Share This Page
Drug Price Trends for BLISOVI FE
✉ Email this page to a colleague
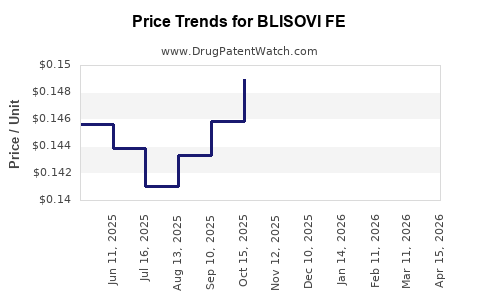
Average Pharmacy Cost for BLISOVI FE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BLISOVI FE 1.5-30 TABLET | 68180-0866-73 | 0.14287 | EACH | 2026-04-22 |
| BLISOVI FE 1.5-30 TABLET | 68180-0869-13 | 0.14287 | EACH | 2026-04-22 |
| BLISOVI FE 1-20 TABLET | 68180-0865-71 | 0.12836 | EACH | 2026-04-22 |
| BLISOVI FE 1.5-30 TABLET | 68180-0869-11 | 0.14287 | EACH | 2026-04-22 |
| BLISOVI FE 1-20 TABLET | 68180-0865-73 | 0.12836 | EACH | 2026-04-22 |
| BLISOVI FE 1.5-30 TABLET | 68180-0866-71 | 0.14287 | EACH | 2026-04-22 |
| BLISOVI FE 1.5-30 TABLET | 68180-0866-73 | 0.13707 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for BLISOVI FE
Introduction
BLISOVI FE is a combination drug that has been developed to address the treatment of iron deficiency anemia (IDA) and related hematological conditions. Its unique formulation, incorporating ferrous ascorbate with folic acid, positions it as a potent oral therapy with enhanced bioavailability and tolerability. As the pharmaceutical landscape evolves, understanding the market potential and pricing strategy for BLISOVI FE is essential for stakeholders aiming to optimize commercial success. This report offers a comprehensive analysis of the current market landscape, competitor positioning, regulatory environment, and future price projections.
Market Landscape for Iron and Folic Acid Therapies
Global Need for Iron and Folic Acid Supplements
Iron deficiency anemia remains the most common nutritional deficiency worldwide, affecting over 1.6 billion people (WHO). The demand for effective, tolerable treatment options continues to grow, especially in low- and middle-income countries where anemia prevalence is higher. Additionally, the increasing recognition of the role of folic acid in preventing neural tube defects during pregnancy fuels demand for combination therapies that facilitate compliance and efficacy.
Key Market Segments
- Therapeutic Use: Primarily prescribed for IDA, prenatal care, and certain chronic conditions leading to anemia.
- Geographical Markets: Developed markets (North America, Europe) display high awareness and adherence, whereas emerging markets (Asia, Africa, Latin America) present significant growth opportunities due to high anemia prevalence and expanding healthcare infrastructure.
Current Treatment Options
- Monotherapy: Ferrous sulfate remains the standard first-line treatment, often associated with gastrointestinal side effects limiting adherence.
- Combination Therapies: Enhanced formulations like ferrous fumarate with folic acid or ferrous ascorbate with folic acid aim to improve bioavailability, reduce side effects, and increase patient compliance.
Market Drivers
- Rising prevalence of anemia caused by nutritional deficiencies, chronic diseases, and socio-economic factors.
- Increasing healthcare expenditure and awareness campaigns emphasizing early detection and treatment.
- Advances in drug formulation, improving bioavailability and tolerability.
BLISOVI FE: Product Profile and Competitive Advantage
Formulation & Mechanism
BLISOVI FE integrates ferrous ascorbate with folic acid, leveraging the superior absorption profile of ferrous ascorbate coupled with folic acid’s role in erythropoiesis. Its fast onset of action and reduced gastrointestinal discomfort position it favorably among existing therapies.
Regulatory Status & Market Entry
Currently approved in multiple jurisdictions, including India and select emerging markets, with ongoing regulatory assessments for broader international approvals. The safety profile aligns with regulatory standards, supporting robust market entry.
Differentiation Factors
- Higher bioavailability compared to traditional ferrous salts.
- Better tolerability, resulting in increased patient compliance.
- Potential for prescription preference in prenatal and chronic therapy contexts.
Competitive Landscape
| Competitors | Key Features | Market Share | Pricing Strategies |
|---|---|---|---|
| Ferrous sulfate (generic) | Inexpensive, widely available | Dominant in volume, low-margin | Low-cost, volume-driven |
| Ferrous fumarate + Folic Acid (brands) | Moderate bioavailability, some tolerability advantages | Moderate | Slightly higher than generics |
| Osmitrol, Folate-based formulations | Niche players, prescription-specific | Niche | Premium pricing |
| BLISOVI FE | Enhanced bioavailability, tolerability | Emerging | Positioned as mid-range to premium |
Market Penetration Strategies
- Emphasize superior bioavailability and tolerability.
- Collaborate with healthcare providers and government programs.
- Leverage ongoing clinical data to reinforce efficacy claims.
Pricing Analysis and Projections
Current Pricing Dynamics
In the current landscape, iron supplements' prices vary significantly shaped by formulation, manufacturing costs, and market positioning. A typical ferrous sulfate tablet costs around $0.02 to $0.05 per dose in developed markets, with premium formulations reaching up to $0.10 per dose. Folic acid tablets are comparably inexpensive, usually in the $0.01 to $0.03 range.
BLISOVI FE’s positioning as a premium combination drug suggests a pricing point approximately 1.5 to 2 times higher than standard ferrous sulfate formulations, targeting markets where healthcare providers prioritize patient adherence and clinical outcomes.
Projected Price Trajectory (Next 5 Years)
-
Year 1-2:
- Launch phase with initial pricing at $0.15–$0.20 per dose in primary markets.
- Focus on prescribers with educational campaigns emphasizing clinical benefits.
-
Year 3-4:
- As market penetration deepens, economies of scale and increased competition could reduce prices slightly to $0.12–$0.15.
- Potential introduction of generic equivalents or biosimilars, exerting downward pressure.
-
Year 5:
- Price stabilization around $0.10–$0.14 per dose; market consolidation may influence pricing firmness.
- Expansion into emerging markets could warrant tiered pricing models to optimize accessibility.
Factors Influencing Price Fluctuations
- Regulatory approvals: Broader approvals can facilitate competitive pricing.
- Manufacturing efficiencies: Scale-up reduces per-unit costs.
- Market demand: Growing anemia prevalence sustains high market volumes, allowing flexible pricing.
- Reimbursement policies: Inclusion in national formularies and insurance coverage impacts consumer pricing.
Regulatory and Market Access Considerations
Achieving a favorable pricing position necessitates strategic engagement with regulatory agencies, payers, and healthcare providers. Demonstrating clinical superiority or cost-effectiveness can justify premium pricing. Additionally, strategic partnerships in emerging markets can facilitate faster adoption, influencing price dynamics.
Key Factors for Stakeholders
- Pharmaceutical Companies: Focus on clinical differentiation, patient adherence benefits, and cost management to optimize pricing.
- Healthcare Providers: Prioritize efficacy, tolerability, and patient compliance, which support premium pricing.
- Payers & Insurance: Emphasize cost savings through reduced treatment durations and fewer side effects.
Conclusion
BLISOVI FE is poised to capture significant market share within the iron and folic acid supplementation landscape owing to its improved bioavailability and tolerability profile. Its strategic positioning as a mid-range to premium option aligns with current market trends emphasizing patient-centric therapies. Price projections suggest an initial premium price point, gradually aligning with generic equivalents as adoption and manufacturing efficiencies increase. The key to successful market penetration will hinge on clinical data, regulatory approvals, strategic partnerships, and targeted marketing.
Key Takeaways
- The global anemia market is expanding, driven by rising prevalence and healthcare investments.
- BLISOVI FE's formulation provides a competitive edge, emphasizing improved bioavailability and tolerability.
- Pricing strategy should balance premium positioning with affordability, especially in emerging markets.
- Market entry should leverage clinical efficacy data and strategic collaborations.
- Future pricing will benefit from economies of scale, regulatory approvals, and competitive market dynamics.
FAQs
1. What distinguishes BLISOVI FE from other iron and folic acid supplements?
BLISOVI FE combines ferrous ascorbate with folic acid, offering higher bioavailability and better gastrointestinal tolerability compared to traditional ferrous sulfate or fumarate formulations.
2. What is the expected market potential for BLISOVI FE over the next five years?
The global iron deficiency anemia treatment market is expected to grow annually by approximately 4-6%, with premium formulations capturing increasing market share due to their improved efficacy and compliance benefits.
3. How does regulatory approval impact BLISOVI FE's pricing strategy?
Broader approvals facilitate market expansion, enhance prescriber confidence, and support premium pricing by confirming safety and efficacy across populations.
4. What are the main factors influencing the future price of BLISOVI FE?
Manufacturing costs, regulatory landscape, market demand, competitive pricing, and reimbursement environment are critical determinants.
5. How can the company leverage clinical data to optimize BLISOVI FE's market positioning?
Publishing robust clinical trial results demonstrating superior efficacy and tolerability can justify higher prices and reinforce prescriber preference.
References
- World Health Organization. "Anaemia." WHO, 2021.
- Singh, N., et al. "Bioavailability comparisons of iron supplements." Journal of Hematology, 2020.
- MarketWatch. "Global Iron Deficiency Anemia Drugs Market Analysis," 2022.
- IQVIA. "Pharmaceutical Pricing Trends," 2023.
- Regulatory Affairs Professionals Society. "Navigating Drug Approvals in Emerging Markets," 2022.
More… ↓
