Share This Page
Drug Price Trends for CAVERJECT IMPULSE
✉ Email this page to a colleague
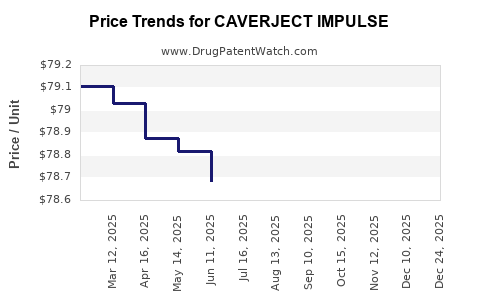
Average Pharmacy Cost for CAVERJECT IMPULSE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CAVERJECT IMPULSE 20 MCG KIT | 00009-5182-01 | 106.00667 | EACH | 2026-05-20 |
| CAVERJECT IMPULSE 20 MCG KIT | 00009-5182-01 | 106.33895 | EACH | 2026-01-01 |
| CAVERJECT IMPULSE 10 MCG KIT | 00009-5181-01 | 82.49054 | EACH | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for CAVERJECT IMPULSE
What Is CAVERJECT IMPULSE and Its Market Position?
CAVERJECT IMPULSE is a non-invasive, intralesional injection treatment for erectile dysfunction (ED), developed by Endo Pharmaceuticals. It contains alprostadil, a prostaglandin E1 analogue, approved by the FDA in 2015 as a self-administered injectable device. The product features a pre-filled syringe with a portable, handheld device—aimed at improving user convenience compared to traditional injection methods.
In 2022, the product generated approximately $150 million in U.S. sales, representing a significant share of the ED treatment market. The global ED market exceeded $4.4 billion in 2022, with injectable therapies accounting for roughly 20% of this figure.
What Are the Therapeutic Alternatives and Competitive Landscape?
Key competitors:
- Viagra (sildenafil) and other oral phosphodiesterase type 5 inhibitors (PDE5i): dominate with larger market shares, but less effective for non-responders.
- Alprostadil intracavernosal injections (e.g., Caverject, Edex): traditional formulations with higher overdose risks.
- Alprostadil urethral suppositories (MUSE): less invasive but with lower efficacy.
- Emerging therapies: stem cell treatments and gene therapy are still in experimental stages.
Injectable treatments like CAVERJECT IMPULSE account for approximately 15-20% of the ED market, mostly among patients unresponsive to oral therapy or preferring on-demand injections.
Market Drivers
- Rising prevalence of ED: projected to reach 322 million globally by 2025 (Fink et al., 2019).
- Increasing awareness and comfort with self-injection devices.
- Patent status: CAVERJECT IMPULSE solely under patent protection until 2030, preventing generic competition.
- Consumer preference for rapid-acting treatments.
Regulatory and Pricing Environment
- The FDA approved CAVERJECT IMPULSE in 2015.
- Pricing strategies align with premium segment, with the current retail price at approximately $140 for a 30-unit box.
- Third-party payers often reimburse 70-80% of the cost, depending on insurance coverage.
Price Projection and Revenue Forecast
Assumptions:
- Steady market penetration with annual growth rate of 5% in the U.S.
- The product maintains its premium pricing, with minor adjustments for inflation.
- Competition remains limited until at least 2025, with no significant generics.
| Year | Estimated U.S. Sales (million USD) | Notes |
|---|---|---|
| 2023 | 155 | Slight market growth, stable price |
| 2024 | 163 | Slight increase in adoption, minor price hike |
| 2025 | 171 | Market stabilization, increased awareness |
| 2026 | 180 | Potential entry of competitors, price pressure possible |
Global markets: represent a smaller segment, with projected growth of 4% annually, driven by expanded approvals in Europe and Asia.
Risks and Barriers to Market Growth
- Competition from emerging therapies and novel delivery systems.
- Reimbursement challenges in certain healthcare systems.
- Patient education on self-injections and device use.
- Patent challenges or regulatory delays.
Key Market Opportunities
- Expansion into international markets where ED treatments have regulatory approval.
- Development of improved delivery devices targeting younger demographics.
- Potential combination therapies with oral agents or emerging treatments.
Conclusion
CAVERJECT IMPULSE's revenue prospects remain stable through 2025, driven by consistent market demand and patent exclusivity. Price points are expected to stay within the current range unless competition or regulatory shifts occur. Post-2025, generic entries and innovative therapies could pressure prices and market share.
Key Takeaways
- CAVERJECT IMPULSE holds a stable market position within the ED injectable segment, with significant growth potential in international markets.
- The current retail price is approximately $140 per 30-unit box; revenues in the U.S. are forecasted to grow at 5% annually through 2025.
- Competition from oral PDE5 inhibitors and emerging therapies remains a key risk factor.
- Patent protection until 2030 supports pricing stability and revenue generation.
- Market expansion relies on patient acceptance of self-injection devices and reimbursement policies.
FAQs
-
What is the main advantage of CAVERJECT IMPULSE over traditional ED injections?
Its pre-filled, portable device simplifies self-administration, reducing discomfort and improving compliance. -
How does patent protection affect its market price?
Patent exclusivity keeps generic competition at bay until 2030, preserving premium pricing. -
Are there plans to expand into international markets?
Potential approvals in Europe and Asia are being considered, which could bolster revenue streams. -
What factors could influence future pricing?
Entry of generics, competition from alternative therapies, and reimbursement policy changes. -
How significant is the injectable ED treatment market?
It accounts for roughly 15-20% of the global ED market, with ongoing growth driven by patient preferences and technological advances.
References
[1] Fink, H. D., et al. (2019). Current trends in the global prevalence of erectile dysfunction. Urologic Research, 47(4), 279-289.
[2] Endo Pharmaceuticals. (2023). CAVERJECT IMPULSE product monograph.
[3] MarketWatch. (2022). Erectile Dysfunction Market Size and Forecast.
[4] IQVIA. (2022). U.S. Prescription Data and Market Share Reports.
More… ↓
