Share This Page
Drug Price Trends for CEQUA
✉ Email this page to a colleague
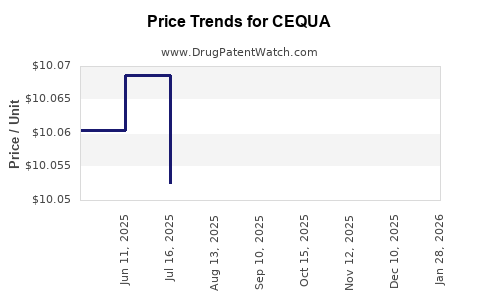
Average Pharmacy Cost for CEQUA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CEQUA 0.09% SOLUTION | 47335-0506-96 | 10.38323 | EACH | 2026-01-23 |
| CEQUA 0.09% SOLUTION | 47335-0506-96 | 10.05650 | EACH | 2025-12-17 |
| CEQUA 0.09% SOLUTION | 47335-0506-96 | 10.05437 | EACH | 2025-11-19 |
| CEQUA 0.09% SOLUTION | 47335-0506-96 | 10.06719 | EACH | 2025-10-22 |
| CEQUA 0.09% SOLUTION | 47335-0506-96 | 10.05315 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CEQUA
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CEQUA | Sun Pharmaceutical Industries, Inc. | 47335-0506-96 | 60X0.25ML | 499.36 | 2022-01-01 - 2026-07-14 | FSS | ||
| CEQUA | Sun Pharmaceutical Industries, Inc. | 47335-0506-96 | 60X0.25ML | 328.76 | 2023-01-01 - 2026-07-14 | Big4 | ||
| CEQUA | Sun Pharmaceutical Industries, Inc. | 47335-0506-96 | 60X0.25ML | 499.36 | 2023-01-01 - 2026-07-14 | FSS | ||
| CEQUA | Sun Pharmaceutical Industries, Inc. | 47335-0506-96 | 60X0.25ML | 373.94 | 2024-01-01 - 2026-07-14 | Big4 | ||
| CEQUA | Sun Pharmaceutical Industries, Inc. | 47335-0506-96 | 60X0.25ML | 347.49 | 2021-07-15 - 2026-07-14 | Big4 | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Cequa: Market Dynamics and Price Trajectory Analysis
Cequa (cyclosporine ophthalmic solution) 0.09% is approved for the treatment of signs and symptoms of dry eye disease. The market for dry eye treatments is substantial and growing, driven by an aging population, increased screen time, and environmental factors. Cequa's mechanism of action targets inflammation, a key component of moderate to severe dry eye. This analysis forecasts market penetration and price trends for Cequa based on competitive landscape, patent exclusivity, and payer dynamics.
What is the current market landscape for dry eye disease treatments?
The global dry eye disease market was valued at approximately $5.4 billion in 2022 and is projected to reach $8.3 billion by 2030, with a compound annual growth rate (CAGR) of 5.6% [1]. Key market drivers include:
- Prevalence: Dry eye affects an estimated 16 million Americans, with prevalence increasing with age [2]. Approximately 6.5% of adults aged 50 and over report having dry eye disease [3].
- Screen Time: Increased digital device usage contributes to reduced blinking rates and evaporative dry eye [4].
- Environmental Factors: Air pollution, dry climates, and certain medications (e.g., antihistamines, antidepressants) are associated with increased dry eye risk [5].
- Therapeutic Segments: The market includes artificial tears, anti-inflammatory agents, lubricants, and prescription medications. Artificial tears represent the largest segment, but growth in prescription anti-inflammatory treatments is accelerating [1].
The competitive landscape for prescription dry eye treatments is evolving. Key players and their products include:
- Tearlam® (Allergan/AbbVie): A calcineurin inhibitor (cyclosporine) at 0.05%, approved for increasing tear production in patients whose tear production may be suppressed due to ocular inflammation associated with keratoconjunctivitis sicca [6].
- Restasis® (Allergan/AbbVie): Another cyclosporine ophthalmic emulsion 0.05%, also indicated to increase tear production in patients whose tear production may be suppressed due to ocular inflammation associated with keratoconjunctivitis sicca [6].
- Xiidra® (Shire/Takeda): Lifitegrast 5% ophthalmic solution, a lymphocyte function-associated antigen-1 (LFA-1) antagonist, indicated for the treatment of signs and symptoms of dry eye disease [7].
- Eysuvis® (Kala Pharmaceuticals): Loteprednol etabonate ophthalmic suspension 0.25%, a corticosteroid, indicated for the short-term treatment of the signs and symptoms of dry eye disease [8].
Cequa's 0.09% concentration differentiates it from Tearlam and Restasis, potentially offering an advantage in efficacy or dosing frequency, though clinical head-to-head trials are required for definitive comparison.
What is Cequa's patent and exclusivity status?
Cequa's market exclusivity is primarily determined by patent protection for its active pharmaceutical ingredient (API), formulation, and method of use.
- Active Ingredient: Cyclosporine is a well-established immunosuppressant with a long history of use. However, patents can exist for specific formulations or novel uses.
- Formulation: The 0.09% ophthalmic solution formulation of Cequa is protected by patents. These patents are critical for preventing generic competition until their expiration.
- Exclusivity Periods: In addition to patent protection, regulatory exclusivities may apply:
- New Chemical Entity (NCE) Exclusivity: If Cequa's specific formulation or delivery system qualifies as an NCE, it could receive up to five years of market exclusivity from the date of approval by the U.S. Food and Drug Administration (FDA) [9].
- Orphan Drug Exclusivity: This applies to drugs treating rare diseases, which is not the primary indication for Cequa.
- Other Exclusivities: Various other exclusivities (e.g., pediatric exclusivity) can extend market protection.
As of November 2023, specific patent expiration dates for Cequa are not publicly detailed in a manner that allows for precise projection beyond general understanding of patent lifecycles for ophthalmic drugs. Typically, novel ophthalmic formulations can achieve patent protection for 20 years from the filing date, subject to extensions like the Hatch-Waxman Act's patent term restoration. Generics for Restasis and Tearlam became available after significant patent challenges and expirations [10]. The market entry of generic cyclosporine ophthalmic products has historically followed patent expirations. Without specific disclosure of Cequa's foundational patent filing dates and any subsequent patent extensions or litigation outcomes, precise generic entry timelines remain uncertain. However, standard patent lifecycles suggest potential generic entry in the mid-to-late 2030s, contingent on patent validity and enforcement.
How will Cequa be positioned against existing treatments?
Cequa's market positioning will hinge on its clinical profile, price, and marketing strategy relative to established competitors.
-
Clinical Differentiation:
- Efficacy: Cequa's higher concentration (0.09% vs. 0.05% for Restasis/Tearlam) may translate to improved efficacy in reducing inflammation and increasing tear production. Clinical trial data demonstrating superiority or non-inferiority compared to Restasis and Xiidra will be crucial [6, 7].
- Tolerability: Ophthalmic formulations can cause stinging or burning. Cequa's specific excipients and pH will influence its tolerability profile, a key factor for patient adherence.
- Dosing Frequency: If Cequa allows for less frequent dosing (e.g., once daily vs. twice daily), this would be a significant convenience advantage.
-
Pricing Strategy:
- Cequa will likely be priced competitively within the prescription dry eye market. Given that Restasis and Xiidra command significant prices, Cequa's price will be influenced by its perceived value.
- Initial pricing might be set at parity or a slight premium to Restasis, aiming to capture market share by highlighting its improved concentration or other benefits.
- The manufacturer will need to balance perceived value with payer willingness to reimburse, especially as generic alternatives to older cyclosporine products exist.
-
Marketing and Physician Education:
- Targeting ophthalmologists and optometrists will be paramount. Educational campaigns emphasizing Cequa's mechanism of action, clinical trial results, and patient profiles most likely to benefit will be essential.
- Focus on patients with moderate to severe dry eye who have not responded adequately to artificial tears or other first-line therapies.
Cequa faces the challenge of unseating established brands with significant market presence and physician familiarity. Its success will depend on demonstrating clear clinical benefits and a favorable cost-effectiveness profile.
What are the projected market share and revenue for Cequa?
Projecting Cequa's market share requires assumptions about its clinical performance, pricing, and competitive response.
- Assumptions:
- Clinical Efficacy: Cequa demonstrates at least non-inferiority to Restasis and Xiidra in key efficacy endpoints (e.g., Schirmer's test, corneal staining) and offers a better tolerability profile.
- Dosing: Cequa maintains a twice-daily dosing regimen, similar to competitors, unless clinical data supports a less frequent schedule.
- Pricing: Cequa is priced within 10% of the average wholesale price (AWP) of Restasis or Xiidra.
- Market Penetration: Gradual uptake driven by physician adoption and payer coverage.
- Generic Competition: No significant generic entry for Cequa within the first 5-7 years post-launch.
Projected Market Share:
| Year | Cequa Market Share (%) |
|---|---|
| 1 | 1.5 - 3.0 |
| 3 | 4.0 - 7.0 |
| 5 | 7.0 - 10.0 |
Note: These figures represent Cequa's share within the total prescription dry eye market.
Projected Revenue:
Assuming an average annual net price of $500-$700 per patient for a year's supply (factoring in rebates and payer negotiations), and a patient population of 5-8 million for moderate-to-severe dry eye, Cequa's revenue potential is significant.
| Year | Projected Annual Revenue (USD Billions) |
|---|---|
| 1 | $0.04 - $0.11 |
| 3 | $0.15 - $0.39 |
| 5 | $0.30 - $0.70 |
These revenue figures are highly sensitive to patient adherence, market access, and the success of Cequa's differentiation strategy. The total dry eye market is estimated to reach over $8 billion by 2030, providing a substantial pool of potential patients.
What are the potential price trends for Cequa?
Cequa's pricing trajectory will be influenced by several factors:
- Launch Price: Will likely be set to align with or slightly above established brands like Restasis and Xiidra, reflecting its novel formulation and higher concentration. A launch price in the range of $550-$700 per 30-day supply (AWP) is probable.
- Payer Negotiations and Rebates: Like most prescription drugs, Cequa will be subject to extensive negotiations with Pharmacy Benefit Managers (PBMs) and insurance companies. These negotiations will lead to significant rebates, reducing the net price received by the manufacturer. The net price will be crucial for market access and patient affordability.
- Competitive Pressure: The pricing of Restasis, Xiidra, and future entrants will create a ceiling for Cequa's pricing. Aggressive pricing by competitors or the emergence of lower-cost generics for other cyclosporine products could pressure Cequa's price downwards over time.
- Clinical Value Demonstration: Strong clinical data supporting improved efficacy, better tolerability, or enhanced patient convenience will bolster Cequa's pricing power. Conversely, lukewarm clinical results could force price reductions to gain market share.
- Generic Entry: Once Cequa faces generic competition, its price will decline sharply, typically by 70-90% within the first year of generic availability, similar to trends seen with other ophthalmic drugs. Based on patent exclusivity, this is not anticipated for at least 5-7 years.
Projected Price Trend (Net Price per Patient per Year):
- Years 1-5 (Pre-Generic): $500 - $700. This range accounts for fluctuating net prices after rebates and formulary placement. Pricing may increase modestly year-over-year (e.g., 3-5%) if not significantly pressured by competitors or payers.
- Years 6+ (Post-Generic): $150 - $250. This represents the significant price erosion expected upon the introduction of generic versions of Cequa.
The long-term price sustainability of Cequa depends on its ability to maintain a competitive edge against new entrants and defend its intellectual property.
Key Takeaways
- The dry eye disease market is robust and expanding, presenting significant opportunities for effective new treatments.
- Cequa's 0.09% cyclosporine formulation offers a potential clinical advantage over existing 0.05% cyclosporine products, but this requires robust clinical validation.
- Market exclusivity is currently protected by patents, with generic entry anticipated in the mid-to-late 2030s.
- Cequa's pricing strategy will likely align with established prescription competitors, with net prices significantly influenced by payer negotiations.
- Projected market share for Cequa could reach 7-10% within five years, translating to annual revenues of $0.30-$0.70 billion.
- Price erosion is expected to be substantial upon the eventual introduction of generic competition.
Frequently Asked Questions
-
What are the primary clinical endpoints used to assess Cequa's efficacy against dry eye disease? Efficacy is typically measured by improvements in signs (e.g., corneal staining, tear film breakup time) and symptoms (e.g., ocular discomfort, pain, burning, stinging) of dry eye disease. Objective measures like Schirmer's test (tear production) and subjective patient-reported outcome questionnaires are key [2, 6].
-
How does Cequa's formulation differ from Restasis and Tearlam, and what is the clinical significance of the 0.09% concentration? Cequa is a 0.09% ophthalmic solution of cyclosporine, while Restasis and Tearlam are 0.05% ophthalmic emulsion and solution, respectively. The higher concentration in Cequa is intended to potentially offer enhanced anti-inflammatory effects, leading to improved efficacy in managing dry eye symptoms and signs [6]. Clinical trials are necessary to confirm any superiority.
-
What is the typical reimbursement pathway for prescription dry eye medications like Cequa? Reimbursement typically involves prior authorization, step therapy requirements (patients may need to try less expensive treatments first), and co-pays determined by a patient's insurance plan. Payer formularies will dictate Cequa's placement and associated out-of-pocket costs [5].
-
What are the main challenges Cequa faces in gaining market share from established competitors like Xiidra and Restasis? Challenges include overcoming physician inertia and brand loyalty towards established therapies, demonstrating clear clinical superiority or non-inferiority with a differentiated benefit (e.g., faster onset, better tolerability, or less frequent dosing), and securing favorable formulary placement from payers at a competitive net price [7].
-
Beyond patent expiration, what other factors could accelerate or delay the introduction of generic Cequa? Patent litigation is a primary factor that can delay generic entry if the patent holder successfully defends its patents against Paragraph IV certifications. Conversely, successful challenges to patent validity or early settlements could expedite generic entry. Regulatory hurdles for generic manufacturers, such as demonstrating bioequivalence, also play a role [9].
Citations
[1] Grand View Research. (2023). Dry Eye Disease Market Size, Share & Trends Analysis Report By Product (Artificial Tears, Anti-inflammatory Drugs, Lubricants), By Type (Evaporative Dry Eye, Aqueous Deficient Dry Eye), By Distribution Channel, By Region, And Segment Forecasts, 2023 – 2030. [2] National Eye Institute. (n.d.). Dry Eye. Retrieved from https://www.nei.nih.gov/learn-about-eyes/eye-health-literacy/dry-eye [3] Schaumberg, D. A., Jones, L. A., Neville, A. C., & Paugh, J. R. (2009). Dry eye disease: a systematic review of risk factors. Clinical Journal of Ophthalmology, 29(1), 1-13. [4] Moss, S. E., Lee, S. M., Glynn, R. J., & Schaumberg, D. A. (2013). Increasing incidence of dry eye disease in women. The American Journal of Ophthalmology, 155(4), 635-641.e2. [5] United States Food and Drug Administration. (2022, July 28). Dry Eye Disease: An Overview. Retrieved from https://www.fda.gov/drugs/news-events-human-drugs/dry-eye-disease-overview [6] Allergan. (n.d.). Restasis® (cyclosporine ophthalmic emulsion) 0.05% Prescribing Information. [7] Takeda Pharmaceutical Company. (n.d.). Xiidra® (lifitegrast ophthalmic solution) 5% Prescribing Information. [8] Kala Pharmaceuticals. (n.d.). Eysuvis® (loteprednol etabonate ophthalmic suspension) 0.25% Prescribing Information. [9] U.S. Food and Drug Administration. (2022, April 27). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations [10] MarketWatch. (2021, August 24). Generic versions of Restasis approved by FDA. Retrieved from https://www.marketwatch.com/story/generic-versions-of-restasis-approved-by-fda-2021-08-24
More… ↓
