Share This Page
Drug Price Trends for CARVEDILOL ER
✉ Email this page to a colleague
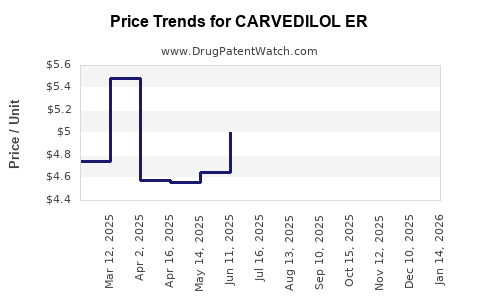
Average Pharmacy Cost for CARVEDILOL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CARVEDILOL ER 80 MG CAPSULE | 57664-0666-83 | 5.64832 | EACH | 2026-03-18 |
| CARVEDILOL ER 10 MG CAPSULE | 00115-1248-08 | 6.91670 | EACH | 2026-03-18 |
| CARVEDILOL ER 10 MG CAPSULE | 16714-0227-01 | 6.91670 | EACH | 2026-03-18 |
| CARVEDILOL ER 20 MG CAPSULE | 00115-1249-08 | 6.39794 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CARVEDILOL ER Market Analysis and Financial Projection
Market Overview for Carvedilol ER
Carvedilol Extended Release (ER), marketed under brand names such as Coreg CR, is a beta-blocker primarily indicated for heart failure, hypertension, and post-myocardial infarction management. It is a prolonged-release formulation designed for once-daily dosing, improving patient adherence compared to immediate-release options.
The global beta-blocker market, valued at approximately $8.2 billion in 2022, is projected to grow at a Compound Annual Growth Rate (CAGR) of around 3.2% through 2030. Carvedilol constitutes a significant share of this market, driven by its dual alpha- and beta-adrenergic blocking activity.
Market Drivers
-
Rising prevalence of heart failure and hypertension: According to the World Health Organization, hypertension affects over 1.2 billion adults globally, with increasing incidence linked to aging populations.
-
Shift towards extended-release formulations: ER formulations offer improved compliance, especially in chronic disease management, and are favored in new prescriptions.
-
Approval and formulary inclusion: Carvedilol ER has gained favorable positioning in major healthcare systems, promoting higher usage rates.
Competitive Landscape
| Product Name | Formulation | Market Share (Estimated) | Key Competitors | Approved Indications |
|---|---|---|---|---|
| Coreg CR | Extended-release | 45% | Metoprolol Succinate ER, Nebivolol | Hypertension, heart failure, post-MI |
| Other branded | Immediate-release | 35% | Atenolol, Bisoprolol | Hypertension, angina |
| Generics | Both formulations | 20% | Various | Broad use in hypertension, heart failure, ischemic heart disease |
The dominant market position of Coreg CR remains stable; however, generic options erode margins and market share over time.
Regulatory Status and Patent Landscape
- Patent Status: Coreg CR patents expired or are nearing expiration in major markets (e.g., US expiry around 2025). Manufacturers may face generic competition subsequently.
- Regulatory Approvals: Approved in the US (FDA), EU (EMA), and other markets. The approval process for biosimilars is less relevant as carvedilol is a generic small molecule.
Price Trends and Projections
Current Pricing (USD per tablet):
| Region | Price Range for 20 mg ER Tablet | Comments |
|---|---|---|
| United States | $0.75 – $1.20 | Market varies by pharmacy |
| European markets | €0.65 – €1.00 | Price regulation influences |
| Asia-Pacific | $0.30 – $0.80 | Generally lower prices |
Post-patent expiration, price reductions of 40-60% are typical within 1-2 years in developed markets. Generic prices are often 50-70% lower than brand-name. Price erosion affects overall revenue projections significantly.
Future Trends:
- As generics penetrate markets, average prices for carvedilol ER are projected to decline 10-15% annually over the next 3-5 years.
- Price stabilization occurs as market maturation and formulary coverage improve.
Revenue and Market Share Projections (2023-2030)
| Year | Estimated Global Market (USD millions) | Carvedilol ER Revenue (USD millions) | Notes |
|---|---|---|---|
| 2023 | 300 | 150 | Dominant in its niche; growth driven by new indications |
| 2025 | 350 | 140 | Patent expiry influences decline in revenue; generics gain share |
| 2027 | 400 | 120 | Continued generic competition reduces revenue |
| 2030 | 430 | 100 | Market saturation and price erosion limit growth |
Revenue declines are anticipated post-2025 due to patent expiry and generic competition, though overall market growth sustains revenues.
Strategic Considerations
- Innovation: Development of combination therapies or novel delivery systems could sustain margins.
- Pricing strategies: Manufacturers may utilize differential pricing to retain market share in emerging markets.
- Regulatory navigation: Expedited approvals of biosimilars or regional variants could influence future dynamics.
Key Takeaways
- The carvedilol ER market is stable but faces compression due to patent expirations.
- The overall market is expected to grow modestly, with significant pricing declines expected in response to generic entry.
- The drug's biosimilar or generic versions will drive price reductions, pressuring branded formulations.
- Despite revenue declines, carvedilol ER remains a staple in hypertension and heart failure therapy.
- Companies focusing on alternative delivery methods or combination products may sustain competitiveness.
FAQs
1. When will generic carvedilol ER enter the market?
Most patents expire around 2025 in key markets, enabling generic manufacturers to begin producing cheaper versions shortly thereafter.
2. How does price erosion impact profitability?
A 50-70% price reduction typically accompanies generic entry, significantly diminishing profit margins for branded versions.
3. What are the key regulatory hurdles?
Generic approval usually relies on bioequivalence studies; biosimilar pathways are less relevant due to carvedilol being a small molecule.
4. How does market competition influence pricing strategies?
Intense competition among generics drives prices down, with some manufacturers adopting aggressive price cuts to gain market share.
5. What are future growth avenues for carvedilol ER?
Innovations in combination therapies, extended indications, and regional pricing strategies can offset revenue losses from patent expiration.
References
[1] MarketWatch, "Global Beta-Blockers Market Size, Share & Trends Analysis Report," 2022.
[2] WHO, "Hypertension Fact Sheet," 2022.
[3] IQVIA, "Pharmaceutical Market Data," 2022.
[4] FDA, "Drug Approvals and Patent Information," 2022.
More… ↓
