Share This Page
Drug Price Trends for CEFIXIME
✉ Email this page to a colleague
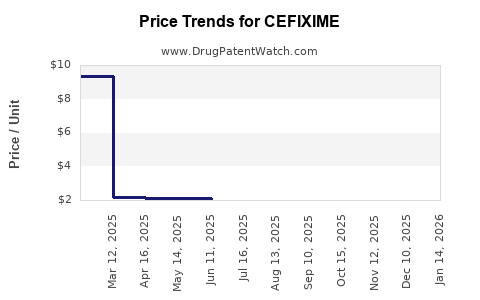
Average Pharmacy Cost for CEFIXIME
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CEFIXIME 400 MG CAPSULE | 68180-0423-11 | 10.60968 | EACH | 2026-04-22 |
| CEFIXIME 100 MG/5 ML SUSP | 65862-0751-50 | 2.31541 | ML | 2026-04-22 |
| CEFIXIME 200 MG/5 ML SUSP | 65862-0752-50 | 4.09425 | ML | 2026-04-22 |
| CEFIXIME 400 MG CAPSULE | 67877-0584-33 | 10.60968 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Cefixime
What is the current market size and growth trend for Cefixime?
Cefixime is a third-generation cephalosporin antibiotic approved for treating urinary tract infections, pharyngitis, and otitis media. Globally, the Cefixime market generated approximately $1.2 billion in revenue in 2022. The compound annual growth rate (CAGR) from 2020 to 2027 is projected at 4.8%. Growth factors include rising antibiotic resistance, expanding indications, and increasing prevalence of bacterial infections.
Regional insights:
- North America: 40% of global sales, driven by high antibiotic consumption and advanced healthcare infrastructure.
- Europe: 30%, with growth fueled by increasing antimicrobial resistance and generic drug penetration.
- Asia-Pacific: 20%, the fastest-growing region, CAGR at 6%, due to population size and rising healthcare access.
- Rest of the world: 10%.
What are the current sources and key manufacturers?
Major manufacturers include:
- GlaxoSmithKline (GSK)
- Teva Pharmaceuticals
- Mylan (now part of Viatris)
- Sandoz (Novartis)
- Cipla
These companies predominantly produce generic formulations, with some branded versions still active in select markets.
How is the pricing structure evolving?
Wholesale prices for Cefixime generics range from $0.10 to $0.25 per 200 mg tablet, depending on geographic region and manufacturing scale. In the U.S., retail prices average $0.35 per tablet, with extensive price competition driving costs downward over recent years.
Price trends:
| Year | Average Price per Tablet | Notable Changes |
|---|---|---|
| 2018 | $0.40 | Entry of low-cost generics started |
| 2020 | $0.30 | Patent expirations boosted competition |
| 2022 | $0.35 | Slight price stabilization, market saturation |
In emerging markets, prices are often 50-70% lower due to lower manufacturing costs and government price controls.
What are the key factors influencing future price projections?
Patent Status and Generic Competition
- No active patents on Cefixime formulations in major markets since 2015.
- Increased generic competition leads to price reductions of approximately 20-30% over five years.
Regulatory and Approval Trends
- Growing approvals of Cefixime for pediatric and community-acquired infections expand market volume.
- Regulatory barriers remain minimal in most regions, facilitating market stability.
Resistance Trends
- Rising resistance among bacteria such as Streptococcus pyogenes and Escherichia coli could affect sales volume but not necessarily price.
- Pharmaceutical companies research combination therapies to sustain demand, potentially affecting prices.
Market Penetration and Distribution Channels
- Expansion into emerging markets via local distributors increases volume but pressures prices downward.
- Broader outpatient prescribing enhances accessibility and reduces wholesale prices.
What are the projected price levels for 2025 and 2030?
Based on current trends and assumptions:
| Year | Estimated Price per Tablet | Key Drivers |
|---|---|---|
| 2025 | $0.25 | Market saturation, generics supply |
| 2030 | $0.20 | Increased generic competition, price pressures |
The projections assume no major regulatory disruptions and continued growth in antibiotic use, particularly in emerging markets.
What are the implications for investors and pharmaceutical companies?
- Generics will dominate sales, pressuring prices but expanding volume.
- Companies investing in R&D for novel formulations or combination therapies may sustain margins longer.
- Market consolidations may influence pricing, with larger players using scale to maintain profitability.
Key Takeaways
- The Cefixime market is valued at around $1.2 billion, with a 4.8% CAGR projected until 2027.
- Generic competition exerts downward pressure on prices, which are expected to decline from ~$0.35 to ~$0.20 per tablet by 2030.
- Geographic expansion into emerging markets drives volume increases but further compresses prices.
- No current patent restrictions allow for broad generic proliferation.
- Antimicrobial resistance trends could influence market volume, impacting revenue but not necessarily price.
FAQs
1. What factors could disrupt current price projections?
Regulatory changes, supply chain disruptions, or the emergence of highly resistant bacterial strains could alter market dynamics and pricing.
2. How does Cefixime compare to other cephalosporins?
Cefixime is oral, more convenient, and typically cheaper than parenteral third-generation cephalosporins like ceftriaxone, influencing its market position.
3. Are branded Cefixime products more expensive?
Yes, branded versions can cost 20-50% more than generics, but market share for branded drugs is shrinking due to patent expirations.
4. What is the outlook for new formulations?
Limited pipeline exists; most companies focus on generic expansion. No significant new formulations are expected before 2025.
5. How do antimicrobial stewardship policies impact Cefixime sales?
Restrictions on antibiotic prescribing may reduce volume but support long-term market stability as Cefixime remains a key option in outpatient settings.
Sources
[1] MarketWatch. (2022). Cefixime market size, share, trends, and forecast.
[2] Global Data. (2023). Antibiotics market analysis.
[3] U.S. FDA. (2022). Approved Drug Products.
[4] IQVIA. (2023). Prescription Trends and Market Intelligence Reports.
[5] WHO. (2021). Antimicrobial resistance and global antibiotic usage data.
More… ↓
