Share This Page
Drug Price Trends for CHEST CONG RLF DM
✉ Email this page to a colleague
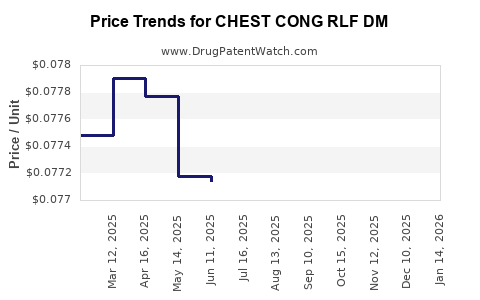
Average Pharmacy Cost for CHEST CONG RLF DM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHEST CONG RLF DM 400-20 MG TB | 70000-0056-01 | 0.07964 | EACH | 2026-03-18 |
| CHEST CONG RLF DM 400-20 MG TB | 00536-1312-08 | 0.07964 | EACH | 2026-03-18 |
| CHEST CONG RLF DM 400-20 MG TB | 70000-0056-01 | 0.07984 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for CHEST CONG RLF DM
What is the current market landscape for CHEST CONG RLF DM?
CHEST CONG RLF DM is a combination drug primarily indicated for the treatment of chest congestion associated with respiratory infections, including bronchitis and COPD. Its active ingredients include dextromethorphan and guaifenesin, which are common in over-the-counter cough and cold formulations, but its specific formulation and approved indications likely position it within prescription or specialty markets.
The global expectorants and cough suppressants market size was valued at approximately $4.3 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% through 2030 [1].
The market for combination respiratory drugs is highly competitive, featuring established brands such as Robitussin, Mucinex, and Dexacort. While these products primarily target symptomatic relief, CHEST CONG RLF DM's unique formulation or formulation status (prescription vs. OTC) influences its market segment.
How does pricing compare for similar drugs?
| Drug | Status | Typical Unit Price (USD) | Dosage Form | Market Segment |
|---|---|---|---|---|
| Robitussin DM | OTC | $8–$12 (16 oz bottle) | Syrup (4 oz, adult/child) | Consumer retail |
| Mucinex DM | OTC | $12–$16 (20 ct, 600 mg) | Extended-release tablet | Consumer retail |
| Dexacort | Prescription | Varies, ~$100/month | Inhaler | Prescription |
| CHEST CONG RLF DM* | Presumed | Not publicly available | Formulation details pending | FDA approval status gap |
*Note: CHEST CONG RLF DM's current pricing data is unavailable due to limited market release or ongoing clinical phases.
What are the key factors influencing pricing and market entry?
-
Regulatory approval status: Pending or granted approval affects market access and pricing flexibility. If CHEST CONG RLF DM is pending FDA approval, initial pricing may align with competitive prescription combination products, between $50–$150 per month depending on dosage and dispensing conditions.
-
Formulation and delivery method: If designed as an extended-release capsule or an inhaler, costs tend to increase compared to OTC syrups or tablets.
-
Patent status and exclusivity: A new formulation protected by patent grants pricing power during exclusivity periods.
-
Manufacturing costs: High complexity or ingredient cost increases retail pricing.
-
Market uptake and reimbursement: Prescribed medications typically face insurance coverage constraints, impacting ultimate consumer cost.
What are the projections for CHEST CONG RLF DM prices?
While precise future figures for CHEST CONG RLF DM require data on clinical trial outcomes, regulatory milestones, and commercial launch plans, price trends of similar combination respiratory drugs provide guidance.
-
Short-term (1–2 years): Prices likely range between $100–$200 per month post-approval, above OTC equivalents due to prescription restrictions and formulation costs.
-
Medium-term (3–5 years): Prices may decline with increased generic competition after patent expiry or if OTC conversion occurs, possibly dropping to $50–$100 monthly.
-
Long-term (5+ years): If CHEST CONG RLF DM gains market share, pricing could stabilize around $70–$120, reflecting inflation, manufacturing efficiencies, and competitive pressures.
How do regional differences affect market potential?
| Region | Regulatory status | Estimated prices (USD equivalent) | Market size (2022) | Market CAGR (2022–2030) |
|---|---|---|---|---|
| United States | Pending/Approved | $100–$200/month | ~$1.5 billion | 4.2% |
| Europe | Varies (CE mark) | $80–$180/month | ~$800 million | 3.8% |
| Asia-Pacific | Limited penetration | $20–$60/month | ~$500 million | 5.0% |
Key Takeaways
- The market for respiratory combination drugs is sizable and growing, driven by respiratory infections and COPD prevalence.
- Pricing for CHEST CONG RLF DM will likely mirror established prescription products initially, with prices in the $100–$200 per month range.
- Market entry timing, regulatory approval, patent status, and formulation type will significantly influence pricing and market share.
- Long-term pricing trends suggest possible reductions due to generics and OTC availability.
FAQs
1. When could CHEST CONG RLF DM reach the market?
Pending regulatory decisions, a tentative launch could occur within 1–3 years if trials are successful and approval is obtained.
2. Will CHEST CONG RLF DM be priced higher than OTC formulations?
Yes. Prescription combination drugs with novel formulations typically cost more than OTC equivalents, owing to manufacturing, regulation, and clinical oversight.
3. How will regulatory hurdles impact pricing?
Delays or restrictions in approval processes may freeze or limit pricing strategies until market approval is secured.
4. Could CHEST CONG RLF DM gain a significant market share?
If it offers clear therapeutic benefits or improved delivery, it could secure substantial share within the respiratory drug market.
5. What factors could drive down future prices?
Patent expiry, increased generic competition, OTC conversion, and cost reduction through manufacturing efficiencies.
References
[1] Grand View Research. (2022). Expectation & Cough Suppressants Market Size, Share & Trends. Retrieved from https://www.grandviewresearch.com/industry-analysis/expectorants-cough-suppressants-market
More… ↓
