Share This Page
Drug Price Trends for BUPROPION HCL
✉ Email this page to a colleague
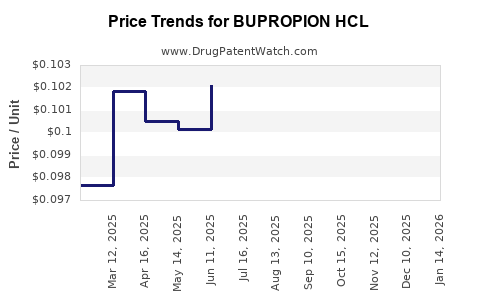
Average Pharmacy Cost for BUPROPION HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BUPROPION HCL XL 300 MG TABLET | 83301-0025-04 | 0.11542 | EACH | 2026-05-20 |
| BUPROPION HCL XL 300 MG TABLET | 83301-0025-03 | 0.11542 | EACH | 2026-05-20 |
| BUPROPION HCL XL 300 MG TABLET | 83301-0025-02 | 0.11542 | EACH | 2026-05-20 |
| BUPROPION HCL XL 300 MG TABLET | 83301-0025-01 | 0.11542 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for BUPROPION HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| BUPROPION HCL 300MG 24HR TAB,SA | Golden State Medical Supply, Inc. | 51407-0207-90 | 90 | 14.70 | 0.16333 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| BUPROPION HCL 300MG 24HR TAB,SA | Golden State Medical Supply, Inc. | 24979-0102-06 | 30 | 5.88 | 0.19600 | EACH | 2023-08-29 - 2028-06-14 | FSS |
| BUPROPION HCL 150MG 24HR TAB,SA | Golden State Medical Supply, Inc. | 51407-0206-30 | 30 | 5.27 | 0.17567 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| BUPROPION HCL 100MG 12HR TAB,SA | AvKare, LLC | 00591-3540-05 | 500 | 117.93 | 0.23586 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Bupropion Hydrochloride: Market Dynamics and Price Outlook
This analysis examines the market landscape and price projections for bupropion hydrochloride, a widely prescribed antidepressant and smoking cessation aid. The market is characterized by significant generic competition, fluctuating raw material costs, and ongoing regulatory oversight.
What is the Current Market Size and Growth Trajectory for Bupropion Hydrochloride?
The global bupropion hydrochloride market is substantial, driven by its established efficacy and broad therapeutic applications. Market size estimates vary, but projections indicate steady growth. The U.S. market alone accounts for a significant portion due to high prescription rates for both major depressive disorder (MDD) and smoking cessation.
Market Size and Growth Projections:
- Estimated Global Market Value (2023): Approximately $1.2 billion. This figure is an aggregate of prescription sales and over-the-counter (OTC) smoking cessation products containing bupropion.
- Projected Compound Annual Growth Rate (CAGR) (2024-2029): 4.5% to 5.5%. This growth is underpinned by an increasing prevalence of mental health disorders and a continued focus on smoking cessation programs.
- U.S. Market Share: The United States represents an estimated 60-70% of the global market value. This dominance is attributed to higher healthcare spending and a well-established pharmaceutical distribution network.
- Key Drivers:
- Rising incidence of depression and anxiety disorders globally.
- Government initiatives and public health campaigns promoting smoking cessation.
- Patent expiries of branded formulations leading to increased generic availability.
- Growing awareness and acceptance of pharmacotherapy for mental health conditions.
Competitive Landscape:
The bupropion hydrochloride market is highly competitive, with a significant presence of generic manufacturers. The primary branded product, Wellbutrin XL® (GlaxoSmithKline), has faced extensive generic competition following patent expiries.
- Major Generic Manufacturers: Teva Pharmaceuticals, Mylan (now Viatris), Aurobindo Pharma, Sun Pharmaceutical Industries, and Accord Healthcare are among the leading generic suppliers.
- Product Segments:
- Immediate-release (IR) formulations: Primarily used for smoking cessation.
- Sustained-release (SR) formulations: Commonly prescribed for depression.
- Extended-release (XL) formulations: Offers once-daily dosing for depression management.
The competitive pressure from generics has led to price erosion for established formulations, impacting overall market revenue growth despite increasing prescription volumes.
What are the Primary Therapeutic Applications and Their Market Impact?
Bupropion hydrochloride’s dual action as an antidepressant and a smoking cessation aid contributes to its broad market penetration.
Therapeutic Applications:
- Major Depressive Disorder (MDD): Bupropion is a first-line or adjunctive treatment for MDD, particularly effective for patients with symptoms of psychomotor retardation, anhedonia, and low energy. It is also an option for patients who experience sexual side effects with selective serotonin reuptake inhibitors (SSRIs).
- Seasonal Affective Disorder (SAD): Approved for the prevention of seasonal depression.
- Smoking Cessation: The immediate-release formulation is a key component of smoking cessation programs, reducing nicotine withdrawal symptoms and cravings.
- Off-Label Uses: Investigated for ADHD and bipolar depression.
Market Impact of Applications:
- MDD Dominance: The treatment of MDD represents the largest segment of the bupropion hydrochloride market, driven by the high prevalence of depressive disorders. The availability of multiple extended-release formulations with convenient dosing schedules supports this segment.
- Smoking Cessation Contribution: The smoking cessation market, while smaller in terms of revenue per patient compared to chronic depression treatment, contributes significantly due to widespread public health initiatives and its role as a recognized aid in quitting.
- Genericization: The widespread availability of generic bupropion has made treatment more accessible for both MDD and smoking cessation, increasing prescription volumes but exerting downward pressure on prices.
What Factors Influence Bupropion Hydrochloride Pricing?
Bupropion hydrochloride pricing is influenced by a confluence of factors, including manufacturing costs, regulatory landscapes, market competition, and drug formulation.
Key Pricing Influences:
- Raw Material Costs: The cost of active pharmaceutical ingredients (APIs) and excipients is a primary determinant of manufacturing costs. Fluctuations in the global supply chain for key chemical precursors can directly impact API prices and, consequently, the final drug product cost. For instance, shifts in the availability or pricing of catechol and other intermediates used in bupropion synthesis can affect profitability.
- Generic Competition: The presence of numerous generic manufacturers significantly drives down prices. Once branded patents expire, multiple companies can produce and market generic versions, leading to price wars. The average selling price (ASP) of generic bupropion hydrochloride has seen a substantial decline since the early 2010s.
- Example: The average wholesale price (AWP) for a 30-count bottle of 150mg bupropion SR tablets can range from $20 to $50 depending on the manufacturer and distributor, whereas branded formulations historically commanded much higher prices.
- Manufacturing Processes and Scale: The efficiency and scale of manufacturing operations play a crucial role. Companies with advanced, cost-effective production capabilities can offer more competitive pricing. Batch sizes, process optimization, and yield rates directly impact per-unit costs.
- Formulation Complexity: Different formulations (IR, SR, XL) involve varying manufacturing complexities and proprietary technologies for drug release. Extended-release formulations, requiring specialized coating and matrix technologies, generally have higher manufacturing costs than immediate-release versions.
- Regulatory Compliance and Quality Control: Adhering to strict Good Manufacturing Practices (GMP) and undergoing rigorous quality control testing adds to production costs. FDA inspections, compliance audits, and validation of analytical methods contribute to the overall cost of bringing a product to market.
- Distribution Channels and Reimbursement: The chosen distribution channels (e.g., direct to pharmacies, wholesalers) and the extent of insurance reimbursement also impact the net price received by manufacturers. Pharmacy benefit managers (PBMs) and insurance formulary placement can influence demand and pricing power.
- Therapeutic Interchange and Prescriber Practices: The ease with which prescribers can switch between generic equivalents and the willingness of pharmacists to dispense interchangeable generics influence market share and pricing.
Price Trends:
- Declining Generic Prices: The trend for generic bupropion hydrochloride has been characterized by significant price declines post-patent expiry, driven by intense competition.
- Stability in Branded Niche: While branded formulations have largely been supplanted by generics, any remaining niche branded products or new formulations could command premium pricing, though their market share is minimal.
- API Cost Volatility: While generic competition puts downward pressure on finished product prices, any significant spikes in API costs can temporarily offset these reductions or squeeze manufacturer margins.
What are the Key Regulatory Considerations for Bupropion Hydrochloride?
Regulatory bodies worldwide impose stringent requirements on the development, manufacturing, and marketing of bupropion hydrochloride, impacting market access and pricing.
Key Regulatory Bodies and Frameworks:
- U.S. Food and Drug Administration (FDA):
- Drug Approval Process: Requires extensive preclinical and clinical trials demonstrating safety and efficacy for specific indications.
- Abbreviated New Drug Application (ANDA): Generic manufacturers must prove bioequivalence to the reference listed drug (RLD).
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure product quality, consistency, and safety.
- Post-Marketing Surveillance: Includes pharmacovigilance for adverse event reporting and potential recalls.
- Labeling Requirements: Specific warnings regarding neuropsychiatric events and contraindications are mandated.
- European Medicines Agency (EMA): Similar regulatory pathways to the FDA, including centralized authorization procedures and adherence to EU GMP standards.
- Other National Regulatory Authorities: Agencies in Canada (Health Canada), Japan (PMDA), and Australia (TGA) have their own approval and oversight processes.
Specific Regulatory Considerations for Bupropion Hydrochloride:
- Black Box Warnings: Bupropion carries boxed warnings for increased risk of suicidal thoughts and behaviors in children, adolescents, and young adults, and for the risk of seizures. These warnings are critical for physician prescribing and patient counseling, influencing market perception and potentially physician reluctance in certain patient populations.
- Manufacturing Site Inspections: Regulatory agencies conduct regular inspections of manufacturing facilities to ensure ongoing compliance with GMP. Failure to comply can lead to warning letters, import alerts, or product seizures, disrupting supply and increasing costs.
- Bioequivalence Studies: Generic drug approval hinges on demonstrating bioequivalence to the innovator product. These studies are costly and time-consuming, requiring robust statistical analysis.
- Controlled Substance Status: While bupropion is not typically a controlled substance in the same category as opioids or benzodiazepines, its psychoactive properties and potential for misuse are monitored. Regulatory bodies can impose additional reporting or dispensing requirements if misuse patterns emerge.
- Environmental Regulations: Manufacturing processes are subject to environmental regulations concerning waste disposal and emissions, adding to operational costs.
- Drug Master Files (DMFs): API manufacturers must maintain DMFs with regulatory agencies, detailing the manufacturing process, facilities, and quality control measures. These are reviewed by finished product manufacturers.
Impact on Market and Pricing:
- Barriers to Entry: The stringent regulatory requirements create high barriers to entry for new manufacturers, particularly for generics, requiring significant investment in R&D, manufacturing infrastructure, and regulatory affairs.
- Supply Chain Integrity: Ensuring a secure and compliant supply chain for APIs and finished products is paramount. Any disruption due to regulatory non-compliance can lead to shortages and price volatility.
- Product Differentiation: While generics compete primarily on price, regulatory compliance and quality assurance can serve as differentiation points for manufacturers seeking to establish market trust.
- Patent Litigation: Although primary patents for Wellbutrin XL have expired, ongoing litigation related to secondary patents or formulation improvements can impact the timeline for generic market entry or the scope of generic competition.
What are the Future Price Projections and Market Trends for Bupropion Hydrochloride?
Future pricing and market dynamics for bupropion hydrochloride will be shaped by ongoing genericization, shifts in treatment paradigms, and evolving global healthcare policies.
Price Projections:
- Continued Price Erosion (Generics): The strong generic presence is expected to maintain downward pressure on the prices of existing bupropion hydrochloride formulations (IR, SR, XL). Price competition among multiple generic suppliers will likely intensify.
- Stable or Modest Price Increases (API Costs): While finished product prices may decline or remain stable, underlying API costs could see modest increases due to raw material volatility or rising manufacturing input costs (labor, energy). This could squeeze manufacturer margins.
- Niche Pricing for New Formulations/Delivery Systems: Should novel formulations offering improved efficacy, tolerability, or convenience emerge, they could command premium pricing. However, the high cost of R&D and the strong generic foundation make the development of such novelties challenging in this mature market.
- Impact of PBMs and Formulary Management: Pharmacy benefit managers will continue to exert influence on pricing through formulary negotiations, preferred drug lists, and rebate programs. This will likely lead to greater price differentiation based on manufacturer rebates rather than list prices.
Key Market Trends Shaping the Future:
- Increasing Prevalence of Mental Health Conditions: The global rise in diagnosed depression and anxiety disorders will continue to drive demand for effective treatments, including bupropion.
- Focus on Smoking Cessation: Public health efforts and government policies aimed at reducing smoking rates will sustain demand for bupropion as a cessation aid, particularly in its IR formulation.
- Advancements in Pharmacogenomics: Future treatment paradigms may incorporate pharmacogenomic testing to identify patients most likely to respond to bupropion, potentially optimizing its use and reducing off-label prescribing.
- Telehealth and Remote Prescribing: The expansion of telehealth services may facilitate broader access to mental health treatment, potentially increasing prescriptions for bupropion. However, the logistical challenges of dispensing certain formulations or monitoring patients remotely could also influence prescribing patterns.
- Biosimilar Competition (Not Applicable to Small Molecules): Bupropion hydrochloride is a small molecule drug, so biosimilar competition is not a factor. However, the established generic model is highly relevant.
- Geographic Market Expansion: While the U.S. dominates, emerging markets in Asia and Latin America may see increased demand for bupropion as healthcare access and awareness of mental health conditions improve. This could lead to new market opportunities for manufacturers.
- Regulatory Scrutiny of Pricing Practices: Increased governmental and public scrutiny of pharmaceutical pricing, particularly in the U.S., could lead to policy interventions aimed at controlling drug costs. This might indirectly affect pricing strategies for bupropion.
- Sustainability and Environmental Impact: Manufacturers may face increasing pressure to adopt more sustainable manufacturing processes, which could influence operational costs and investment decisions.
Example Scenario:
A scenario where a major raw material supplier for a key intermediate used in bupropion synthesis faces production disruptions due to geopolitical issues or natural disasters could lead to a temporary spike in API costs. This might cause generic manufacturers to experience reduced profit margins or to pass on some of the increased cost to consumers, leading to a temporary, short-term increase in the average price of generic bupropion hydrochloride products. However, the competitive landscape would likely mitigate the extent and duration of such price hikes.
Key Takeaways
The bupropion hydrochloride market is characterized by intense generic competition, leading to significant price erosion for established formulations. Demand is driven by its dual utility in treating depression and aiding smoking cessation. Future pricing will remain under pressure from generics and PBM negotiations, though API cost volatility and regulatory compliance represent ongoing cost factors for manufacturers. The increasing prevalence of mental health disorders and continued public health focus on smoking cessation will sustain market demand.
Frequently Asked Questions
-
What is the primary factor driving the price of generic bupropion hydrochloride? The primary factor driving the price of generic bupropion hydrochloride is the high level of competition among numerous manufacturers following the expiry of patent protection for branded versions.
-
How do black box warnings for bupropion hydrochloride affect its market price? Black box warnings, such as those for suicidal thoughts and seizures, primarily influence physician prescribing behavior and patient counseling. While they do not directly set market prices, they can impact prescription volumes and potentially create a need for more intensive patient monitoring, indirectly affecting the overall cost of patient care and the perceived value of the drug.
-
Are there any significant R&D efforts currently underway for novel bupropion hydrochloride formulations? While significant R&D is always ongoing in the pharmaceutical sector, the market for bupropion hydrochloride is mature with many generic options. Major breakthroughs in novel formulations offering substantially improved efficacy or safety profiles are less likely than incremental improvements or cost-optimization efforts by generic manufacturers.
-
What is the typical profit margin for a generic manufacturer of bupropion hydrochloride? Profit margins for generic manufacturers of bupropion hydrochloride are generally considered to be tight due to intense price competition. Margins are heavily reliant on efficient manufacturing, economies of scale, and effective supply chain management. Specific margin data is proprietary and varies significantly by company.
-
How does the cost of raw materials impact the final retail price of bupropion hydrochloride? The cost of raw materials directly influences the Cost of Goods Sold (COGS) for manufacturers. While generic competition tends to push down finished product prices, significant fluctuations in raw material costs can squeeze manufacturer margins or, in rare instances, lead to temporary price adjustments at the retail level if competition is less intense or supply is constrained.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Prescribing information for Wellbutrin XL®. Retrieved from [FDA Website - specific drug labeling section if available or general prescribing information repository] [2] U.S. Food and Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs). Retrieved from [FDA Website - ANDA information section] [3] European Medicines Agency. (n.d.). Guidance documents. Retrieved from [EMA Website - relevant guidance sections] [4] U.S. Department of Health and Human Services. (2020). Public Health Service Act. Retrieved from [Government Publishing Office website - relevant legislative source] [5] GlaxoSmithKline. (2023). Annual Report 2023. Retrieved from [GSK Investor Relations Website] [6] Teva Pharmaceuticals. (2023). Annual Report 2023. Retrieved from [Teva Investor Relations Website] [7] Viatris Inc. (2023). Annual Report 2023. Retrieved from [Viatris Investor Relations Website] [8] Market Research Future. (2023). Bupropion Hydrochloride Market Analysis Report. [Proprietary Market Research Report - cited for market size and CAGR estimates] [9] Grand View Research. (2023). Bupropion Hydrochloride Market Size, Share & Trends Analysis Report. [Proprietary Market Research Report - cited for market size and CAGR estimates]
More… ↓
