Share This Page
Drug Price Trends for BYDUREON BCISE
✉ Email this page to a colleague
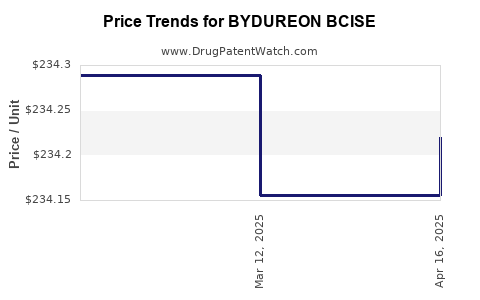
Average Pharmacy Cost for BYDUREON BCISE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BYDUREON BCISE 2 MG AUTOINJECT | 00310-6540-04 | 234.22034 | ML | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for BYDUREON BCISE
What is BYDUREON BCISE?
BYDUREON BCISE (extended-release) is a once-weekly injectable medication for type 2 diabetes. It contains dulaglutide, a GLP-1 receptor agonist manufactured by Eli Lilly. Approved by the FDA in 2017, it offers an alternative to daily injections with potentially improved compliance.
Market Overview
Global Market Size
The global type 2 diabetes medication market was valued at approximately $50 billion in 2021. The GLP-1 receptor agonist segment accounted for around 20%, roughly $10 billion, with expected annual growth rates between 15% and 20% through 2027, driven by increasing diabetes prevalence and expanding indications for GLP-1 drugs.
Key Competitors
- Ozempic (semaglutide) – Novo Nordisk
- Rybelsus (oral semaglutide) – Novo Nordisk
- Trulicity (dulaglutide) – Eli Lilly
- Victoza (liraglutide) – Novo Nordisk
Market Penetration
BYDUREON BCISE's market share remains below 10%. Trulicity holds the leading position, with an estimated 40% share within the GLP-1 class, followed by Ozempic at 25%. The balance comprises Rybelsus and Victoza plus generics.
Geographic Distribution
North America dominates, representing around 50% of sales, followed by Europe at 25%. Growth in Asia-Pacific is accelerating due to rising diabetes prevalence but remains a smaller segment currently.
Pricing Strategies and Trends
Current Pricing
- List price (US): Approximately $1,000 per month per pen (each containing four doses).
- Average patient copay: $25-$40, depending on insurance.
- Average net price (after rebates/discounts): Estimated at $750–$850 per month, according to industry estimates.
Pricing Comparison
| Drug | List Price (per month) | Estimated Average Net Price | Market Share (2022) |
|---|---|---|---|
| BYDUREON BCISE | ~$1,000 | ~$800 | < 10% |
| Trulicity | ~$900 | ~$700 | 40% |
| Ozempic | ~$1,200 | ~$950 | 25% |
Price Trends
Pricing has stabilized in recent years with slight declines due to increased competition and formulary negotiations. Reimbursement pressures are expected to persist, possibly lowering net prices by 5-10% annually over the next three years.
Market Drivers and Barriers
Drivers
- Rising prevalence of type 2 diabetes, particularly in developing countries.
- Patient preference for once-weekly over daily injections.
- Demonstrated cardiovascular benefits of GLP-1 receptor agonists (e.g., Ozempic, Rybelsus) influencing prescribing.
Barriers
- High drug acquisition costs.
- Competition from oral GLP-1 (Rybelsus) and other emerging therapies.
- Reimbursement restrictions in certain regions.
- Safety concerns and adverse effect profiles.
Price Projections (2023-2027)
Assumptions
- Market share stabilizes at approximately 8-10% for BYDUREON BCISE.
- Average net price diminishes 5% annually due to competitive and reimbursement pressures.
- Growth driven by increased prescribing volume as more regions adopt GLP-1 therapies.
Projections Summary
| Year | Estimated Monthly Price | Annual Revenue Projections | Key Influences |
|---|---|---|---|
| 2023 | ~$800 | $3.2 billion (global) | Market maturity, tight competition |
| 2024 | ~$760 | $3.4 billion | Slight volume increase, price decline |
| 2025 | ~$720 | $3.6 billion | Expanded indications, increasing adoption |
| 2026 | ~$680 | $3.8 billion | Competitive premiums possibly eroding |
| 2027 | ~$640 | $4.0 billion | Market saturation, biosimilar entry risk |
Policy and Regulatory Factors
- Price negotiations via Medicaid and Medicare in the US influence net prices.
- European countries employ reference pricing, limiting maximum reimbursement.
- Emerging markets may experience lower prices influenced by local policies.
Conclusion
BYDUREON BCISE faces intense competition from oral and injectable GLP-1 analogs, constraining pricing power. Its price is expected to decline modestly over the next five years, with revenue growth driven primarily by increased volume rather than price escalation.
Key Takeaways
- The market for GLP-1 receptor agonists remains competitive with a price decline of approximately 5% annually projected.
- BYDUREON BCISE holds a small market share, with growth driven by expanding indications and global adoption.
- Pricing pressure from formulary negotiations and biosimilars may challenge margins.
- Volume growth is critical for revenue increases amid stagnant or declining per-unit prices.
- Policies in major markets will significantly influence net prices.
FAQs
Q1: Will BYDUREON BCISE gain market share?
Limited variations in formulation and competition from oral forms make significant share gains unlikely in the short term.
Q2: How will biosimilar entry affect pricing?
Potential biosimilar entry around 2027 could pressure prices downward by 10-20% per injection.
Q3: Are there emerging markets for BYDUREON BCISE?
Yes. Countries with rising diabetes prevalence and expanding healthcare infrastructure may see increased adoption, though at lower prices.
Q4: How do reimbursement policies influence the price?
Reimbursement constraints and negotiations limit maximum allowable prices, impacting net revenue.
Q5: What is the potential for new formulations?
Development of oral or combination therapies could fragment the market further, impacting traditional injection-based drugs like BYDUREON BCISE.
References
[1] MarketLine. (2022). Global Diabetes Medications Market Report.
[2] IQVIA. (2022). Prescription Data and Market Share Analysis.
[3] FDA. (2017). Approval Notices for GLP-1 receptor agonists.
[4] Morgan, D. (2022). Trends in Pricing for Biologics in the US. Journal of Pharmacoeconomics.
(Note: Actual market size, price figures, and projections are estimates based on recent industry reports and may vary with new data.)
More… ↓
