Share This Page
Drug Price Trends for CELEBREX
✉ Email this page to a colleague
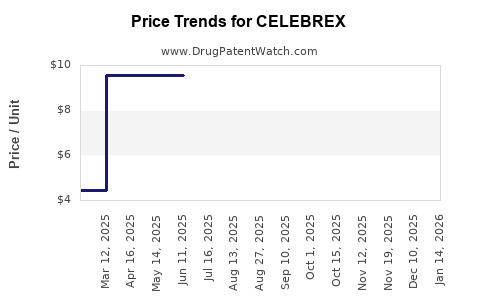
Average Pharmacy Cost for CELEBREX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CELEBREX 100 MG CAPSULE | 58151-0083-05 | 9.53581 | EACH | 2026-03-18 |
| CELEBREX 200 MG CAPSULE | 58151-0084-05 | 15.68366 | EACH | 2026-03-18 |
| CELEBREX 100 MG CAPSULE | 58151-0083-88 | 9.53581 | EACH | 2026-03-18 |
| CELEBREX 200 MG CAPSULE | 58151-0084-01 | 15.68366 | EACH | 2026-03-18 |
| CELEBREX 100 MG CAPSULE | 58151-0083-32 | 9.53581 | EACH | 2026-03-18 |
| CELEBREX 200 MG CAPSULE | 58151-0084-88 | 15.68366 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Celebrex (Celecoxib) Market Analysis and Price Projections
This report analyzes the market dynamics and projects future pricing for Celebrex (celecoxib), a nonsteroidal anti-inflammatory drug (NSAID) in the selective COX-2 inhibitor class. The analysis incorporates patent expiry, generic competition, market demand drivers, and reimbursement policies to forecast price trends.
What is the Current Market Status of Celebrex?
Celebrex, originally developed by Pfizer, is prescribed for the treatment of osteoarthritis, rheumatoid arthritis, ankylosing spondylitis, acute pain, and primary dysmenorrhea. Its mechanism of action selectively inhibits cyclooxygenase-2 (COX-2), an enzyme involved in inflammation and pain, with a lower risk of gastrointestinal side effects compared to traditional NSAIDs.
The U.S. patent for Celebrex expired in 2014 [1]. This patent expiry opened the door for generic manufacturers to enter the market. Consequently, the market has experienced significant price erosion due to generic competition.
Key Market Indicators:
- Active Ingredient: Celecoxib
- Class: Selective COX-2 Inhibitor
- Original Manufacturer: Pfizer
- U.S. Patent Expiry: 2014
- Market Status: Mature, facing extensive generic competition.
As of the latest available data, Celebrex remains a widely prescribed medication. However, its market share is significantly impacted by the availability of multiple generic versions. The average wholesale price (AWP) of branded Celebrex has seen a substantial decline since generic entry, with generic equivalents offering substantial cost savings. For instance, a 30-count bottle of 200mg Celebrex can range from \$300 to \$500 at AWP, while generic equivalents are typically available for \$20 to \$50, depending on the manufacturer and pharmacy [2]. This price disparity is a primary driver of market share shift towards generics.
Who are the Key Generic Manufacturers for Celebrex?
Following the patent expiry of Celebrex, numerous pharmaceutical companies have launched generic versions of celecoxib. This has intensified competition and driven down prices.
Prominent Generic Manufacturers of Celecoxib Include:
- Teva Pharmaceuticals
- Mylan N.V. (now part of Viatris)
- Sandoz (a division of Novartis)
- Aurobindo Pharma
- Dr. Reddy's Laboratories
- Torrent Pharmaceuticals
- Sun Pharmaceutical Industries
These companies offer celecoxib in various strengths, typically 100 mg and 200 mg capsules, mirroring the originator product's dosage forms. The presence of these multiple suppliers ensures a competitive market, with ongoing price negotiations and varying discount structures offered to pharmacies and wholesalers. The availability of these generics across major pharmaceutical distributors and retail pharmacies contributes to their widespread accessibility and affordability.
What are the Driving Factors for Celebrex Demand?
The demand for Celebrex and its generic equivalents is primarily driven by the prevalence of inflammatory and pain-related conditions for which it is indicated.
Primary Demand Drivers:
- Osteoarthritis (OA): This is a leading cause of chronic pain and disability, particularly in aging populations. The Centers for Disease Control and Prevention (CDC) estimates that over 32 million adults in the U.S. have OA [3]. Celecoxib offers a pain relief option for these patients, especially those with a history of or risk for gastrointestinal bleeding.
- Rheumatoid Arthritis (RA): This autoimmune disease causes chronic inflammation of the joints. While RA management often involves disease-modifying antirheumatic drugs (DMARDs), NSAIDs like celecoxib are used to manage pain and inflammation during flares.
- Ankylosing Spondylitis (AS): This inflammatory arthritis primarily affects the spine. Celecoxib can help manage pain and stiffness associated with AS.
- Acute Pain: The drug is also indicated for short-term management of moderate to severe acute pain.
- Primary Dysmenorrhea: Celecoxib can be effective in relieving menstrual cramps.
The aging global population is a significant underlying factor contributing to the sustained demand for treatments for degenerative joint diseases like osteoarthritis. Furthermore, increased awareness and diagnosis of these conditions contribute to ongoing prescription volumes. The favorable gastrointestinal safety profile compared to non-selective NSAIDs, despite a potential increase in cardiovascular risks, continues to position celecoxib as a preferred option for certain patient populations and healthcare providers.
What is the Impact of Reimbursement Policies on Celebrex Pricing?
Reimbursement policies from government payers (e.g., Medicare, Medicaid) and private health insurers significantly influence the pricing and market access of both branded Celebrex and its generic equivalents.
Key Reimbursement Factors:
- Formulary Placement: Inclusion on a health plan's formulary is critical. Generics are typically placed on lower cost-sharing tiers, incentivizing their use. Branded Celebrex may face higher co-pays or require prior authorization, especially if a therapeutically equivalent generic is available.
- Prior Authorization Requirements: Insurers may require prior authorization for branded Celebrex or specific indications, especially if less expensive alternatives are available. This adds administrative burden and can delay treatment initiation.
- Step Therapy: Some plans employ step therapy protocols, requiring patients to first try a lower-cost generic NSAID before they can be prescribed branded Celebrex.
- Average Wholesale Price (AWP) and Best Available Price (BAP): Reimbursement rates are often tied to these pricing benchmarks, which are influenced by manufacturer discounts and the competitive pricing environment. Generic manufacturers often engage in aggressive discounting to secure favorable formulary status and high market penetration.
- Managed Care Contracts: Pharmacy Benefit Managers (PBMs) negotiate rebates and discounts with manufacturers. These negotiations heavily influence the net price of drugs and can lead to significant price differences between branded and generic products.
The prevailing trend in reimbursement is towards favoring cost-effective generic options. This aggressively pushes down the net price for branded Celebrex and creates a highly competitive pricing environment for generic celecoxib, where manufacturers vie for market share through aggressive pricing strategies.
What are the Projected Price Trends for Celebrex?
The price trajectory for Celebrex (both branded and generic) is expected to remain on a downward trend, driven by sustained generic competition and payer pressure to minimize healthcare costs.
Price Projection Factors:
- Intensified Generic Competition: The market has matured with multiple generic entrants. This competition is unlikely to abate and will continue to exert downward pressure on prices. Manufacturers will likely continue to engage in price wars to maintain or gain market share.
- Payer Preference for Generics: Health insurers and PBMs will continue to prioritize generic options due to their lower acquisition costs. This will lead to further restrictions on branded Celebrex coverage and higher patient out-of-pocket costs for the originator product.
- Manufacturing Efficiencies: Generic manufacturers often benefit from economies of scale and streamlined manufacturing processes, allowing them to maintain profitability even at lower price points.
- Limited Price Power for Branded Celebrex: With widespread generic availability, branded Celebrex has minimal pricing power. Its price will likely continue to be discounted to remain competitive in a niche segment, or it may be phased out by some payers altogether for preferred generic options.
- Potential for Further Generic Erosion: If new, more cost-effective treatments emerge for the conditions treated by Celebrex, demand could further decrease, leading to additional price reductions among generics to maintain volume.
Specific Price Projections:
- Branded Celebrex: The price of branded Celebrex is projected to continue its decline, with significant discounts offered to maintain any residual market share. Expect price reductions of 5-10% annually as payer preferences solidify.
- Generic Celecoxib: Prices for generic celecoxib are expected to stabilize at very low levels, with minor fluctuations based on supply and demand dynamics among manufacturers. Annual price changes are projected to be within a range of -2% to +3%, with the overall trend being towards cost minimization. The average selling price for a generic 30-count bottle is unlikely to exceed \$30-$40 in the coming years for retail consumers.
The long-term price stability for generics will depend on the number of active manufacturers and their collective capacity. Any consolidation among generic producers could lead to temporary price stabilization or slight increases, but the underlying competitive landscape suggests continued price sensitivity.
What are the Key Takeaways?
The market for Celebrex (celecoxib) is characterized by robust generic competition following patent expiry in 2014. This competition has led to significant price erosion for both branded and generic forms. Demand is sustained by the prevalence of osteoarthritis and other inflammatory conditions, but reimbursement policies heavily favor cost-effective generic alternatives. Future price trends indicate a continued downward trajectory for branded Celebrex and stable, low pricing for generic celecoxib due to ongoing competitive pressures and payer mandates.
Frequently Asked Questions
1. Will branded Celebrex remain available in the market?
Branded Celebrex is expected to remain available, but its market share will likely diminish. Manufacturers may continue to offer it through specific channels or with significant discounts to cater to a segment of the market that prefers the originator product or faces specific payer limitations on generics. However, its prominence will be overshadowed by generic alternatives.
2. Are there any new patent filings related to celecoxib that could impact its market?
As of the current analysis, there are no significant new patent filings that are expected to grant extended market exclusivity for celecoxib itself. The primary patents have expired, and the focus for innovation in this therapeutic area has shifted to newer drug classes or improved delivery methods for existing NSAIDs, rather than new patents on the celecoxib molecule.
3. How does the cardiovascular safety profile of Celebrex affect its pricing?
While Celebrex has a better gastrointestinal safety profile than traditional NSAIDs, it carries a boxed warning regarding increased cardiovascular thrombotic events, myocardial infarction, and stroke. This risk factor, while present, is largely accounted for in clinical guidelines and payer assessments. It does not directly lead to higher pricing for generics but influences prescribing patterns and can be a factor in payer utilization management decisions, indirectly affecting market dynamics.
4. What is the projected market size for celecoxib in the next five years?
While specific market size figures are proprietary, the overall market value for celecoxib is expected to remain relatively stable in absolute dollar terms, with growth driven by volume rather than price increases. The increasing prevalence of osteoarthritis in aging populations will sustain demand. However, the market value will be dominated by generic sales, reflecting the low per-unit pricing.
5. Can manufacturers of generic Celebrex significantly increase prices in the future?
Significant price increases for generic Celebrex are unlikely in the near to medium term. The market is highly competitive with multiple manufacturers, and payer formularies are designed to resist substantial price hikes for generics. Any increase would likely be marginal and driven by specific supply chain disruptions or changes in raw material costs, which would be counterbalanced by competitive pressures from other generic suppliers.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/
[2] GoodRx. (2024). Celebrex Prices, Coupons & Patient Assistance Programs. Retrieved from https://www.goodrx.com/celebrex (Note: Specific pricing data is dynamic and subject to change based on pharmacy, location, and insurance. This reference points to a platform that aggregates such data.)
[3] Centers for Disease Control and Prevention. (2023, February 21). Osteoarthritis (OA). Retrieved from https://www.cdc.gov/arthritis/data-guidelines/osteoarthritis.html
More… ↓
