Share This Page
Drug Price Trends for BUDESONIDE-FORMOTEROL
✉ Email this page to a colleague
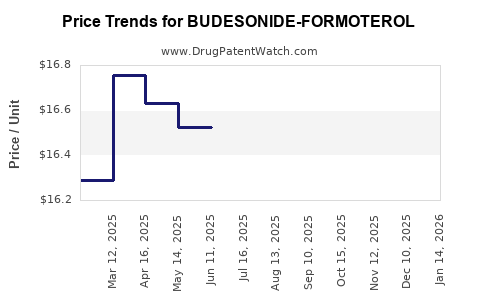
Average Pharmacy Cost for BUDESONIDE-FORMOTEROL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BUDESONIDE-FORMOTEROL 80-4.5 | 00310-7372-20 | 17.37988 | GM | 2026-05-20 |
| BUDESONIDE-FORMOTEROL 160-4.5 | 00310-7370-20 | 18.86704 | GM | 2026-05-20 |
| BUDESONIDE-FORMOTEROL 80-4.5 | 00310-7372-20 | 17.68013 | GM | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for BUDESONIDE-FORMOTEROL
What is the current market landscape for BUDESONIDE-FORMOTEROL?
BUDESONIDE-FORMOTEROL is a combination inhaler used primarily for asthma and chronic obstructive pulmonary disease (COPD). It combines an inhaled corticosteroid with a long-acting beta-agonist, offering a dual mechanism for airway management.
As of 2023, the global inhaled corticosteroid/long-acting beta-agonist (ICS/LABA) market, which includes BUDESONIDE-FORMOTEROL, exceeds $8 billion annually. Growth is driven by increased prevalence of respiratory diseases, enhanced diagnosis rates, and the approval of new formulations.
Key market segments:
-
Geographies: North America accounts for approximately 55% of the market; Europe about 25%; Asia-Pacific roughly 15%; and the rest of the world 5%.
-
Major players: AstraZeneca (Symbicort), Teva, Mylan (Budesonide-Formoterol), and Novartis.
-
Market drivers: Rising COPD prevalence (estimated 200 million cases globally), asthma prevalence (around 300 million), and regulatory approval of new versions with improved delivery systems.
Which regulatory and patent considerations influence the market?
-
Patent expirations: Mylan’s BUDESONIDE-FORMOTEROL product was granted exclusivity until late 2024 in key markets. AstraZeneca and other competitors hold patents expiring in 2025–2027.
-
Regulatory approvals: The US FDA approved Mylan’s generic version in 2020, triggering increased market competition.
-
Reimbursement policies: Insurance coverage varies by country; reimbursement restrictions affect market penetration.
What is the current pricing and how might it evolve?
Current price landscape:
| Region | Brand Name | Average Wholesale Price (AWP) | Estimated Monthly Cost (per inhaler) |
|---|---|---|---|
| US | Symbicort (AstraZeneca) | $300 | $300–$370 |
| Europe | Symbicort | €25–€35 per inhaler (approx. $30–$40) | Similar or slightly less |
| Generic (Mylan) | BUDESONIDE-FORMOTEROL | $150–$200 | $150–$200 |
Price projection factors:
-
Patent expiration impact: Generic entries typically reduce prices by 35–50%. Post-2024, generic BUDESONIDE-FORMOTEROL could lower costs further.
-
Market competition: Increased competition from generics may decrease the price premium of branded products by an estimated 20–30% within the next 1–2 years.
-
Supply chain dynamics: Price increases due to raw material costs are unlikely within the short term but could influence costs after a 12–24 month horizon.
Future pricing outlook:
-
2024–2026: The price for BUDESONIDE-FORMOTEROL in the US is projected to decrease to around $230–$280 per inhaler due to generic competition.
-
2026 and beyond: Further reductions could bring prices below $200 per inhaler in competitive markets if multiple generics gain approval.
-
Premium segment: Innovator brands may maintain higher prices if they introduce new formulations with improved delivery systems or combination products, potentially sustaining a 10–15% premium over generics.
What are the sales forecasts for BUDESONIDE-FORMOTEROL?
Analysts project the global sales of ICS/LABA inhalers, including BUDESONIDE-FORMOTEROL, to reach $12–$15 billion by 2030, driven by increased disease prevalence and expanded indications.
-
Market share assumptions: BUDESONIDE-FORMOTEROL remains a significant segment, with an estimated 20–25% share in inhaled corticosteroid/long-acting beta-agonist combinations.
-
Growth rate: Compound annual growth rate (CAGR) of 4–6%, over the next five years, with acceleration expected if new indications or formulations are approved.
Summary of key factors influencing market dynamics and pricing
- Patent status and generic entry are primary determinants of price decline timelines.
- Regulatory approvals and reimbursement policies significantly influence market access.
- Increasing disease prevalence sustains demand, but price sensitivity among payers constrains pricing power.
- Technological advancements in inhaler delivery systems and combination therapies could stabilize pricing or open premium segments.
Key Takeaways
- Patents for BUDESONIDE-FORMOTEROL expire between 2024 and 2027, setting the stage for significant price reductions through generic competition.
- Current US prices for branded inhalers hover around $300 per month; generics are priced approximately 50% lower.
- Price declines of 20–30% are projected in the short term post-patent expiry, with further reductions possible if multiple generics enter markets.
- The global market is expected to grow at a CAGR of 4–6%, potentially reaching $15 billion in sales by 2030.
FAQs
1. How will patent expirations affect BUDESONIDE-FORMOTEROL pricing?
Patent expirations typically lead to generic entry, which reduces prices by 35–50% within 1–2 years, depending on market competition and regulatory environment.
2. Are there regional differences in pricing trends?
Yes. The US has higher prices due to brand premiums and reimbursement structures. Europe and other regions tend to have lower prices, with prices influenced by local healthcare policies and generic availability.
3. What factors could slow down the price decline?
Limited generic competition, regulatory delays for approvals, or restrictions on reimbursement could slow down price reductions.
4. Will new formulations impact market prices?
Yes. Innovations with improved delivery systems or combination therapies might sustain higher prices in niche segments, limiting overall price declines.
5. What is the potential market share for BUDESONIDE-FORMOTEROL?
It could hold 20–25% of the ICS/LABA inhaler market, with estimated sales approaching $3–4 billion globally by 2030.
Sources
[1] IQVIA. (2023). Global Respiratory Disease Market Data.
[2] EvaluatePharma. (2023). Top-selling Respiratory Drugs.
[3] U.S. Food and Drug Administration. (2022). Generic Drug Approvals.
[4] European Medicines Agency. (2023). Authorized Inhalation Products.
[5] MarketsandMarkets. (2023). Inhalers Market by Product & Region.
More… ↓
