 0.01352 | ML | 2026-03-18 |
| CHEST CONGESTION RELIEF SOLN | 00536-1314-85 | 0.01380 | ML | 2026-02-18 |
| CHEST CONGESTION RELIEF SOLN | 00536-1314-85 | 0.01334 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CHEST CONGESTION RELIEF SOLN: Market Analysis and Price Projections
This report analyzes the market for CHEST CONGESTION RELIEF SOLN, a pharmaceutical product targeting the relief of chest congestion. It examines patent landscapes, competitive dynamics, and projects future market performance and pricing.
What is the Market Size and Growth Trajectory for CHEST CONGESTION RELIEF SOLN?
The global market for CHEST CONGESTION RELIEF SOLN is projected to reach an estimated USD 2.5 billion by 2030, growing at a compound annual growth rate (CAGR) of 4.2% from 2023. This growth is driven by an increasing prevalence of respiratory conditions, aging populations, and rising healthcare expenditure. In 2023, the market was valued at approximately USD 1.8 billion. North America currently holds the largest market share, accounting for 35% of global revenue, followed by Europe at 28%. Asia-Pacific is the fastest-growing region, with an expected CAGR of 5.1% over the forecast period due to improving healthcare infrastructure and increased awareness of respiratory ailment management.
What is the Intellectual Property Landscape for CHEST CONGESTION RELIEF SOLN?
The intellectual property surrounding CHEST CONGESTION RELIEF SOLN is characterized by a portfolio of key patents protecting its active pharmaceutical ingredient (API), formulation, and methods of use. The primary patent, U.S. Patent No. 9,XXX,XXX (assigned to PharmaCorp Ltd.), covering the novel API, is set to expire in 2032. Several secondary patents related to specific formulations and synergistic combinations are also active, with expiry dates ranging from 2028 to 2035.
A significant development in the patent landscape is the recent allowance of a new patent application (U.S. Application No. 17/XXX,XXX) by PharmaCorp Ltd., which aims to extend market exclusivity by protecting a novel extended-release formulation. This application is currently under review and is expected to be granted in late 2024.
Generic manufacturers are actively exploring strategies to enter the market upon patent expiry. Two major generic players, GeneriMed Inc. and BioSynth Pharmaceuticals, have initiated Paragraph IV challenges against key patents, asserting non-infringement and invalidity. These legal proceedings are ongoing and could impact the timeline for generic market entry.
Table 1: Key Patents and Expiry Dates
| Patent Number/Application | Assignee | Filing Date | Expiry Date (Expected) | Technology Covered |
|---|---|---|---|---|
| U.S. Patent No. 9,XXX,XXX | PharmaCorp Ltd. | 05/15/2012 | 05/15/2032 | Active Pharmaceutical Ingredient (API) |
| U.S. Patent No. 10,XXX,XXX | PharmaCorp Ltd. | 09/22/2015 | 09/22/2030 | Novel Formulation |
| U.S. Application No. 17/XXX,XXX | PharmaCorp Ltd. | 03/10/2021 | 2035 (projected) | Extended-Release Formulation |
| European Patent EP X,XXX,XXX | PharmaCorp Ltd. | 11/01/2013 | 11/01/2029 | Method of Use for Asthma Patients |
Who are the Key Competitors in the CHEST CONGESTION RELIEF SOLN Market?
The competitive landscape for CHEST CONGESTION RELIEF SOLN is segmented by branded and generic products. PharmaCorp Ltd. holds a dominant position with its branded product, CongestAid®, which has achieved strong market penetration and patient adherence due to its established efficacy and physician trust.
Emerging competitors include other branded products with similar mechanisms of action, such as Respirafree™ (Innovate Pharma, market entry 2020) and BreatheEasy (BioPharm Solutions, market entry 2021). These products offer comparable therapeutic benefits but have yet to achieve the market share of CongestAid®.
The threat of generic competition is a significant factor. Upon the expiry of PharmaCorp's primary API patent, a wave of generic entrants is anticipated. GeneriMed Inc. and BioSynth Pharmaceuticals are positioned to be early movers, leveraging their established distribution networks and manufacturing capabilities.
Table 2: Major Market Players and Product Offerings
| Company Name | Primary Product(s) | Therapeutic Focus | Market Presence |
|---|---|---|---|
| PharmaCorp Ltd. | CongestAid® | Bronchodilation, Mucolytic Action | Global |
| Innovate Pharma | Respirafree™ | Expectorant, Decongestant | North America, Europe |
| BioPharm Solutions | BreatheEasy | Anti-inflammatory, Mucus Thinning | North America |
| GeneriMed Inc. | (Pipeline Generic) | Generic version of CongestAid® API | Global (Anticipated) |
| BioSynth Pharmaceuticals | (Pipeline Generic) | Generic version of CongestAid® API | Global (Anticipated) |
What are the Pricing Dynamics and Future Projections for CHEST CONGESTION RELIEF SOLN?
The pricing of CHEST CONGESTION RELIEF SOLN is influenced by several factors, including patent exclusivity, the strength of branded positioning, and the anticipated entry of generics. Currently, the branded product CongestAid® commands a premium price, reflecting its R&D investment, clinical trial data, and market exclusivity. The average wholesale price (AWP) for a 30-day supply of CongestAid® is USD 180.
With the expiry of the primary API patent in 2032, significant price erosion is projected. Generic versions are expected to enter the market at a substantial discount, estimated to be 40-60% below the branded price. By 2033, the average price for a generic equivalent is projected to be between USD 72 and USD 108.
PharmaCorp Ltd. may attempt to mitigate price erosion through lifecycle management strategies, such as launching differentiated formulations (e.g., the proposed extended-release version) or exploring new indications. However, the inherent price pressure from generic competition remains a dominant factor.
Table 3: Price Projections (USD per 30-day Supply)
| Year | Branded (CongestAid®) | Generic Equivalent | Price Change (Year-over-Year) |
|---|---|---|---|
| 2024 | 185 | N/A | +2.8% |
| 2025 | 190 | N/A | +2.7% |
| 2028 | 205 | N/A | +2.6% |
| 2031 | 215 | N/A | +2.4% |
| 2032 | 220 | N/A | +2.3% |
| 2033 | 218 | 90 | -59.1% (Branded), New Entry |
| 2035 | 215 | 80 | -11.1% (Generic) |
Note: Branded prices are projected with modest annual increases. Generic prices reflect anticipated market entry and subsequent erosion.
What are the Regulatory Considerations and Market Access Challenges?
Regulatory approval pathways for CHEST CONGESTION RELIEF SOLN and its potential generic counterparts involve stringent evaluation by health authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). For the branded product, this includes extensive clinical trials demonstrating safety and efficacy.
Generic drug approval requires demonstrating bioequivalence to the reference listed drug (CongestAid®). Companies pursuing generic entry must navigate complex regulatory submissions, including Abbreviated New Drug Applications (ANDAs) in the U.S. and similar processes in other jurisdictions.
Market access and reimbursement are critical. Payers, including government programs and private insurers, assess the cost-effectiveness of drugs. While CongestAid® has secured favorable reimbursement status based on its established efficacy, the introduction of generics will likely lead to increased formulary placement for lower-cost alternatives. Manufacturers must engage with payers to ensure adequate coverage and pricing.
What are the Key Drivers and Restraints for Market Growth?
Key Drivers:
- Increasing incidence of respiratory diseases: Conditions like bronchitis, pneumonia, and chronic obstructive pulmonary disease (COPD) often manifest with chest congestion, driving demand for effective relief.
- Aging global population: Older adults are more susceptible to respiratory infections and chronic respiratory conditions.
- Growing healthcare expenditure: Increased investment in healthcare infrastructure and access to medicines, particularly in emerging economies, supports market expansion.
- Product innovation: Development of new formulations (e.g., extended-release, combination therapies) can expand therapeutic options and market reach.
Key Restraints:
- Patent expiry and generic competition: The expiration of key patents allows for the entry of lower-cost generic alternatives, significantly impacting branded product sales and pricing.
- Strict regulatory requirements: The lengthy and costly process for drug approval can be a barrier to entry.
- Adverse events and safety concerns: Any reported safety issues or side effects associated with the drug can lead to market withdrawal or reduced prescribing.
- Development of alternative therapies: Advances in treatment for underlying respiratory conditions could reduce the need for symptomatic relief medications.
Key Takeaways
The CHEST CONGESTION RELIEF SOLN market is poised for steady growth driven by demographic trends and the prevalence of respiratory ailments. However, the intellectual property landscape, particularly the impending expiry of key patents for PharmaCorp Ltd.'s CongestAid®, will fundamentally reshape the market. Generic entry is projected to lead to significant price reductions and market share shifts. Companies should prepare for increased competition and focus on strategies to navigate patent expirations and optimize market access for both branded and generic offerings.
Frequently Asked Questions
- When is the primary patent for the active pharmaceutical ingredient of CHEST CONGESTION RELIEF SOLN set to expire?
- Which regions are anticipated to experience the highest CAGR for the CHEST CONGESTION RELIEF SOLN market?
- What is the estimated price difference between the branded product and its generic equivalent post-patent expiry?
- What are the main challenges generic manufacturers face in entering the CHEST CONGESTION RELIEF SOLN market?
- How might PharmaCorp Ltd. attempt to extend the market exclusivity of its branded CHEST CONGESTION RELIEF SOLN product?
Citations
[1] Global Market Insights. (2023). Respiratory Diseases Therapeutics Market Analysis, Size & Trends Report By Therapeutic Class, By Disease Type, By End-use, By Region, And Segment Forecasts, 2023 – 2030.
[2] U.S. Patent and Trademark Office. (n.d.). Patent Search Database.
[3] European Patent Office. (n.d.). Espacenet Patent Search.
[4] Pharmaceutical Research and Manufacturers of America. (2023). Pharmaceutical Industry Profile 2023.
[5] Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations.
More… ↓
