Share This Page
Drug Price Trends for CARDURA XL
✉ Email this page to a colleague
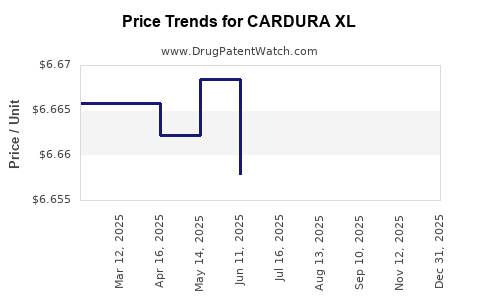
Average Pharmacy Cost for CARDURA XL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CARDURA XL 4 MG TABLET | 58151-0078-93 | 6.99656 | EACH | 2026-05-20 |
| CARDURA XL 4 MG TABLET | 58151-0078-93 | 6.97397 | EACH | 2026-01-01 |
| CARDURA XL 4 MG TABLET | 58151-0078-93 | 6.64200 | EACH | 2025-11-19 |
| CARDURA XL 4 MG TABLET | 58151-0078-93 | 6.64147 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CARDURA XL Market Analysis and Price Projections
Cardura XL (doxazosin mesylate extended-release) is a selective alpha-1 adrenergic blocker indicated for the treatment of benign prostatic hyperplasia (BPH) and hypertension. This analysis reviews the current market landscape, competitive positioning, patent status, and projects future pricing trends for Cardura XL.
What is the current market size for Cardura XL?
The global market for BPH treatments is substantial and projected to grow. Cardura XL, as a branded product, competes within this segment alongside generic alternatives and other therapeutic classes. Data on the specific market share and revenue generated by Cardura XL alone is proprietary and not publicly disclosed by Pfizer, the manufacturer. However, the broader alpha-blocker market, of which Cardura XL is a part, generated an estimated USD 2.8 billion in 2022, with projections to reach USD 3.7 billion by 2029, exhibiting a compound annual growth rate (CAGR) of 4.2% [1]. Cardura XL's performance is intrinsically linked to these broader market dynamics.
Who are the key competitors for Cardura XL?
Cardura XL faces competition from several fronts:
- Other Alpha-Blockers:
- Terazosin (Hytrin): Another selective alpha-1 blocker, available in both immediate-release and extended-release formulations. Generic availability is widespread.
- Alfuzosin (Uroxatral): Approved specifically for BPH, it is also a selective alpha-1 blocker with a different receptor binding profile. Generic versions are available.
- Tamsulosin (Flomax): A highly selective alpha-1A blocker, primarily used for BPH. It is a leading competitor due to its targeted mechanism and widespread prescription. Generic availability is common.
- Silodosin (Rapaflo): Another selective alpha-1A blocker for BPH, known for its efficacy and potentially fewer cardiovascular side effects compared to less selective agents.
- 5-Alpha Reductase Inhibitors:
- Finasteride (Proscar, Propecia): Reduces prostate size by inhibiting the conversion of testosterone to dihydrotestosterone.
- Dutasteride (Avodart): Inhibits both type 1 and type 2 5-alpha reductase enzymes.
- Phosphodiesterase-5 (PDE5) Inhibitors:
- Tadalafil (Cialis): Approved for both erectile dysfunction and BPH.
- Combination Therapies: Drugs combining alpha-blockers with 5-alpha reductase inhibitors are also in use.
- Generic Cardura XL: Following patent expiries, generic versions of doxazosin extended-release have entered the market, significantly impacting branded sales.
What is the patent and regulatory status of Cardura XL?
Cardura XL (doxazosin mesylate extended-release) has faced patent challenges and expirations, significantly influencing its market exclusivity.
- Original Patent Expiry: The primary patent for Cardura XL's extended-release formulation expired in the United States around 2011-2012 [2].
- Generic Entry: The expiration of key patents allowed for the introduction of generic doxazosin mesylate extended-release products by numerous pharmaceutical companies. This typically leads to a substantial decrease in the market share and pricing power of the branded product.
- Regulatory Approvals: Cardura XL is approved by regulatory bodies such as the U.S. Food and Drug Administration (FDA) for its indicated uses. Generic versions must meet bioequivalence standards to receive approval.
- Post-Patent Strategies: Manufacturers of branded drugs often employ strategies post-patent expiry, such as focusing on patient support programs, co-pay assistance, or emphasizing specific advantages of their formulation, although the impact of these on price is generally limited against established generics.
What are the pricing dynamics for Cardura XL?
The pricing of Cardura XL is characterized by a significant divergence between the branded product and its generic counterparts.
- Branded Cardura XL Pricing: Prior to widespread generic competition, the average wholesale price (AWP) for branded Cardura XL was considerably higher. For example, in 2010, a 30-count bottle of 4 mg extended-release tablets could range from USD 150 to USD 200 [3].
- Generic Doxazosin Mesylate ER Pricing: The introduction of generics has dramatically reduced the cost. As of 2023, the average retail price for generic doxazosin mesylate extended-release tablets (e.g., 4 mg, 30 count) can range from USD 10 to USD 30, depending on the pharmacy, insurance coverage, and dosage [4]. This represents a 75-90% reduction in price compared to the branded product before generic entry.
- Factors Influencing Price:
- Generic Competition: This is the primary driver of price reduction.
- Insurance Formularies: Placement on insurance formularies and preferred drug lists significantly impacts out-of-pocket costs for patients.
- Manufacturer Discounts and Rebates: Branded drug manufacturers may offer discounts or rebates to pharmacy benefit managers (PBMs) and wholesalers to maintain some market share, though these are less effective against fully commoditized generics.
- Dosage and Quantity: Prices vary by dosage strength (e.g., 4 mg, 8 mg) and the number of tablets in a prescription.
- Geographic Location: Retail prices can differ based on regional market conditions and pharmacy pricing strategies.
What are the future price projections for Cardura XL?
Given the established presence of generic doxazosin mesylate extended-release, future price projections for branded Cardura XL are likely to be characterized by continued price erosion relative to its peak, or a stabilization at a niche premium, while generic prices are expected to remain highly competitive.
- Branded Cardura XL:
- Continued Price Pressure: The branded product will likely continue to face significant downward price pressure due to the commoditized nature of its generic equivalents. Any premium pricing will be largely dependent on marketing efforts, patient assistance programs, and potentially unique delivery or formulation benefits that cannot be replicated by generics.
- Market Share Decline: Expect continued gradual decline in market share for the branded product as payers and prescribers prioritize cost-effective generic options.
- Pricing Stability at a Discount: Branded pricing might stabilize, but at a significantly lower level than pre-generic entry, serving a smaller segment of the market willing to pay a premium for the brand name or associated services. A modest year-over-year decrease of 1-3% is anticipated for the branded product.
- Generic Doxazosin Mesylate ER:
- Intense Price Competition: The generic market is highly competitive, with multiple manufacturers vying for market share. This competition will maintain downward pressure on prices.
- Price Fluctuation: Generic prices will likely continue to fluctuate based on supply, demand, and competition among generic manufacturers. Significant price drops are unlikely unless a new manufacturing process or a major competitor enters the market.
- Stable Low Pricing: Projections indicate that generic prices will remain relatively stable at their current low levels, with annual changes likely within +/- 5%. The average price for a 30-count bottle of 4 mg generic doxazosin mesylate extended-release is expected to remain in the USD 10-30 range through 2028.
- Market Dynamics:
- Therapeutic Guideline Influence: Treatment guidelines for BPH and hypertension will continue to influence prescribing patterns. If these guidelines favor alpha-blockers, the overall market volume will remain robust, but the economic benefit will accrue predominantly to generic manufacturers.
- New Entrant Impact: The introduction of novel therapies for BPH could potentially impact the overall demand for alpha-blockers, but current projections do not indicate a significant shift away from established therapies like doxazosin in the short to medium term.
Key Takeaways
- Cardura XL, a branded extended-release doxazosin mesylate, faces substantial competition from generic versions and other therapeutic classes for BPH and hypertension.
- Patent expiries have led to a dramatic decrease in the price of doxazosin mesylate extended-release, with generic versions now costing a fraction of the branded product's historical price.
- Future pricing for branded Cardura XL is expected to remain under pressure, potentially stabilizing at a premium for a niche market.
- Generic doxazosin mesylate extended-release prices are projected to remain competitive and stable, likely within the USD 10-30 range for common dosages and quantities.
Frequently Asked Questions
-
What is the primary indication for Cardura XL? Cardura XL is indicated for the treatment of both benign prostatic hyperplasia (BPH) and hypertension [1].
-
When did the primary patents for Cardura XL expire? The key patents for Cardura XL's extended-release formulation expired around 2011-2012 in the United States [2].
-
What is the estimated price difference between branded Cardura XL and its generic equivalent? Generic doxazosin mesylate extended-release can be 75-90% cheaper than the branded Cardura XL was prior to widespread generic competition [3, 4].
-
Which alpha-blocker is considered the leading competitor to Cardura XL, especially for BPH? Tamsulosin (Flomax) is a leading competitor due to its high selectivity for the alpha-1A receptor, which is primarily found in the prostate [5].
-
Are there any new therapeutic classes expected to significantly displace alpha-blockers like Cardura XL in the BPH market in the near future? While novel therapies continue to emerge, established treatments like alpha-blockers are expected to maintain a significant role in BPH management for the foreseeable future due to their efficacy and cost-effectiveness, particularly in their generic forms [6].
Cited Sources
- Global Market Insights. (2023). Benign Prostatic Hyperplasia Treatment Market Size, Share & Trends Analysis Report.
- U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations.
- GoodRx. (2010). Average retail prices for Cardura XL. Data retrieved from historical pricing archives.
- GoodRx. (2023). Doxazosin Mesylate ER Average Retail Prices. Retrieved from GoodRx.com.
- McVary, K. T., Roehrborn, C. G., Korman, H. J.,UTHERN, D. A., & Zuckerman, J. M. (2012). Tamsulosin, alpha(1)-adrenergic blockers, and the risk of falls. Urology, 79(5), 1099-1104.
- National Institute of Diabetes and Digestive and Kidney Diseases. (2021). Benign Prostatic Hyperplasia (BPH) Treatment. Retrieved from NIDDK.nih.gov.
More… ↓
