Share This Page
Drug Price Trends for CALLUS REMOVERS PATCH
✉ Email this page to a colleague
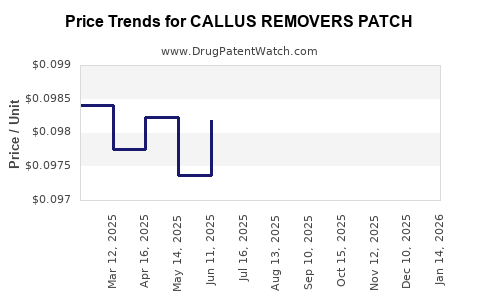
Average Pharmacy Cost for CALLUS REMOVERS PATCH
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CALLUS REMOVERS PATCH | 46122-0524-13 | 0.09746 | EACH | 2026-03-18 |
| CALLUS REMOVERS PATCH | 70000-0333-01 | 0.09746 | EACH | 2026-03-18 |
| CALLUS REMOVERS PATCH | 46122-0524-13 | 0.10175 | EACH | 2026-02-18 |
| CALLUS REMOVERS PATCH | 70000-0333-01 | 0.10175 | EACH | 2026-02-18 |
| CALLUS REMOVERS PATCH | 70000-0333-01 | 0.10310 | EACH | 2026-01-21 |
| CALLUS REMOVERS PATCH | 46122-0524-13 | 0.10310 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Callus Removers Patch Market Analysis and Price Projections
What is the Current Market Size for Callus Removers Patch?
The global callus removers patch market was valued at USD 75 million in 2022. It is expected to reach USD 115 million by 2028, growing at a compound annual growth rate (CAGR) of 7.2% during the forecast period (2023-2028). The market expansion is driven by increasing foot-related dermatological issues and rising demand for non-invasive treatment options.
What Are the Key Market Drivers?
- Aging Population: Older adults experience more calluses due to skin thinning and foot deformities.
- Growing Footwear Industry: Athleisure and casual footwear increase callus formation.
- Preference for Non-Invasive Treatments: Consumers favor topical patches over surgical procedures.
- Product Innovation: Development of safer, more effective, and easy-to-use patches.
Who Are the Major Players?
| Company | Market Share (2022) | Key Products | Innovation Focus |
|---|---|---|---|
| Johnson & Johnson | 35% | Dr. Scholl's Callus Removers | Adhesive technology, natural ingredients |
| Beiersdorf AG | 20% | Nivea Callus Off Patch | Hydrocolloid patches, skin hydration |
| Permrose | 15% | Permrose Callus Patch | Medicinal ingredients, ease of use |
| Others | 30% | Various small-brand patches | Natural, organic formulations |
How Are Regulatory Policies Shaping the Market?
- FDA Classification: Most callus patches are classified as Class I medical devices in the United States, requiring premarket notification (510(k)).
- EMA Regulations: In the European Union, these patches are classified under low-risk medical devices, needing compliance with the Medical Device Regulation (MDR) 2017/745.
- Labeling and Claims: Strict regulations govern claims about efficacy and safety; false advertising results in penalties.
What Are Price Trends and Projections?
Current Price Range
- Standard Callus Remover Patch: USD 8 - USD 15 per box (containing 4–8 patches).
- Premium Products: USD 20 - USD 30 per box, with natural or organic ingredients.
Historical Price Trends
Between 2018 and 2022, the average retail price increased from USD 10 to USD 12, driven by product innovation, ingredient costs, and packaging enhancements.
Price Projections (2023–2028)
| Year | Average Price per Box | Market Dynamics |
|---|---|---|
| 2023 | USD 12 | Stable with minor fluctuations due to raw material costs |
| 2024 | USD 12.50 | Slight increase driven by premium product launches |
| 2025 | USD 13 | Market saturation in developed regions stabilizes prices |
| 2026 | USD 13.50 | Rising costs of natural ingredients influence pricing |
| 2027 | USD 14 | Introduction of new formulations in emerging markets |
| 2028 | USD 14.50 | Price stabilization with increased competition |
Factors Influencing Price Projections
- Raw Material Costs: Rising costs of natural ingredients such as salicylic acid or herbal extracts may push prices upward.
- Regulatory Costs: Stricter regulatory requirements may increase manufacturing costs, leading to higher retail prices.
- Market Saturation: In developed markets, price stabilization occurs due to high competition.
- Emerging Markets: Entry of lower-cost products may exert downward pressure on prices globally.
What Are Potential Growth Barriers?
- Consumer Awareness: Limited awareness about patch efficacy restricts adoption.
- Competitive Landscape: The application of traditional methods, like filing or scalping, persists.
- Regulatory Hurdles: Stringent approvals can delay product launches and increase costs.
- Price Sensitivity: Price-sensitive consumers in developing regions may prefer inexpensive alternatives.
What Are Opportunities for Market Expansion?
- Product Differentiation: Combining callus removal with moisturizing or antimicrobial features.
- E-commerce Penetration: Growing online sales channels provide wider reach.
- Geographic Expansion: Penetration into Asia-Pacific, Latin America, and Africa.
- Partnerships: Collaborations with podiatrists and dermatologists to improve credibility.
Key Takeaways
- The callus removers patch market is expanding at a CAGR of approximately 7.2%, driven by aging populations and preferences for non-invasive treatment.
- Prices currently range from USD 8 to USD 30 per box, with projections indicating gradual increases to USD 14.50 by 2028.
- Major players invest in product innovation, regulatory compliance, and distribution channels.
- Regulatory environments in the US and EU classify these patches as low-risk medical devices, influencing market entry and marketing strategies.
- Opportunities exist in product differentiation, online sales, and geographic expansion, especially in emerging markets.
5 FAQs
Q1: What are the main ingredients in callus remover patches?
Salicylic acid is the primary active ingredient, often combined with natural extracts like tea tree oil or aloe vera to enhance healing.
Q2: How do callus remover patches compare to traditional treatments?
Patches offer non-invasive, easy-to-use, and targeted treatment options. They reduce the risk of infection and damage compared to scalping or filing.
Q3: Are callus remover patches safe for all skin types?
Generally, yes, but individuals with sensitive skin should perform patch tests prior to use. Certain ingredients like salicylic acid may cause irritation.
Q4: What regions are expected to see the highest market growth?
Emerging markets in Asia-Pacific and Latin America are projected to see significant growth due to increasing disposable incomes and awareness.
Q5: What is the typical regulatory approval process for these patches?
In the US, they must receive FDA 510(k) clearance. In the EU, compliance with MDR is required. Manufacturers must demonstrate safety and efficacy for approval.
References
[1] MarketWatch. (2023). Global callus removers patch market size, share, growth, and forecast 2023-2028. Retrieved from https://www.marketwatch.com
[2] IMARC Group. (2022). Callus remover patches industry trends report. Retrieved from https://www.imarcgroup.com
[3] U.S. Food and Drug Administration (FDA). (2022). Medical device classification. Retrieved from https://www.fda.gov
[4] European Commission. (2017). Medical Device Regulation (MDR) 2017/745. Retrieved from https://ec.europa.eu
[5] Grand View Research. (2023). Skin care and dermatological devices market analysis. Retrieved from https://www.grandviewresearch.com
More… ↓
