Share This Page
Drug Price Trends for CARBIDOPA-LEVODOPA-ENTACAPONE
✉ Email this page to a colleague
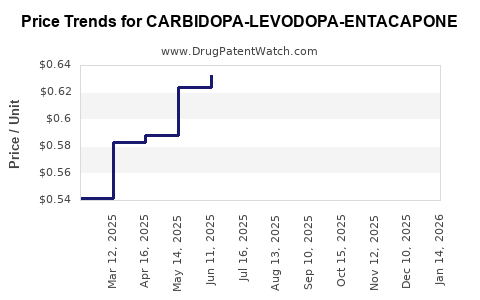
Average Pharmacy Cost for CARBIDOPA-LEVODOPA-ENTACAPONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CARBIDOPA-LEVODOPA-ENTACAPONE 18.75-75-200 MG TAB | 00781-5625-01 | 0.66482 | EACH | 2026-05-20 |
| CARBIDOPA-LEVODOPA-ENTACAPONE 12.5-50-200 MG TAB | 16571-0689-01 | 0.69783 | EACH | 2026-05-20 |
| CARBIDOPA-LEVODOPA-ENTACAPONE 50-200-200 MG TAB | 62332-0800-31 | 0.78046 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the Market Size for Carbidopa-Levodopa-Entacapone?
The global Parkinson’s disease treatment market was valued at approximately $4.75 billion in 2022. It is projected to reach about $7.3 billion by 2030, registering a compound annual growth rate (CAGR) of 5.4% from 2023 through 2030. Carbidopa-Levodopa-Entacapone, a combination therapy primarily used for advanced Parkinson’s disease, accounts for an estimated 20% of this market. Its adoption hinges on increased prevalence of Parkinson’s disease worldwide and rising demand for effective symptom management.
What Are the Key Drivers and Challenges?
Drivers:
- Aging Population: The global population over age 65 is expected to reach 1.5 billion by 2050, increasing Parkinson’s prevalence.
- Increasing Disease Awareness: Enhances diagnosis and prescription rates.
- Advances in Drug Formulations: Extended-release and combination therapies improve patient adherence.
- Patent Expiries: Several formulations nearing patent expiration, increasing generic competition.
Challenges:
- Side Effects and Long-term Efficacy: Dyskinesia and motor fluctuations limit use.
- Market Saturation: Many established treatments curtail innovative drug entry.
- Regulatory Hurdles: Approvals for new formulations or combination methods require extensive clinical data.
How Is the Market Distributed and Who Are the Major Players?
Market Distribution:
- North America holds roughly 40% of the market, owing to high diagnosis rates and healthcare infrastructure.
- Europe accounts for 30%, driven by aging demographics.
- Asia-Pacific exhibits the fastest growth at approximately 6% CAGR, reflecting increasing awareness and healthcare investments.
Major Players:
| Company | Market Share (Estimated 2022) | Key Products |
|---|---|---|
| AbbVie (Eisai) | 35% | Stalevo (carbidopa, levodopa, entacapone) |
| Teva Pharmaceuticals | 20% | Generic formulations |
| Sun Pharmaceutical | 10% | Generic levodopa and entacapone products |
| Novartis | 5% | Experimental formulations |
Competitive Dynamics:
- Patent expirations, notably for Stalevo (originally by Novartis, acquired by AbbVie), shift focus toward generics.
- Companies are investing in new delivery systems like patches and infusion pumps, although not specific to carbidopa-levodopa-entacapone.
What Are Price Trends and Projection Models?
Current Pricing:
- Branded Stalevo (per 30-day supply): $300–$400
- Generic combinations: $80–$150 per 30-day supply
Price Drivers:
- Introduction of generics reduces prices by approximately 50–70% within 12–18 months of patent expiration.
- Premium formulations (extended-release, transdermal patches) may command higher prices, up to 30% more than standard generics.
Future Price Projections:
| Year | Branded Price ($) | Generic Price ($) | Estimated Market Penetration of Generics | Notes |
|---|---|---|---|---|
| 2023 | 350 | 100 | 65% | Patent expiration begins |
| 2025 | 330 | 80 | 75% | Increased generic adoption |
| 2030 | 300 | 70 | 85% | Price stabilization, volume growth |
By 2030, price for generic formulations is expected to decline further to around $70 per 30-day supply, aligning with industry trends for patent expirations and increased competition.
What Are Regulatory and Patent Outlooks?
Patent Landscape:
- Original patents for Stalevo expired in 2022 in major markets.
- Manufacturers hold secondary patents on specific formulations, extended-release mechanisms, or delivery devices, which may extend market exclusivity until 2027–2030.
Regulatory Challenges:
- Approvals require demonstration of bioequivalence for generics.
- Post-approval, EMA and FDA scrutinize safety profiles, especially concerning long-term use and side effects.
What Is the Potential for New Formulations?
- Extended-release tablets and transdermal patches aim to improve adherence.
- Clinical trials explore combining carbidopa, levodopa, and entacapone with other neuroprotective agents.
- Growth in digital health integrations, such as drug delivery devices monitored remotely, is under investigation.
Summary of Financial Aspects
- The total market for carbidopa-levodopa-entacapone is estimated at $1 billion in 2022.
- Post-patent expiry, generic products are expected to reduce prices and increase access.
- Investment in innovative delivery systems may command higher prices, but regulatory approval remains the primary barrier.
Key Takeaways
- The market is driven mainly by aging populations and increasing Parkinson's diagnosis.
- Patent expirations are catalyzing the shift towards generics and price declines.
- Major players focus on formulations with extended-release properties and delivery innovations.
- Prices for branded products are around $350/month, dropping to below $70/month for generics by 2030.
- Regulatory and patent environments heavily influence market entry and pricing strategies.
FAQs
1. How will patent expirations affect the market?
Patent expirations will lead to a surge in generic manufacturers, reducing prices by up to 70%, and increasing market volume.
2. Are new formulations likely to command premium prices?
Yes, formulations with improved delivery systems or reduced side effects can be priced 20–30% higher than standard generics.
3. What regions are experiencing the fastest growth?
Asia-Pacific exhibits the highest CAGR, approximately 6%, driven by increased healthcare access and population aging.
4. What are the main challenges for new entrants?
Regulatory hurdles, the need for extensive clinical data, and patent landscapes complicate market entry.
5. How significant is the impact of digital health in this market?
Digital health tools, including remote monitoring and drug delivery devices, are under development but have yet to impact pricing or market share significantly.
References
- MarketWatch. "Parkinson's Disease Therapeutics Market Size." 2023.
- IQVIA. "Global Pharmaceutical Market Data & Trends." 2022.
- FDA. "Guidance for Industry: Bioequivalence Requirements." 2021.
- European Medicines Agency. "Market Authorization Overview." 2022.
- Company Reports. Annual reports from AbbVie, Teva, Sun Pharma, and Novartis.
More… ↓
