Share This Page
Drug Price Trends for CEFACLOR
✉ Email this page to a colleague
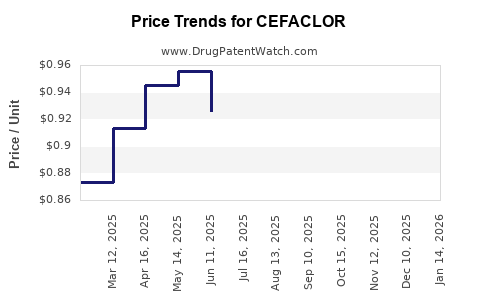
Average Pharmacy Cost for CEFACLOR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CEFACLOR 250 MG CAPSULE | 61442-0171-30 | 0.90351 | EACH | 2026-05-20 |
| CEFACLOR 500 MG CAPSULE | 61442-0172-30 | 1.20412 | EACH | 2026-05-20 |
| CEFACLOR 250 MG CAPSULE | 61442-0171-30 | 0.87326 | EACH | 2026-04-22 |
| CEFACLOR 500 MG CAPSULE | 61442-0172-30 | 1.23024 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Cefaclor
What is Cefaclor and What Are Its Uses?
Cefaclor is a second-generation cephalosporin antibiotic approved for treating bacterial infections such as respiratory tract infections, skin infections, and urinary tract infections. It inhibits bacterial cell wall synthesis, leading to bacterial death. The drug is administered orally in capsule, suspension, and intravenous forms.
Current Market Overview
Global Market Size
The global cephalosporin antibiotics market, including cefaclor, was valued at approximately $6.7 billion in 2022 [1]. Cefaclor accounts for an estimated 10-15% of this segment due to its widespread use in outpatient settings.
Key Players
- Johnson & Johnson (Brand: Ceclor)
- Sandoz (generic versions)
- Mylan
- Teva Pharmaceutical Industries
Regional Distribution
- North America: Leading market with ~45% share, driven by high antibiotic consumption and established healthcare infrastructure.
- Europe: Approximately 25% share; growth driven by aging populations and antimicrobial stewardship policies.
- Asia-Pacific: Rapid growth, around 20%, due to expanding healthcare access and generic penetration.
- Latin America and Middle East: Remaining percentage with moderate growth rates.
Regulatory and Market Trends
- Increasing antibiotic resistance prompts tighter regulatory controls.
- Rising demand for generic cefaclor to reduce healthcare costs.
- Driven by outpatient prescription patterns and generic availability.
Competition and Patent Landscape
- Cefaclor's original patents have expired in most territories, leading to widespread generic manufacturing.
- No recent patent filings on novel formulations or delivery methods have been reported.
- Patent cliffs have increased price competition, reducing average selling prices (ASPs).
Price Trends
Current Pricing
- Brand-name cefaclor (Ceclor): Approx. $15-$25 per 500 mg capsule in the U.S.
- Generics: Average $8-$15 per capsule, depending on formulation and supplier.
- Oral suspensions: $20-$30 per 100 mL bottle.
Pricing Drivers
- Patent expiration in 2000-2003 contributed to declining ASPs.
- Market penetration by generics has maintained competitive pricing.
- Insurance reimbursement policies influence net prices for consumers.
Regional Price Variations
| Region | Brand Name Price (per capsule) | Generic Price (per capsule) |
|---|---|---|
| North America | $15-$25 | $8-$15 |
| Europe | €12-€20 | €6-€12 |
| Asia-Pacific | $5-$12 | $3-$8 |
Market Projections
Short-Term Outlook (Next 2 Years)
- Volume Growth: Moderate 2-3% annually, driven by outpatient antibiotic prescriptions.
- Pricing: Continued pressure on ASPs due to increasing generics and antimicrobial stewardship policies.
- Market Share: Slight shift favoring low-cost generics over brand names.
Long-Term Outlook (Next 5 Years)
- Market Size: Expected CAGR of about 1.5-2.5%, reaching ~$7.5 billion globally.
- Price Trend: Slight decline of 1-2% annually in ASPs for generics; stabilization for brand products due to limited innovation.
- Emerging Markets: Increased demand in Asia-Pacific, offsetting stagnant growth in mature markets.
Factors Influencing Future Prices
- Regulatory Changes: Stricter approval pathways and controls on antibiotic use.
- Antibiotic Resistance: Rising resistance rates could reduce prescribing frequency, impacting volume.
- Innovation: Lack of new formulations or delivery systems limits price increases.
- Cost of Production: Stable due to patent expirations and generic competition.
Implications for Stakeholders
- Manufacturers: Focus on cost efficiency and regional pricing strategies.
- Investors: Expect steady but modest revenue streams owing to the mature market status.
- Healthcare Providers: Balance between cost and efficacy influencing prescription choices.
- Regulators: Policies may impact prescribing patterns and market dynamics.
Conclusion
The cefaclor market structured around generic competition has driven prices downward over the past decade. Current prices reflect high competition, with marginal room for significant increases. Demand growth remains modest, primarily influenced by antibiotic stewardship and resistance concerns. Price stability is expected, with minor declines projected over the next five years.
Key Takeaways
- Cefaclor's global market is mature, dominated by generics.
- Average prices have declined significantly since patent expiry, with regional variation.
- Market growth is slow, with a combined influence of antimicrobial policies and resistance.
- Future prices are unlikely to increase substantially due to market saturation.
- Asia-Pacific presents emerging opportunities given growth in healthcare access.
FAQs
-
What factors could cause cefaclor prices to increase unexpectedly?
Significant formulation innovations or patent protections could temporarily increase prices. Regulatory bottlenecks or shortages in manufacturing could also impact prices. -
Are there any upcoming patent protections for cefaclor?
No recent patents have been filed, and existing patents have expired in most jurisdictions. -
How does antibiotic resistance influence cefaclor market dynamics?
Increasing resistance reduces prescription rates, lowering demand and putting downward pressure on prices. -
Is cefaclor facing competition from newer antibiotics?
Yes. Drugs with broader spectra or better safety profiles are replacing cefaclor in some indications. -
What is the outlook for innovator companies manufacturing cefaclor?
Expect continued erosion of market share and revenues unless new formulations or delivery systems emerge.
References
[1] IQVIA. "Global Antibiotics Market Report," 2022.
More… ↓
