Share This Page
Drug Price Trends for BROMFENAC SODIUM
✉ Email this page to a colleague
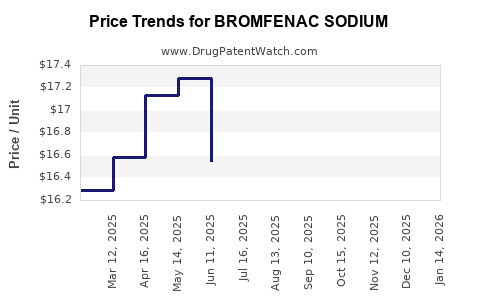
Average Pharmacy Cost for BROMFENAC SODIUM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BROMFENAC SODIUM 0.07% EYE DRP | 68180-0433-02 | 12.85173 | ML | 2026-05-20 |
| BROMFENAC SODIUM 0.07% EYE DRP | 62332-0583-03 | 12.85173 | ML | 2026-05-20 |
| BROMFENAC SODIUM 0.09% EYE DRP | 72266-0142-01 | 16.05726 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Bromfenac Sodium
What is Bromfenac Sodium?
Bromfenac sodium is a non-steroidal anti-inflammatory drug (NSAID) primarily used as an ophthalmic solution. It treats postoperative ocular inflammation and pain, particularly following cataract surgery. The medication inhibits cyclooxygenase enzymes, decreasing prostaglandin synthesis, leading to reduced inflammation.
Market Overview
Current Market Size
- Estimated global ophthalmic NSAID market was valued at approximately $450 million in 2022.
- Bromfenac sodium represented roughly 30% of this segment, translating to $135 million in revenue in 2022.
Key Players
- Akorn (subsidiary of Fresenius Kabi): Commercializes BromSite, a combination of bromfenac and dexamethasone.
- Hikma Pharmaceuticals: Markets Bromfenac in some territories.
- Alcon: Markets proprietary NSAID formulations with bromfenac as an active component.
- Generic manufacturers: Increasing presence in emerging markets.
Market Drivers
- Rising number of cataract surgeries.
- Greater adoption of NSAIDs as standard postoperative care.
- Growing awareness of postoperative inflammation management.
- Patent expirations on branded formulations expanding generic entry.
Market Challenges
- Competition from alternative NSAIDs (nepafenac, ketorolac, diclofenac).
- Patent protections on branded drugs.
- Regulatory scrutiny and approval processes in different jurisdictions.
- Pricing pressure due to generic competition.
Price Trends (Historical & Current)
- Brand-name Bromfenac (e.g., BromSite): Pricing ranges from $60 to $100 per 5 mL bottle.
- Generics: Prices drop to $20–$40 per bottle, with variations based on region.
- Market average: An average retail price for Bromfenac sodium ophthalmic solution is approximately $35 per bottle in the U.S. market.
Future Price Projections
Short-term (1-2 years)
- Expectations: Slight decline in average price due to increased generic competition.
- Projected price: $25–$35 per bottle in developed markets.
- Despite this, branded products may maintain premium pricing in exclusive contracts or hospital settings.
Medium-term (3-5 years)
- Market saturation: Higher penetration by generics could push prices down to $15–$25 per bottle.
- Innovations: Development of sustained-release formulations could command higher prices, potentially $50–$70 per dose**.
Long-term (5+ years)
- Price stabilization: As patents expire, prices may decline further, but innovative delivery systems could sustain premium pricing.
- Market evolution: Increased use in emerging markets and combination drugs could influence average prices upward or downward, depending on regional dynamics.
Market Share and Revenue Forecasts
| Year | Estimated Market Size | Bromfenac Sodium Revenue | Note |
|---|---|---|---|
| 2022 | $135 million | $135 million | Current market |
| 2025 | $500 million (global ophthalmic NSAID) | $150–$200 million | Growth driven by surgeries and generic entry |
| 2030 | $600 million | $180–$250 million | Expansion into emerging markets |
Regional Variations
- United States: Stable, high prices; reliance on branded drugs persists.
- Europe: Similar to the U.S., but with more aggressive generic penetration.
- Emerging markets (e.g., India, China): Prices significantly lower ($10–$20 per bottle); increasing adoption.
Regulatory and Patent Outlook
- Current patents on branded formulations are set to expire between 2024 and 2028.
- Pending approval of generic bromfenac formulations in multiple regions.
- Patent litigations may influence pricing and market dynamics.
Strategic Implications
- Entry of generics pressures pricing but broadens market access.
- Innovations in formulation delivery (such as sustained-release implants) could preserve premium pricing.
- Partnerships with surgical centers and healthcare providers offer growth avenues.
Key Takeaways
- The global ophthalmic NSAID market, including Bromfenac sodium, is expanding amid growing surgical procedures.
- Prices are declining in response to generic competition, with a 2022 average around $35 per bottle.
- Price projections indicate further reductions in the short term, with potential stabilization or increases resulting from new formulations or regional factors.
- Patent expirations from 2024 onward are critical turning points for market share and pricing.
- Regional variations significantly impact revenue and pricing strategies.
FAQs
1. What are the primary competitors to bromfenac sodium in the ophthalmic NSAID market?
Nepafenac, ketorolac, and diclofenac dominate as alternative NSAIDs in ophthalmology.
2. How does patent law affect bromfenac sodium pricing?
Patent expirations typically lead to increased generic competition, reducing prices and expanding access.
3. Will new formulations impact bromfenac sodium prices?
Innovations such as sustained-release devices can command higher prices but may initially increase R&D costs and market entry risk.
4. What regions present the most growth opportunities?
Emerging markets like India and China are expanding their ophthalmic surgical procedures, creating growth opportunities for bromfenac sodium.
5. How significant is the impact of regulatory approvals on market dynamics?
Regulatory approval processes influence entry timing for generics and new formulations, directly affecting prices and market share.
References
- MarketsandMarkets. (2022). Ophthalmic drugs market size.
- IQVIA. (2022). Ophthalmic NSAID market analysis.
- U.S. FDA. (2023). Patent status and approval timelines for ophthalmic drugs.
- EvaluatePharma. (2022). Pharmaceutical pricing and market access report.
- European Medicines Agency. (2023). Regulatory guidelines for ophthalmic products.
More… ↓
