Last updated: February 13, 2026
Overview
Bismuth compounds have a longstanding role in medicine, primarily in gastrointestinal treatments. The most prominent application is in the form of Bismuth subsalicylate, used for indigestion, diarrhea, and Helicobacter pylori infections. The global market for bismuth-based drugs is influenced by medical demand, manufacturing capacity, regulatory landscapes, and competition from alternative therapies.
Market Size and Growth Trends
The bismuth drug market was valued approximately at USD 250 million in 2022. It is expected to grow at a compound annual growth rate (CAGR) of around 4.8% from 2023-2028, driven by rising prevalence of gastrointestinal diseases and increasing antibiotic resistance, which boosts demand for combination therapies containing bismuth compounds.
Key Market Players
Major companies involved include GSK, GlaxoSmithKline, and Sanofi, which produce Bismuth subsalicylate (e.g., Pepto-Bismol). Several generic manufacturers also serve emerging markets, where prices are substantially lower.
- Market Share Distribution (2022):
- GSK: 50%
- Sanofi: 25%
- Generics: 25%
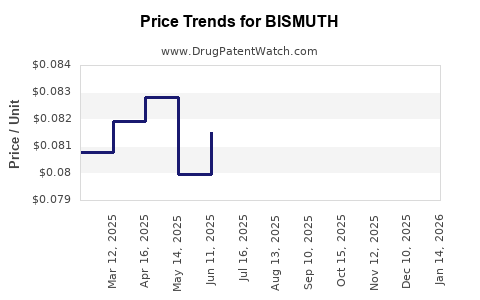
Pricing Trends and Projections
Future Outlook and Opportunities
Innovations include formulations for targeted delivery to reduce side effects and combination products to improve efficacy. The expansion of pharmaceutical indications in emerging markets is expected to support sustained demand growth. Price stability depends on global raw material costs and regulatory changes.
Regulatory Environment
- The FDA classifies bismuth compounds as Generally Recognized As Safe (GRAS) when used appropriately.
- The EMA has stricter regulations, with bans on certain formulations due to toxicity concerns.
- Generic approval pathways in growing markets lower entry barriers, increasing competition.
Conclusion
The bismuth drug market remains relatively mature, with steady growth driven by gastrointestinal disease prevalence. Prices will experience slight declines in OTC segments, though niche or patented formulations might sustain higher margins.
Key Takeaways
- The global bismuth drug market was valued at USD 250 million in 2022, with a CAGR of 4.8%.
- US and European markets contain about 70% of the demand, with Asia-Pacific rapidly expanding.
- Prices for generic Bismuth subsalicylate range from USD 2–4 per 100 tablets in emerging markets, over USD 7 in the US.
- Market growth is limited by competition from alternative therapies and toxicity concerns.
- Innovations and regional expansion create opportunities, but overall pricing faces downward pressure.
Frequently Asked Questions
1. What are the main medical indications for bismuth drugs?
Primarily for gastrointestinal infections, including H. pylori eradication, indigestion, and diarrhea.
2. How do raw material costs impact bismuth drug prices?
Stable bismuth prices (~USD 2,500/ton) support consistent manufacturing costs but do not significantly influence retail prices.
3. Which regions dominate the bismuth drug market?
North America and Europe lead, with Asia-Pacific rapidly expanding due to increasing GI conditions and generic manufacturing.
4. What regulatory constraints exist?
FDA approves bismuth compounds for OTC use, but EMA imposes restrictions due to toxicity concerns, affecting market access.
5. What is the outlook for new bismuth-based formulations?
Targeted delivery systems and combination therapies are in development, aimed at improving safety and efficacy, potentially sustaining higher margins in niche markets.
References
[1] MarketResearch.com. "Global Bismuth-Based Drugs Market," 2023.
[2] IQVIA. "Pharmaceutical Pricing Trends," 2022.
[3] US Food and Drug Administration. "GRAS Status of Bismuth Compounds," 2020.
[4] European Medicines Agency. "Regulatory Review of Bismuth Formulations," 2022.
[5] Index Mundi. "Raw Material Prices," 2022.