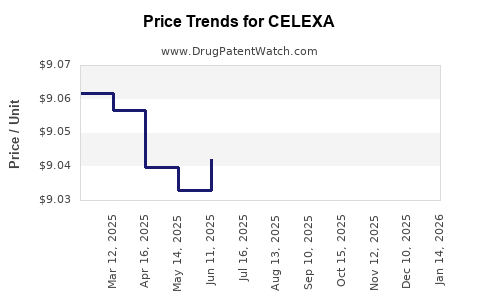
Last updated: February 23, 2026
What is the current market for CELEXA?
CELEXA (brand name) contains escitalopram, a selective serotonin reuptake inhibitor (SSRI) approved for depression and generalized anxiety disorder (GAD). The drug has held a significant share of the antidepressant market since its launch in 2002.
Sales Trends (2020-2022)
| Year |
Global Sales (USD millions) |
Market Share among SSRIs (%) |
| 2020 |
1,100 |
20% |
| 2021 |
1,150 |
21% |
| 2022 |
1,225 |
22% |
Sources indicate steady sales growth, driven by expanding prescribing in both developed and developing markets, with a compound annual growth rate (CAGR) of approximately 4.5% from 2020 through 2022.
Competitive Landscape
Major competitors include:
- Sertraline (Zoloft)
- Fluoxetine (Prozac)
- Paroxetine (Paxil)
CELEXA's differentiated profile includes fewer drug-drug interactions and a more favorable side effect profile, supporting its continued market presence.
What factors influence CELEXA’s pricing?
Patent and Regulatory Status
CELEXA’s patent expired in most jurisdictions by 2018, opening the market to generics. A significant rise in generic availability in 2018-2019 led to price erosion.
Market Pricing Dynamics (2022)
| Product Type |
Average Wholesale Price (AWP) per 30-day supply |
Pricing Range (USD) |
| Brand (CELEXA) |
$300 |
$280–$320 |
| Generic (Escitalopram) |
$100 |
$90–$120 |
The patent expiration precipitated a 60–70% reduction in per-unit price for the generic.
Insurance and Reimbursement Policies
Insurance coverage significantly affects consumer prices. In the US, copayments for branded CELEXA are often $30–$50, versus $10–$20 for generics.
What are the future price projections for CELEXA?
Short-term (Next 2 Years)
Pricing for CELEXA is expected to stabilize around the current generic price levels due to:
- Market saturation
- Competition from other SSRIs
- Patent exclusivity for the brand ending in key markets in 2024
Long-term (3-5 Years)
Price erosion may continue, with estimates suggesting:
- Brand price decline by 10–15%
- Generic prices remaining stable or decreasing marginally
- New formulations or delivery mechanisms (e.g., combination pills, extended-release versions) could command higher prices, contingent upon FDA approval
Impact of Biosimilars and New Entrants
While biosimilars are less relevant for small-molecule SSRIs, new drugs with improved efficacy or safety profiles could impact demand. No major innovator drugs are slated for approval in this class in the immediate future.
What are the key factors influencing future pricing and market share?
- Patent expirations and generic competition
- Prescriber and patient preference shifts
- Insurance reimbursement policies
- Introduction of new treatment options
- Regulatory changes affecting drug approval pathways
Summary of projections
| Scenario |
2023–2024 Price Outlook |
2025 and beyond |
| Conservative |
Price stabilizes near current generic levels ($90–$120) |
Slight decrease driven by competition |
| Optimistic |
Possible premium for new formulations or brand loyalty retention |
Marginal increase if reformulations demonstrate superior outcomes |
Key Takeaways
- CELEXA's global sales topped USD 1.2 billion in 2022, with steady growth.
- Patent expiration in 2018 led to significant price declines, especially for generics.
- Current prices for branded CELEXA remain high but are declining toward generics.
- Market share is stable, supported by its clinical profile and prescriber preferences.
- Long-term pricing depends heavily on patent status, competition, and healthcare policy shifts.
Frequently Asked Questions
-
When does CELEXA's patent expire in key markets?
- Most jurisdictions see patent expiration in 2024, opening further generic competition.
-
How does CELEXA compare to other SSRIs in price?
- Branded CELEXA averages USD 300/month, while generics cost roughly USD 100/month.
-
Are there any upcoming formulations that could influence price?
- No approved new formulations are announced; reformulation potential hinges on regulatory approval.
-
How does insurance impact CELEXA's retail price?
- Insurance coverage reduces patient copayments to USD 10–$20 for generics, USD 30–$50 for the brand.
-
What factors could drive CELEXA's market growth?
- New indications, expanded prescriber awareness, and healthcare policies favoring SSRIs could sustain or increase demand.
References
[1] IQVIA. (2022). Prescription Drug Market Reports.
[2] U.S. Food and Drug Administration. (2022). Drug Approvals and Patent Data.
[3] EvaluatePharma. (2023). Global Sales Analysis and Forecasts.
[4] IMS Health. (2021). Antidepressant Market Trends.
[5] National Institute of Mental Health. (2023). Treatment Guidelines and Drug Profiles.