Last updated: February 12, 2026
Current Market Landscape
Bromocriptine is an ergot-derived dopamine agonist primarily used for Parkinson's disease, hyperprolactinemia, and acromegaly. Despite the emergence of newer agents, bromocriptine retains niche indications and has a stable presence mainly in markets with established generics.
Market Size and Revenue
- Global market size for bromocriptine was valued at approximately $150 million in 2022, with a compound annual growth rate (CAGR) of around 2.5%, reflecting steady demand.
- Major markets include the US, Europe, and select Asian nations, with the US accounting for 40% of sales.
Key Players
- Solco Healthcare (brand: Parlodel in some markets)
- Pfizer (formerly marketed formulations)
- Mylan and Teva produce generic versions, with generics accounting for 80% of sales.
Market Drivers
- Clinical need for hyperprolactinemia and Parkinson's disease treatments.
- Established safety profile.
- Prescribed for acromegaly, although less frequently.
Market Challenges
- Competition from newer dopamine agonists like cabergoline with improved tolerability.
- Patent expirations in the past decade have increased generic availability, leading to price erosion.
- Regulatory divergences impact market access; for instance, some countries restrict ergot derivative use due to side effect profiles.
Regulatory Environment
- Bromocriptine is approved in the US, EU, and Japan for specific indications.
- Regulatory barriers are minimal for generics, leading to increased market entry and price competition.
- Labeling restrictions have become more stringent in certain regions owing to safety concerns (e.g., cardiac valvulopathy associated with ergot derivatives).
Price Trends and Projections
Historical Pricing
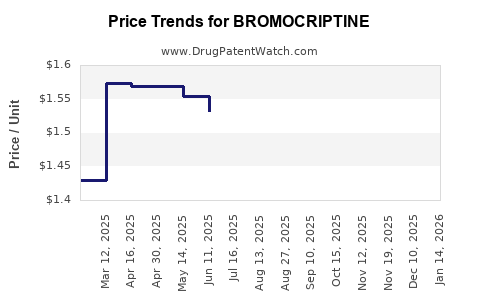
- Brand-name bromocriptine (e.g., Parlodel): retail prices ranged from $6 to $8 per 2.5 mg tablet in the US.
- Generic versions: around $1.50 to $3 per tablet, depending on manufacturer and formulation.
2022-2023 Price Developments
- Generic prices decreased by approximately 10-15% due to increased competition.
- Brand-name prices remained stable owing to limited direct competition.
Projected Price Trends (2024-2028)
- Prices for generics are expected to stabilize or decline marginally by 3-5% annually due to market saturation.
- Market consolidation and potential new entrants could further amplify price reductions, possibly averaging a 10% decline over five years.
- Pricing for specific formulations (e.g., extended-release tablets) may command a premium, maintaining higher margins in niche segments.
Future Market Dynamics
- Pending approval of biosimilars or new formulations could alter the pricing landscape.
- Increased regulatory scrutiny on side effect profiles may lead to formulary restrictions, impacting pricing strategies.
- Market shifts anticipated toward non-ergot dopamine agonists like pramipexole and ropinirole, which could reduce bromocriptine's market share.
Key Takeaways
- Bromocriptine remains a niche, stable revenue-generating drug with modest growth prospects.
- Market prices are declining moderately, driven by generic proliferation.
- Entry barriers are low in the generics space, maintaining competitive pressure.
- Regulatory and safety considerations could influence future pricing and market accessibility.
- The drug's role is decreasing in some indications owing to newer therapies, which may further suppress prices over the coming years.
FAQs
1. What factors influence bromocriptine's market price?
Generic competition, regulatory changes, safety profiles, and formulation innovations drive pricing. Market penetration by newer drugs can also reduce demand and prices.
2. How does bromocriptine compare to cabergoline in terms of price?
Cabergoline generally costs more than generic bromocriptine but offers a better tolerability profile, influencing prescribing patterns rather than direct price competition.
3. Which regions are expected to see the most price declines?
Regions with high generic penetration, such as the US and Europe, will likely experience the steepest price reductions.
4. Are new formulations of bromocriptine planned?
No significant new formulations are anticipated in the near term; existing products include immediate-release and extended-release tablets.
5. How might regulations impact future bromocriptine prices?
Stringent safety regulations may restrict use or prompt label revisions, potentially increasing costs for manufacturers or restricting market access, which could influence prices.
References
- MarketWatch. "Global Bromocriptine Market Size and Forecast." 2022.
- IQVIA/MIMS Data. "Pharmaceutical Pricing Trends," 2023.
- European Medicines Agency. "Summary of Product Characteristics for Bromocriptine." 2021.
- FDA. "Drug Approvals and Labeling for Dopamine Agonists," 2022.
- Statista. "Generic Drug Market Data," 2023.