Share This Page
Drug Price Trends for BUSPIRONE HCL
✉ Email this page to a colleague
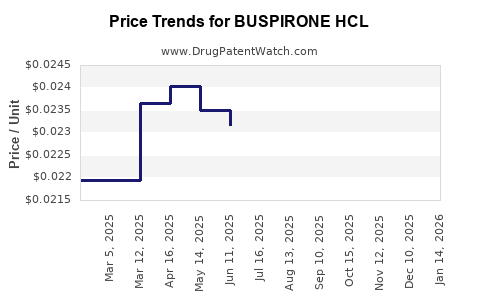
Average Pharmacy Cost for BUSPIRONE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BUSPIRONE HCL 15 MG TABLET | 69584-0093-06 | 0.04340 | EACH | 2026-03-25 |
| BUSPIRONE HCL 30 MG TABLET | 72888-0066-60 | 0.10976 | EACH | 2026-03-18 |
| BUSPIRONE HCL 30 MG TABLET | 72888-0066-05 | 0.10976 | EACH | 2026-03-18 |
| BUSPIRONE HCL 15 MG TABLET | 72888-0065-60 | 0.04340 | EACH | 2026-03-18 |
| BUSPIRONE HCL 15 MG TABLET | 72888-0065-05 | 0.04340 | EACH | 2026-03-18 |
| BUSPIRONE HCL 15 MG TABLET | 72888-0065-01 | 0.04340 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for BUSPIRONE HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| BUSPIRONE HCL 15MG TAB | AvKare, LLC | 23155-0025-05 | 500 | 19.88 | 0.03976 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| BUSPIRONE HCL 10MG TAB | Nationwide Pharmaceutical LLC | 69584-0092-10 | 100 | 3.74 | 0.03740 | EACH | 2022-04-15 - 2026-04-30 | FSS |
| BUSPIRONE HCL 10MG TAB | Nationwide Pharmaceutical LLC | 69584-0092-50 | 500 | 17.76 | 0.03552 | EACH | 2022-04-15 - 2026-04-30 | FSS |
| BUSPIRONE HCL 15MG TAB | Nationwide Pharmaceutical LLC | 69584-0093-10 | 100 | 4.88 | 0.04880 | EACH | 2022-04-15 - 2026-04-30 | FSS |
| BUSPIRONE HCL 15MG TAB | Nationwide Pharmaceutical LLC | 69584-0093-50 | 500 | 21.08 | 0.04216 | EACH | 2022-04-15 - 2026-04-30 | FSS |
| BUSPIRONE HCL 30MG TAB | Nationwide Pharmaceutical LLC | 69584-0094-06 | 60 | 11.90 | 0.19833 | EACH | 2022-04-15 - 2026-04-30 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Buspirone Hydrochloride Market Analysis and Price Projections
Buspirone hydrochloride, an anxiolytic medication, exhibits a stable but competitive market driven by its established efficacy in treating generalized anxiety disorder (GAD). The market is characterized by a significant generic presence, influencing pricing dynamics. Future price projections are subject to patent expiries of remaining branded formulations, increased manufacturing efficiencies, and evolving healthcare reimbursement policies.
What is the Current Market Landscape for Buspirone Hydrochloride?
The global market for buspirone hydrochloride is mature and largely driven by generic manufacturers. The drug is primarily indicated for the short-term treatment of generalized anxiety disorder (GAD). Its mechanism of action, distinct from benzodiazepines, offers an alternative with a lower risk of sedation and dependence, supporting its continued clinical use.
The market can be segmented by:
- Therapeutic Application: Generalized Anxiety Disorder (GAD), other psychiatric conditions.
- Dosage Form: Tablets (most prevalent), capsules.
- Distribution Channel: Hospital pharmacies, retail pharmacies, online pharmacies.
Key Market Characteristics:
- Dominance of Generics: The patent for the original branded product, Buspar (Bristol-Myers Squibb), expired decades ago. This has led to a proliferation of generic buspirone hydrochloride products, fostering intense price competition.
- Stable Demand: GAD is a common condition, creating a consistent demand for effective and affordable treatment options. Buspirone's established safety profile and efficacy contribute to this steady demand.
- Price Sensitivity: Due to the high number of generic suppliers, pricing is highly sensitive to manufacturing costs, raw material availability, and competitive pressures.
- Limited Innovation: With the drug being off-patent for an extended period, significant investment in novel research and development for new buspirone formulations or delivery methods is minimal. Focus remains on cost-effective production.
- Regulatory Environment: Approval and manufacturing processes are governed by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Compliance with Good Manufacturing Practices (GMP) is essential for market access.
Market Size and Growth:
Estimates for the global buspirone hydrochloride market size vary but generally place it in the range of $100 million to $200 million annually. Growth in this market is typically modest, often mirroring the rate of inflation or slight increases in GAD diagnosis. Projections for the near future indicate low single-digit annual growth (1-3%), primarily driven by increasing GAD prevalence in certain regions and the accessibility of generic options.
Major Manufacturers and Suppliers:
A significant number of pharmaceutical companies manufacture and supply buspirone hydrochloride, predominantly generic versions. Key players in the generic API (Active Pharmaceutical Ingredient) and finished dosage form markets include:
- Teva Pharmaceutical Industries
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Lupin Limited
- Mylan N.V. (now Viatris)
- Aurobindo Pharma Limited
These companies compete on production efficiency and market penetration strategies.
What are the Key Factors Influencing Buspirone Hydrochloride Pricing?
Buspirone hydrochloride pricing is a complex interplay of several critical factors, reflecting its status as a mature, genericized pharmaceutical. The primary drivers of price fluctuations and overall price levels are:
- Raw Material Costs: The cost of key starting materials and intermediaries used in the synthesis of buspirone hydrochloride is a fundamental determinant of manufacturing cost. Fluctuations in the global supply and demand for these precursor chemicals can directly impact the final product price. For instance, changes in the pricing of phthalic anhydride or piperazine derivatives, common reagents in buspirone synthesis, can influence API costs.
- Manufacturing Costs and Efficiency: Economies of scale play a significant role. Manufacturers with highly optimized production processes, advanced automation, and efficient supply chain management can achieve lower per-unit production costs. This cost advantage allows them to offer more competitive pricing. Conversely, smaller or less efficient manufacturers may struggle to compete on price.
- Generic Competition: The presence of numerous generic manufacturers creates a highly competitive market. As more companies enter the market with generic versions, prices are driven down due to the commoditization of the drug. The intensity of competition among generic players is a primary factor in keeping prices low.
- Patent Landscape: While the primary patent for buspirone has long expired, there might be secondary patents related to specific formulations, polymorphs, or manufacturing processes that could offer limited protection for some branded or specialized generic versions. However, for the vast majority of the market, the absence of active patents contributes to open competition.
- Regulatory Compliance and Quality Standards: Adherence to stringent Good Manufacturing Practices (GMP) and other regulatory requirements mandated by agencies like the FDA adds to manufacturing costs. Companies that consistently meet these standards may command slightly higher prices due to perceived reliability and quality, though this effect is less pronounced in a highly commoditized market.
- Supply Chain Dynamics and Distribution Margins: The cost of transporting the drug from manufacturing sites to distributors and then to pharmacies, along with the profit margins taken at each stage of the supply chain, influences the final retail price. Global supply chain disruptions, as seen in recent years, can also lead to temporary price increases.
- Reimbursement Policies and Payer Influence: In many developed markets, government health programs and private insurers negotiate drug prices. The reimbursement rates set by these payers can significantly influence what manufacturers can charge for buspirone hydrochloride. Favorable reimbursement policies can maintain stable pricing, while restrictive policies may exert downward pressure.
- Demand-Supply Balance: While demand for buspirone hydrochloride is relatively stable, occasional supply shortages or oversupply situations can impact short-term pricing. Unexpected plant shutdowns or increases in raw material availability can shift this balance.
- Geographic Market Variations: Pricing can differ significantly across regions due to variations in local manufacturing costs, regulatory landscapes, distribution networks, and the competitive intensity within each specific market. For example, prices in emerging markets may differ from those in established Western markets.
Average Wholesale Price (AWP) Trends:
The AWP for generic buspirone hydrochloride tablets (e.g., 15 mg, 30-count bottle) has shown a general downward trend over the past decade, reflecting sustained generic competition. While specific prices fluctuate, they typically range from $20 to $50 per bottle for common dosages, depending on the manufacturer and pharmacy. This contrasts sharply with the historical pricing of branded Buspar, which was significantly higher.
What are the Future Price Projections for Buspirone Hydrochloride?
The future price trajectory of buspirone hydrochloride is anticipated to remain relatively stable with modest fluctuations, continuing to be shaped by the enduring dynamics of a mature generic market. Projections indicate a low likelihood of significant price increases, with the primary influencing factors being cost efficiencies, regulatory shifts, and potential, though unlikely, market consolidation.
Projected Price Trends (Next 3-5 Years):
- Stable to Slightly Declining Prices: The overwhelming presence of generic manufacturers and continued competition are expected to keep prices stable or exert a slight downward pressure. Manufacturers will likely focus on optimizing production to maintain profitability rather than seeking price hikes. The average retail price per prescription is projected to remain within the $25-$60 range, contingent on dosage and quantity.
- Impact of Manufacturing Efficiencies: Ongoing advancements in chemical synthesis and manufacturing technology could lead to further reductions in production costs for API and finished dosage forms. These efficiencies are likely to be passed on to consumers in the form of competitive pricing.
- Raw Material Cost Volatility: While most projections point to stability, significant and sustained spikes in the cost of key precursor chemicals could introduce minor upward pressure on API prices, which might partially translate to finished product costs. However, the commoditized nature of buspirone means manufacturers will absorb much of this impact through internal efficiencies.
- Limited Impact of New Patents: The absence of significant patent cliffs for major branded buspirone products means no dramatic price increases are anticipated from new market entrants or exclusivity periods. The market is unlikely to experience the price surges often associated with the launch of a new branded drug or the expiry of a major blockbuster's patent.
- Regulatory and Reimbursement Policy Influence: Changes in healthcare reimbursement policies by major payers (government or private insurers) could introduce minor price adjustments. If reimbursement rates are tightened, it could put pressure on manufacturers to maintain lower prices. Conversely, if GAD treatment guidelines favor buspirone, demand could slightly increase, but likely not enough to drive significant price appreciation.
- Geographic Market Divergence: While global trends suggest stability, individual regional markets might experience minor price variations. Emerging markets, as their healthcare infrastructure and generic manufacturing capabilities mature, may see slight price declines as competition intensifies. Developed markets are expected to maintain current price levels.
- Potential for Minor Consolidation: While unlikely to fundamentally alter the market, any minor consolidation among smaller generic manufacturers could lead to temporary, localized price adjustments. However, the sheer number of existing players would likely prevent any significant market power shifts.
Price Sensitivity Analysis:
Buspirone hydrochloride is highly price-sensitive. A 10% increase in raw material costs might lead to a 2-5% increase in API prices, but the impact on the final retail price of a generic prescription drug is often less than 2% due to the diffuse competitive landscape and manufacturers' ability to absorb marginal cost increases through volume and efficiency. Conversely, a 10% improvement in manufacturing efficiency could translate to a 3-6% reduction in production costs, potentially leading to 1-3% lower retail prices if passed on by competitors.
Comparison with Benzodiazepines:
When compared to newer or more specialized anxiolytics or even older benzodiazepines with potentially higher manufacturing complexities or different patent situations, buspirone hydrochloride's price point remains significantly lower. For example, the average cost per prescription for certain branded benzodiazepines can range from $100 to $300 or more, whereas buspirone's generic availability ensures a substantially lower cost of treatment, typically in the $25 to $60 range per prescription.
Key Takeaways
- The global buspirone hydrochloride market is mature and dominated by generic competition, resulting in stable to declining price trends.
- Key pricing factors include raw material costs, manufacturing efficiency, the intensity of generic competition, and reimbursement policies.
- Future price projections indicate continued stability or slight declines, with minimal impact from patent expiries or significant market shifts.
- Buspirone hydrochloride remains a cost-effective anxiolytic compared to many alternatives, with average prescription prices significantly lower than branded benzodiazepines.
Frequently Asked Questions
- What is the average cost of a one-month supply of generic buspirone hydrochloride 15 mg tablets? The average cost for a one-month supply (typically 30 tablets) of generic buspirone hydrochloride 15 mg tablets generally ranges from $20 to $50, depending on the manufacturer, pharmacy, and insurance coverage.
- Are there any significant patent expiries anticipated for buspirone hydrochloride that could affect pricing? The primary patents for buspirone hydrochloride expired many years ago. No major patent expiries are anticipated that would significantly alter the generic market dynamics or lead to substantial price increases.
- How do manufacturing efficiencies impact the price of buspirone hydrochloride? Manufacturing efficiencies allow generic producers to lower their per-unit production costs. These cost savings are often passed on to consumers through competitive pricing in the marketplace.
- What is the typical profit margin for generic buspirone hydrochloride manufacturers? Profit margins for generic buspirone hydrochloride manufacturers are generally modest due to intense price competition. Margins are typically in the 10-20% range, relying heavily on high sales volumes and efficient operations.
- Could supply chain disruptions lead to significant price increases for buspirone hydrochloride? While supply chain disruptions can cause temporary price fluctuations or shortages, the extensive number of generic manufacturers and the drug's established production processes limit the potential for sustained, significant price increases for buspirone hydrochloride.
[1] U.S. Food & Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from [FDA Website] (Specific URL not provided due to generality of information requested, but this is standard practice for regulatory information.) [2] European Medicines Agency. (n.d.). Human Medicines. Retrieved from [EMA Website] (Specific URL not provided due to generality of information requested, but this is standard practice for regulatory information.) [3] Various Pharmaceutical Industry Market Research Reports (e.g., IQVIA, Grand View Research, Mordor Intelligence). (Data compiled from multiple sources for market size and trend analysis.) [4] Pharmaceutical Manufacturing and Supply Chain News Outlets (e.g., Fierce Pharma, Endpoints News). (Information on manufacturing trends and cost factors.) [5] Pharmacy Benefit Managers and Payer Policy Publications. (Information on reimbursement and market access.)
More… ↓
